Abstract
Nocturnal blood pressure (BP) changes are an indicator of autonomic dysfunction. We aim to investigate the correlation between nocturnal blood pressure (BP) variability, vitamin D levels and Parkinson’s disease severity (PD) in this study. Thirty-five patients with PD participated in the study. Disease severity was evaluated by United Parkinson’s Disease Rating Scale (UPDRS) and Hoehn and Yahr Scale (HYS). Equivalent levodopa dose was calculated and 25-hydroxyvitamin D levels were measured. The Non-Motor Symptom Questionnaire (NMSQ) was applied to all patients. Ambulatory BP monitoring for 24 h was established. Patients were divided into three groups according to nocturnal BP results: dippers (normal finding—a decline in mean nighttime BP of more than 10%); non-dippers (pathological—a decline in mean nighttime BP of less than 10%); reverse dippers (pathological—an increase in mean nighttime BP) .The mean score of the NMSQ was higher in the group with HYS > 2 (p = 0.050). Four patients were dipper, 17 patients were non-dipper and 16 patients were reverse dipper. There was no significant difference between the three groups in terms of age, gender, disease duration, age of the disease onset, disease stage, disease duration, dopamine agonist usage, levodopa equivalent dose, vitamin D level and NMSQ scores. NMSQ scores are high in advanced PD. Ambulatory BP monitoring is useful in detecting autonomic dysfunction. The number of patients with non-dipping and reverse dipping is high in PD, independent from PD severity, drug dose, vitamin D and the other NMS symptoms.
Similar content being viewed by others
Introduction
The motor symptoms of Parkinson’s disease (PD) are well known, but recently non-motor symptoms (NMS) including cardiovascular, gastrointestinal, urinary and sexual symptoms are of more interest. These symptoms can cause marked disability and affect the quality of life [1]. Especially, cardiac autonomic dysfunction (AD) plays a crucial role in disability and sudden death [2].
The change in blood pressure (BP) is one of the most remarkable findings for AD. In healthy subjects, BP is lower at nighttime which has been described as ‘dipping’ [3]. If BP does not decrease at nighttime, it is called ‘non-dipping’ and BP increase is called ‘reverse dipping’ [4]. In PD, the prevalence of non-dipping is high and goes up to 90% [5, 6].
Abnormal circadian falls in BP occur more often in PD with AD compared to PD without AD [7]. It was believed that the AD is seen in the late phase of the disease, but recent studies showed that it can occur in early phase of the disease, and even before the onset of motor symptoms [8,9,10].
Vitamin D plays a crucial role as it has a neuroprotective effect on autonomic function [11]. A recent study showed that serum vitamin D levels decreased in PD patients with orthostatic hypotension (OH) [12]. Additionally, it was shown that vitamin D level could influence the BP and cardiac autonomy. By this way, vitamin D can play a role in the pathophysiology of OH in PD [13, 14]. But still, there are some controversiesregarding the effect of vitamin D levels on both PD and autonomic system. In this study, we aimed to investigate the correlation between NMS, nocturnal BP changes, vitamin D levels and severity of PD.
Materials and methods
This is a prospective study; including 35 patients with idiopathic PD. All patients were recruited from Istanbul Medipol University, Faculty of Medicine, Neurology Department from January 2016 to March 2017. The diagnosis of PD was confirmed by a movement disorders specialist (N.H.Y.), according to the United Kingdom Parkinson’s Disease Society Brain Bank Criteria [15]. The exclusion criteria were: age below 18, patients with cognitive impairment, pyramidal, cerebellar and vertical gaze abnormality, a medical condition which can affect BP (malignancy, coronary heart disease, uncontrolled hypertension), taking drugs such as alpha-1- blockers and vitamin D supplement. Disease severity was evaluated by United Parkinson’s Disease Rating Scale (UPDRS) and Hoehn and Yahr Scale (HYS). Equivalent levodopa dose was calculated for each patient [16]. The cardiological examination of the patients, evaluated by electrocardiography and transthoracic echocardiography, was all normal.
