Abstract
Introduction
Congenital myasthenic syndromes (CMS) are a heterogeneous group of genetic disorders affecting the safety factor which required for neuromuscular transmission. Here we reported our experience in children with CMS.
Methods
We retrospectively collected the data of 18 patients with CMS who were examined in our outpatient clinic between January 2021 and January 2022. The diagnosis of CMS was based on the presence of clinical symptoms such as abnormal fatigability and weakness, absence of autoantibodies against acetylcholine receptor and muscle-specific kinase, electromyographic evidence of neuromuscular junction defect, molecular genetic confirmation, and response to treatment.
Results
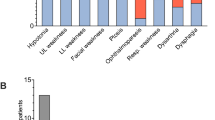
The most common mutations were in the acetylcholine receptor (CHRNE) gene (8/18) and choline acetyltransferase (ChAT) (2/18) gene. Despite targeted gene sequencing and whole exome sequencing (WES) were underwent, we couldn’t detect a genetic mutation in three out of patients. The most commonly determined initial finding was eyelid ptosis, followed by fatigable weakness, and respiratory insufficiency. Although the most commonly used drug was pyridostigmine, we have experienced that caution should be exercised as it may worsen some types of CMS.
Discussion
We reported in detail the phenotypic features of very rare gene mutations associated with CMS and our experience in the treatment of this disease. Although CMS are rare genetic disorder, the prognosis can be very promising with appropriate treatment in most CMS subtypes.
Similar content being viewed by others
References
Engel AG, Shen XM, Selcen D, Sine SM (2015) Congenital myasthenic syndromes: pathogenesis, diagnosis, and treatment. Lancet Neurol 14(4):420–434
Hacer D, Shen XM, Serdaroglu-Oflazer P et al (2018) Congenital myasthenic syndromes in Turkey: clinical clues and prognosis with long term follow-up. Neuromuscul Disord 28:315–322
McMacken G, Abicht A, Evangelista T, Spendiff S, Lochmu¨ ller H, (2017) The increasing genetic and phenotypical diversity of congenital myasthenic syndromes. Neuropediatrics 48:294–308
Abicht A, Dusl M, Gallenmuller C et al (2012) Congenital myasthenic syndromes: achievements and limitations of phenotype-guided gene-after-gene sequencing in diagnostic practice: a study of 680 patients. Hum Mutat 33(10):1474–1484
Pattrakornkul N, Ittiwut C, Boonsimma P et al (2020) Congenital myasthenic syndromes in the Thai population: clinical findings and novel mutations. Neuromuscul Disord 30:851–858
Yiş U, Becker K, Kurul S et al (2017) Genetic Landscape of congenital myasthenic syndroms from Turkey: novel mutations and clinical insights. J of Child Neurol 32(8):759–765
Estephan EP, Sobreira CFR, Dos Santos ACJ et al (2018) A common CHRNE mutation in Brazilian patients with congenital myasthenic syndrome. J Neurol 265:708–713
Oztürk A, Deymeer F, Serdaroğlu P et al (2003) Distribution of extremity muscle weakness in myasthenia gravis: sparing of tibialis anterior muscle. Acta Myologica Myopathies Cardiomyopathies: Official J Mediterr Soc Myol 22(2):58–60
Ramdas S, Beeson D (2021) Congenital myasthenic syndromes: where do we go from here? Neuromuscul Disord 31(10):943–954
McMacken G, Whittaker RG, Evangelista T, Abicht A, Dusl M, Lochmüller H (2018) Congenital myasthenic syndrome with episodic apnoea: clinical, neurophysiological and genetic features in the long-term follow-up of 19 patients. J Neurol 265(1):194–203
Huze C, Bauche S, Richard P et al (2009) Identification of an Agrin mutation that causes congenital myasthenia and affects synapse function. AJHG 85:155–167
Maselli RA, Fernandez JM, Arredondo J et al (2012) LG2 Agrin mutation causing severe congenital myasthenic syndrome mimics functional characteristics of non-neural (z-) Agrin. Hum Genet 131:1123–1135
Palace J, Lashley D, Newsom-davis J et al (2007) Clinical features of the DOK7 neuromuscular junction synaptopathy. Brain 130:1507–1515
Estephan EP, Zambon AA, Thompson R et al (2022) Congenital myasthenic syndrome: correlation between clinical features and molecular diagnosis. Eur J Neurol 29(3):833–842
Belaya K, Finlayson S, Slater C et al (2012) Mutations in DPAGT1 cause a limb-girdle congenital myasthenic syndrome with tubular aggregates. Am J Hum Genet 91:193–201
Dagher R, Massie R, Gentil BJ (2021) MTP deficiency caused by HADHB mutations: pathophysiology and clinical manifestations. Mol Genet Metabol 133(1):1–7
Liewluck T, Mundi MS, Mauermann ML (2013) Mitochondrial trifunctional protein deficiency: a rare cause of adult-onset rhabdomyolysis. Muscle Nerve 48:989–991
Nicolau S, Kao JC, Liewluck T (2019) Trouble at the junction: when myopathy and myasthenia overlap. Muscle Nerve 60:648–657
Li J, Suda K, Ueoka I et al (2019) Neuron-specific knockdown of Drosophila HADHB induces a shortened lifespan, deficient locomotive ability, abnormal motor neuron terminal morphology and learning disability. Exp Cell Res 379:150–158
Regal L, Martensson E, Maystadt I et al (2018) PREPL deficiency: delineation of the phenotype and development of a functional blood assay. Genet Med 20:109–118
LAU, Jarred. SYT11 as a Novel Gene in Congenital Myasthenic Syndromes. 2022. PhD Thesis. Université d'Ottawa/University of Ottawa.
GülMert G, Özcan N, Hergüner Ö et al (2021) Congenital myasthenic syndrome in Turkey: clinical and genetic features in the long-term follow-up of patients. Acta Neurol Belg 121(2):529–534
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare they have conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yildiz, E.P., Kilic, M.A., Yalcin, E.U. et al. Genetic and clinical evaluation of congenital myasthenic syndromes with long-term follow-up: experience of a tertiary center in Turkey. Acta Neurol Belg 123, 1841–1847 (2023). https://doi.org/10.1007/s13760-022-02090-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-022-02090-0