Abstract
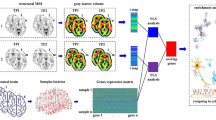
Deep brain stimulation of the anterior nucleus of the thalamus (ANT-DBS) has been shown to be effective and safe in the long-term treatment of refractory epilepsy. However, the mechanisms by which ANT-DBS controls epilepsy at the gene expression level (e.g., which regulatory mechanisms are altered) is not well understood. Nine rats were randomly assigned to the control group, the kainic acid (KA) group, and the DBS group. Temporal lobe epilepsy in rats was induced by a stereotaxic KA injection (KA group). The DBS group received the KA injection followed by treatment with ANT-DBS. Video-electroencephalogram (EEG) was used to monitor seizures. Total RNA samples were isolated from the hippocampus of three groups. Microarray was used to detect differentially regulated mRNAs. GO and pathway analysis were performed to analyze the functional categories and affected pathways. qPCR was used to prove the reliability of the microarray results. The differentially expressed genes the KA group and the DBS group, relative to the control group, were screened and a total of 2910 genes were identified. These genes were involved in functional categories such as ion channel activity (P = 5.01 × 10−8), gated channel activity (P = 1.42 × 10−7), lipid binding (P = 4.97 × 10−5), and hydrolase activity (P = 5.02 × 10−5) and pathways such as calcium signaling pathway (P = 2.09 × 10−8), glutamatergic synapse (P = 4.09 × 10−8) and NOD-like receptor signaling pathway (P = 2.70 × 10−6). Differentially expressed mRNAs might play a role in the pathogenesis of temporal lobe epilepsy. Calcium signaling pathways, synaptic glutamate, and NOD-like receptor signaling pathway play a central role in normal-epilepsy-ANT-DBS treatment series. ANT-DBS achieves its antiepileptic effects by modulating target genes involved in a variety of functions and pathways.






Similar content being viewed by others
Abbreviations
- ANT:
-
Anterior nucleus of the thalamus
- DBS:
-
Deep brain stimulation
- KA:
-
Kainic acid
- SD:
-
Standard deviation
- MTLE:
-
Mesial temporal lobe epilepsy
- qPCR:
-
Quantitative real time PCR
- TS:
-
Total seizures
- PS:
-
Partial seizures
- GS:
-
Generalized seizures
References
Wiebe S, Blume WT, Girvin JP, Eliasziw M (2001) Effectiveness, and efficiency of surgery for temporal lobe epilepsy study, a randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med 345(5):311–318
Sander JW (2004) The use of antiepileptic drugs–principles and practice. Epilepsia 45(Suppl 6):28–34
Engel J, McDermott MP, Wiebe S, Langfitt JT, Stern JM, Dewar S, Sperling MR, Gardiner I, Erba G, Fried I, Jacobs M, Vinters HV, Mintzer S, Kieburtz KG (2012) Early randomized surgical epilepsy trial study early surgical therapy for drug-resistant temporal lobe epilepsy: a randomized trial. JAMA 307(9):922–930
Schramm J, Delev D, Wagner J, Elger CE, von Lehe M (2012) Seizure outcome, functional outcome, and quality of life after hemispherectomy in adults. Acta Neurochir (Wien) 154(9):1603–1612
Jain P, Tomlinson G, Snead C, Sander B, Widjaja E (2018) Systematic review and network meta-analysis of resective surgery for mesial temporal lobe epilepsy. J Neurol Neurosurg Psychiatry 89:1138–1144
Ryvlin P, Cross JH, Rheims S (2014) Epilepsy surgery in children and adults. Lancet Neurol 13(11):1114–1126
Englot DJ, Rolston JD, Wright CW, Hassnain KH, Chang EF (2016) Rates and predictors of seizure freedom with vagus nerve stimulation for intractable epilepsy. Neurosurgery 79(3):345–353
O.C. Cockerell, J.W. Sander, S.D. Shorvon, Remission of epilepsy. The NGPS. National General Practice Study of Epilepsy, Lancet 346(8984) (1995) 1228