The Non-Motor Symptom Questionnaire (NMSQ) containing 30 items was used to evaluate AD. It was applied to all the participants and the participants answered the questions as present or absent. Scoring range was 0–30 (0 = no AD, 1–5 = mild AD, 6–9 = medium AD, 10–13 = severe AD, ≥ 14 AD). The answer to the 20th question evaluating OH was also noted (Question 20: do you feel drowsiness or weakness when sitting or standing up from the supine position?).
Peripheral blood samples for 25-hydroxyvitamin D were collected from patients at 9 am after a 12-h fasting. The 25-hydroxyvitamin D levels were analyzed using electrochemiluminescence. Levels under 20 ng/ml were accepted as vitamin D deficiency.
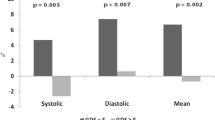
All participants underwent 24-h ambulatory BP monitoring (ABPM) by automated, validated and calibrated oscillometric portable monitor. The ambulatory BP was measured every 15 min at daytime, every 30 min at nighttime. Blood pressure recording started in the morning and continued for 24 h. During monitoring, patients performed their usual daily activities, when the cuff was inflated, the patient was informed to relax the arm where the cuff was located. Dipping values of 10–20% were considered as normal, while for 0–10% values, dipping was considered as absent and named as ‘non-dipping’ [17]. Mean BP was calculated, for each night and day, as the diastolic pressure plus one-third of systolic pressure minus the diastolic pressure. The percentage difference in nighttime BP was calculated as follows: (mean daytime BP−mean nighttime BP⁄mean daytime BP × 100). Negative values for dipping are considered as reversed dipping. Patients were divided into three groups: dippers (a decline in mean nighttime BP of more than 10%); non-dippers (a decline in mean nighttime BP of less than 10%); reverse dippers (an increase in mean nighttime BP).
The study protocol was approved by the Ethical Committee of the Istanbul Medipol University, Istanbul, Turkey. Informed consent was obtained from all subjects. The study was performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Statistical analysis
Descriptive statistics were used to summarize clinical characteristics. Continuous variables were expressed as mean ± standard deviation or median and range. Normal distribution was tested by Kolmogorov–Smirnov test. Kruskal–Wallis test was performed to compare non-normal distributed variables among the groups. Pearson’s Chi-squared test was used for testing categorical variables. p value of < 0.05 was taken significant for all tests. Statistical Package for the Social Sciences (SPSS version 11.0, SPSS Inc., Chicago, IL, USA) was used for analysis.
Results
Twenty-four of the patients were male (68.57%), and 11 were female (31.43%). Two patients were in HYS 1, 14 were in HYS 2, 15 were in HYS 3 and 4 were in HYS 4. Twenty-eight patients were taking dopamine agonist (80.0%), while all of them were taking levodopa with different dosages.
The demographical data and characteristics of the patients are shown in Table 1.
According to the result of NMSQ score: 1–5 points: 4 patients, 6–9 points: 6 patients, 10–13: 10 patients, 14 points and above: 15 patients. Eighteen of the patients (51.43%) answered ‘yes’ to the question 20. Four patients were dipper (11.43%), 17 (48.57%) patients were non-dipper and 16 (45.71%) patients were reverse dipper. There were 21 people (60.00%) with vitamin D levels below 20, 14 people (40.00%) with 20 or more.
When the patients were divided into two subgroups according to disease stages, the ages of patients with HYS > 2 were significantly higher and the duration of the disease was longer (p = 0.014, 0.007, respectively). The mean score of the NMSQ score was higher in the group with HYS > 2 (p = 0.050). (Table 2).
When the patients were divided into three groups as dipper, dipper and reverse dipper according to the results of ABPM, there was no significant difference between the three groups in terms of age, gender, disease duration, age of the disease onset, disease stage, disease duration, dopamine agonist usage, levodopa equivalent dose, vitamin D level and NMSQ scores. (Table 3).
