Abstract
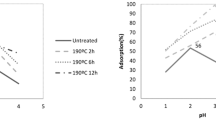
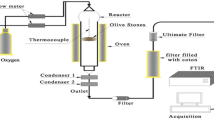
Partial pyrolysis alters the chemical and textural properties of the lignocellulosic material. This work reports the effect of partial pyrolysis of olive wood on adsorption isotherms, kinetics and thermodynamics of chloro and nitrophenols. Shape of adsorption isotherms of the partially pyrolyzed sorbents was L3 for phenol; L2 for 2-nitrophenol and 2,4-dinitrophenol; H3 for 2-chlorophenol, 3-chlorophenol and 4-nitrophenol; and H2 for 4-chlorophenol. The pyrolyzed olive wood sorbents obeyed Langmuir and Freundlich models. Pyrolysis raised adsorption capacity, favorability and spontaneity; the adsorption became more exothermic; the randomness decreased. The adsorption was mainly physical; it occurred first by film diffusion then by pore-filling. Adsorption followed second-order rate kinetics. Adsorption of phenols on olive wood seemed to be governed by hydrophobic interaction. Washing the pyrolyzed olive wood with ethanol caused a decrease in adsorption capacity, favorability and spontaneity, and the adsorption became less exothermic. This indicated that pyrolysis produced species on the olive wood surface that played a significant role in phenols adsorption.




Similar content being viewed by others
References
Abia AA, Horsfall M, Didi O (2003) The use of chemically modified and unmodified cassava waste for the removal of Cd, Cu and Zn ions from aqueous solution. Bioresour Technol 90:345–348
Ahmaruzzaman M (2008) Adsorption of phenolic compounds on low-cost adsorbents: a review. Adv Colloid Interface 143:48–67
Aravindhan R, Rao JR, Nair BU (2009) Application of a chemically modified green macro alga as a biosorbent for phenol removal. J Environ Manag 90:1877–1883
Bansode RR, Losso JN, Marshall WE, Rao RM, Portier RJ (2004) Pecan shell-based granular activated carbon for treatment of chemical oxygen demand (COD) in municipal wastewater. Bioresour Technol 94:129–135
Bayramoglu G, Gursel I, Tunali Y, Arica MY (2009) Biosorption of phenol and 2-chlorophenol by Funalia trogii pellets. Bioresour Technol 100:2685–2691
Brandt S, Zen AP, Deckwer WD (1997) Adsorption and desorption of pentachlorophenol on cells of Mycobacterium chlorophenolicum PCP-1. Biotechnol Bioeng 55:480–489
Bridgwater AV (1994) Thermogravimetric analysis of the components of biomass: advances in thermochemical biomass conversion. Blackie Academic and Professional, London
Cruz CCV, Da Costa ACA, Henriques CA, Luna AS (2004) Kinetic modeling and equilibrium studies during cadmium biosorption by dead Sargassum sp. Biomass. Bioresour Technol 91:249–257
de Hoces MC, de Castro FH, Garcia GB, Rivas G (2006) Equilibrium modeling of removal of cadmium ions by olive stones. Environ Prog 25:261–266
El-Sheikh AH, Newman AP, Al-Daffaee H, Phull S, Cresswell N (2004) Characterization of activated carbon prepared from a single cultivar of Jordanian olive stone by chemical and physicochemical techniques. J Anal Appl Pyrol 71:151–164
El-Sheikh AH, Alzawahreh AM, Sweileh JA (2011) Preparation of an efficient sorbent by washing then pyrolysis of olive wood for simultaneous solid phase extraction of chloro-phenols and nitro-phenols from water. Talanta 85:1034–1042
Fourest E, Volesky B (1997) Alginate properties and heavy metal biosorption by marine algae. Appl Biochem Biotechnol 67:215–226
Freundlich HMF (1906) Liber die adsorption in lo¨sungen. Phys Chem 57:385–470
Frimmel FH, Huber L (1996) Influence of humic substances on the aquatic sorption of heavy metals on defined minerals phases. Environ Int 22:507–517
Giles CH, Smith DA (1974) General treatment and classification of the solute sorption isotherms. J Colloid Interface 47:755–765
Grioui N, Halouani K, Zoulalian A, Halouani F (2007) Experimental study of thermal effect on olive wood porous structure during carbonization. Maderas Ciencia y tecnología 9:15–28