Kwan P, Brodie MJ (2000) Early identification of refractory epilepsy. N Engl J Med 342(5):314–319
Georgiadis I, Kapsalaki EZ, Fountas KN (2013) Temporal lobe resective surgery for medically intractable epilepsy: a review of complications and side effects. Epilepsy Res Treat 2013:752195
M. Panebianco, A. Rigby, J. Weston, A.G. Marson, Vagus nerve stimulation for partial seizures, Cochrane Database Syst Rev (4) (2015) CD002896
Englot DJ, Chang EF, Auguste KI (2011) Vagus nerve stimulation for epilepsy: a meta-analysis of efficacy and predictors of response. J Neurosurg 115(6):1248–1255
Heck CN, King-Stephens D, Massey AD, Nair DR, Jobst BC, Barkley GL, Salanova V, Cole AJ, Smith MC, Gwinn RP, Skidmore C, Van Ness PC, Bergey GK, Park YD, Miller I, Geller E, Rutecki PA, Zimmerman R, Spencer DC, Goldman A, Edwards JC, Leiphart JW, Wharen RE, Fessler J, Fountain NB, Worrell GA, Gross RE, Eisenschenk S, Duckrow RB, Hirsch LJ, Bazil C, O’Donovan CA, Sun FT, Courtney TA, Seale CG, Morrell MJ (2014) Two-year seizure reduction in adults with medically intractable partial onset epilepsy treated with responsive neurostimulation: final results of the RNS System Pivotal trial. Epilepsia 55(3):432–441
Bergey GK, Morrell MJ, Mizrahi EM, Goldman A, King-Stephens D, Nair D, Srinivasan S, Jobst B, Gross RE, Shields DC, Barkley G, Salanova V, Olejniczak P, Cole A, Cash SS, Noe K, Wharen R, Worrell G, Murro AM, Edwards J, Duchowny M, Spencer D, Smith M, Geller E, Gwinn R, Skidmore C, Eisenschenk S, Berg M, Heck C, Van Ness P, Fountain N, Rutecki P, Massey A, O’Donovan C, Labar D, Duckrow RB, Hirsch LJ, Courtney T, Sun FT, Seale CG (2015) Long-term treatment with responsive brain stimulation in adults with refractory partial seizures. Neurology 84(8):810–817
Valentín A, García Navarrete E, Chelvarajah R, Torres C, Navas M, Vico L, Torres N, Pastor J, Selway R, Sola R, Alarcon G (2013) Deep brain stimulation of the centromedian thalamic nucleus for the treatment of generalized and frontal epilepsies. Epilepsia 54(10):1823–1833
McLachlan R, Pigott S, Tellez-Zenteno J, Wiebe S, Parrent A (2010) Bilateral hippocampal stimulation for intractable temporal lobe epilepsy: impact on seizures and memory. Epilepsia 51(2):304–307
Krishna V, King NK, Sammartino F, Strauss I, Andrade DM, Wennberg RA, Lozano AM (2016) Anterior nucleus deep brain stimulation for refractory epilepsy: insights into patterns of seizure control and efficacious target. Neurosurgery 78(6):802–811
Meng DW, Liu HG, Yang AC, Zhang K, Zhang JG (2016) Stimulation of anterior thalamic nuclei protects against seizures and neuronal apoptosis in hippocampal ca3 region of kainic acid-induced epileptic rats. Chin Med J (Engl) 129(8):960–966
Shi L, Yang AC, Li JJ, Meng DW, Jiang B, Zhang JG (2015) Favorable modulation in neurotransmitters: effects of chronic anterior thalamic nuclei stimulation observed in epileptic monkeys. Exp Neurol 265:94–101
G. Zhu, D. Meng, Y. Chen, T. Du, Y. Liu, D. Liu, L. Shi, Y. Jiang, X. Zhang, J. Zhang, Anterior nucleus of thalamus stimulation inhibited abnormal mossy fiber sprouting in kainic acid-induced epileptic rats, Brain Res 1701 (2018) 28–35
Fisher RS, Velasco AL (2014) Electrical brain stimulation for epilepsy. Nat Rev Neurol 10(5):261–270
Schulze-Bonhage A (2017) Brain stimulation as a neuromodulatory epilepsy therapy. Seizure 44:169–175
Salanova V (2015) Long-term efficacy and safety of thalamic stimulation for drug-resistant partial epilepsy. Neurology 84(10):1017–1025
Liu HG, Yang AC, Meng DW, Zhang K, Zhang JG (2012) Effect of anterior nucleus of thalamus stimulation on glucose metabolism in hippocampus of epileptic rats. Chin Med J 125(17):3081–3086
Liu HG, Yang AC, Meng DW, Chen N, Zhang JG (2012) Stimulation of the anterior nucleus of the thalamus induces changes in amino acids in the hippocampi of epileptic rats. Brain Res 1477:37–44
Chen YC, Zhu GY, Wang X, Shi L, Jiang Y, Zhang X, Zhang JG (2017) Deep brain stimulation of the anterior nucleus of the thalamus reverses the gene expression of cytokines and their receptors as well as neuronal degeneration in epileptic rats. Brain Res 1657:304–311
Selvakumar T, Alavian KN, Tierney T (2015) Analysis of gene expression changes in the rat hippocampus after deep brain stimulation of the anterior thalamic nucleus. J Vis Exp. https://doi.org/10.3791/52457
Lerche H, Shah M, Beck H, Noebels J, Johnston D, Vincent A (2013) Ion channels in genetic and acquired forms of epilepsy. J Physiol 591(4):753–764
Bernard C, Anderson A, Becker A, Poolos NP, Beck H, Johnston D (2004) Acquired dendritic channelopathy in temporal lobe epilepsy. Science 305(5683):532–535
Bading H (2013) Nuclear calcium signalling in the regulation of brain function. Nat Rev Neurosci 14(9):593–608
Dudek FE, Sutula TP (2007) Epileptogenesis in the dentate gyrus: a critical perspective. Prog Brain Res 163:755–773
Papadopoulos T, Schemm R, Grubmuller H, Brose N (2015) Lipid binding defects and perturbed synaptogenic activity of a Collybistin R290H mutant that causes epilepsy and intellectual disability. J Biol Chem 290(13):8256–8270
Hung YW, Hung SW, Wu YC, Wong LK, Lai MT, Shih YH, Lee TS, Lin YY (2015) Soluble epoxide hydrolase activity regulates inflammatory responses and seizure generation in two mouse models of temporal lobe epilepsy. Brain Behav Immun 43:118–129
Martinon F, Burns K, Tschopp J (2002) The inflammasome: a molecular platform triggering activation of inflammatory caspases and processing of proIL-beta. Mol Cell 10(2):417–426
Minkiewicz J, de Rivero Vaccari JP, Keane RW (2013) Human astrocytes express a novel NLRP2 inflammasome. GLIA 61(7):1113–1121
Tan CC, Zhang JG, Tan MS, Chen H, Meng DW, Jiang T, Meng XF, Li Y, Sun Z, Li MM, Yu JT, Tan L (2015) NLRP1 inflammasome is activated in patients with medial temporal lobe epilepsy and contributes to neuronal pyroptosis in amygdala kindling-induced rat model. J Neuroinflammation 12:18
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates, 6th edn. Academic Press/Elsevier, Amsterdam
Racine RJ (1972) Modification of seizure activity by electrical stimulation. II. motor seizure. Electroencephalogr Clin Neurophysiol 32(3):281–294
Phelan KD, Shwe UT, Williams DK, Greenfield LJ, Zheng F (2015) Pilocarpine-induced status epilepticus in mice: a comparison of spectral analysis of electroencephalogram and behavioral grading using the Racine scale. Epilepsy Res 117:90–96
Acknowledgements
This study was supported by the National Natural Science Funds for Distinguished. Young Scholars (No. 81501118; 81501186) and Beijing Municipal Administration of Hospitals’Ascent Plan (DFL20150503). The funders had no role in the study design, data collection, analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
This study was approved by the Beijing Neurosurgical Institute ethics committee. All animal experiments were performed in accordance with the Guidance for Animal Experimentation of the Capital Medical University and Beijing Guidelines for the care and use of laboratory animals. All efforts have been made to minimize animal suffering during the procedures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, DF., Chen, YC., Zhu, GY. et al. Effects of anterior thalamic nuclei stimulation on gene expression in a rat model of temporal lobe epilepsy. Acta Neurol Belg 120, 1361–1370 (2020). https://doi.org/10.1007/s13760-019-01240-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-019-01240-1