Hajaligol M, Waymack B, Kellogg D (2002) Pyrolysis behaviour and kinetics of biomass derived materials. J Anal Appl Pyrol 62:331–349
Ho YS, McKay G (2000) The kinetics of sorption of divalent metal ions onto sphagnum moss peat. Water Res 34:735–742
Igwe JC, Nwokennaya EC, Abia AA (2005) The role of pH in heavy metal detoxification by biosorption from aqueous solutions containing chelating agents. Afr J Biotechnol 4:1109–1112
Khan AR, Singh RP (1987) Adsorption thermodynamics of carbofuran on Sn(IV) arsenosilicate in H+, Na+ and Ca2+ forms. Colloid Surface A 24:33–42
Lagergren S (1898) Zur theorie der sogenannten adsorption geloester stoffe. K Sven Vetenskapsakad Handl 24:1–39
Langmuir I (1918) The adsorption of gases on plane surfaces of glass, mica, and platinum. JACS 40:1361–1368
Martin-Lara MA, Pagnanelli F, Mainelli S, Calero M, Toro L (2008) Chemical treatment of olive pomace: effect on acid-base properties and metal biosorption capacity. J Hazard Mater 156:448–457
Mathialagan T, Viraraghavan T (2008) Biosorption of chlorophenols: a review. Int J Environ Pollut 34:164–194
Nadavala SK, Swayampakula K, Boddu VM, Abburi K (2009) Biosorption of phenol and o-chlorophenol from aqueous solutions on to chitosan–calcium alginate blended beads. J Hazard Mater 162:482–489
Navarro AE, Portales RF, Sun-Kou MR, Llanos BP (2008) Effect of pH on phenol biosorption by marine seaweeds. J Hazard Mater 156:405–411
Overend RP, Chornet E (1999) 4th conference on biomass: a growth opportunity in green energy and value-added products. Pergamon-Elsevier Science, Oxford
Pan B, Xing B (2008) Adsorption mechanisms of chemicals on carbon nanotubes. Environ Sci Technol 42:9005–9013
Perez-Bonilla M, Salido S, van Beek TA, Linares-Palomino PJ, Altarejos J, Nogueras M, Sanchez A (2006) Isolation and identification of radical scavengers in olive tree (Olea europaea) wood. J Chromatogr A 1112:311–318
Raji C, Amirudhan TS (1998) Batch Cr(VI) removal by polyacrylamide-grafted saw dust: kinetics and thermodynamics. Water Res 32:3772–3780
Saka C, Sahin O, Küçük MM (2012) Applications on agricultural and forest waste adsorbents for the removal of lead (II) from contaminated waters. Int J Environ Sci Technol 9:379–394
Sanchez AG, Ayuso EA, De Blas OJ (1999) Sorption of heavy metals from industrial waste water by low-cost mineral silicates. Clay Miner 34:469–477
Shin EW, Karthikeyan KG, Tshabalala MA (2005) Orthophosphate sorption onto lanthanum-treated lignocellulosic sorbents. Environ Sci Technol 39:6273–6279
US Environmental Protection Agency (1988) Technology screening guide for treatment of CERCLA soils and sludges. Publication EPA/540/2-88/004, Washington DC
Vazquez G, Gonzalez-Alvarez J, Garcia AI, Freire MS, Antorrena G (2007) Adsorption of phenol on formaldehyde-pretreated Pinus pinaster bark: equilibrium and kinetics. Bioresour Technol 98:1535–1540
Weber WJ, Morris JC (1963) Kinetics of adsorption on carbon from solution. J Sanit Eng Div 89:31–39
Wu J, Yu H (2006) Biosorption of 2,4-dichlorophenol from aqueous solution by Phanerochaete chrysosporium biomass: isotherms, kinetics and thermodynamics. J Hazard Mater 137:498–508
Zghida H, Gauthier R, Helal A, Bartegi A (2006) Modeling of adsorption of anionic surfactants onto cationized lignocellulosic materials. J Appl Polym Sci 99:82–87
Acknowledgments
The authors are grateful for the Faculty of Graduate Studies and Scientific Research at the Hashemite University for supporting this project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
El-Sheikh, A.H. Partial pyrolysis of olive wood to improve its sorption of chlorophenols and nitrophenols. Int. J. Environ. Sci. Technol. 11, 1459–1472 (2014). https://doi.org/10.1007/s13762-013-0333-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-013-0333-x