Discussion
Autonomic dysfunction is common in PD. It occurs before the onset of the disease, and present as varying intensities in each stage and sometimes cause more functional disability than motor symptoms [18]. NMSQ is a method that allows evaluating AD in PD patients rapidly and presents the symptoms that the patients subjectively reported [19]. Only one question in the questionnaire examines the presence of cardiovascular dysfunction. Approximately half of the participants answered ‘yes’ to this question in our study. In epidemiological studies using the NMSQ questionnaire, at least one NMS finding is seen in 99% of the patients during the disease course [20], while the rate of cardiovascular findings varies between 14–36% [20,21,22]. NMSQ scores were found to be higher in patients with long disease duration and more severe disease. Similar to previous studies, in our study, NMSQ scores were significantly higher in the patient group with HYS of > 2.
Altered circadian BP changes can be presented in left ventricular hypertrophy, heart failure, myocardial infarction, microalbuminuria, cerebrovascular and vascular dementia [23]. Ambulatory BP monitoring is a method used to detect the existence of the cardiac dysautonomia (non-dipping/reverse dipping, OH and post-prandial hypotension and elevated BP variability) [24]. On the other hand, reverse dipping is a more risky clinical entity that causes stroke and cardiovascular mortality in patients with essential hypertension more common than non-dipping [25,26,27]. In PD, non-dipping/reverse dipping, OH and BP changes are common both because of the disease itself and the medication used for the treatment. Ejaz et al. found that 93% of the patients have circadian BP disorder, while the rate of 88% was reported by Sommer et al. and 80% by Oh et al. [5, 28, 29]. The rate of reverse dipper in PD was 33% in a single study and it was reported as a more accurate finding of cardiac dysautonomia compared to other parameters [30]. In our study, 31 patients (88.57%) had a circadian BP disorder, while 50% of these 35 patients had reverse dipping. Therefore, it was thought that the presence of reverse dipping was an important finding for cardiac dysautonomia and PD patients should be screened for this reason.
In this study, non-dippers were found to be lower than other studies (39%), but less frequently observed reverse dippers was higher. It was thought that reverse dipper blood pressure changes may be important in this type of patients.
The effects of anti-PD drugs on BP are variable. In one study, the administration of levodopa significantly lowered mean arterial BP [31], whereas, in other studies, OH was not found to be associated with levodopa treatment [32, 33]. Dopamine agonists have been shown to cause acute asymptomatic OH [34]. Increasing drug doses and combined therapies increase the risk of OH development, but in the long term, tolerance to these drugs occurs [35]. In this study, patients were grouped into three groups: reverse dipper, non-dipper, dipper; and these groups had shown no significant differences in terms of PD treatment. On this basis, the use of dopamine agonists in PD and equivalent dose of levodopa were thought to have no effect on changes in BP.
Like PD, circadian blood pressure changes can occur with many diseases. Nocturnal movement, sleep disorders like obstructive sleep apnea syndrome or getting up at night for urinary problems are some of these (36,37). However, one of the limiting aspects of this study was the lack of including disorders other than PD which can cause nocturnal blood pressure changes.
The reason for cardiac changes in PD is mostly AD [9]. As a result of affected sympathetic noradrenergic neurons independent of dopaminergic effect, cardiac and extra-cardiac noradrenergic denervation are responsible for the presence of cardiac dysautonomia [35]. Oh et al. showed that sympathetic noradrenergic dysfunction occurs independent of striatal dopamine uptake in Positron Emission Tomography [29]. In our study, when we compared nocturnal BP changes with PD severity, stage of disease duration, age at onset, we observed no significant difference. Therefore, our work supported the hypothesis that AD in PD has a different pathogenesis from cardinal motor findings.
When we looked at the relationship between NMSQ score and nocturnal BP changes, considering the NMSQ score to have an association with cardiac dysautonomia, the patients who have dipper hypertension had higher mean NMSQ score. But the difference was insignificant between the groups. The reason for this was thought to be that cardiac AD was represented with only one question in NMSQ and that the cardiac AD that presented in patients was frequently subclinical.
Vitamin D is a neuroprotective molecule [39]. Vitamin D levels were found to be low in studies conducted with PD [40, 41]. While there are findings showing that low vitamin D levels have an effect on the pathogenesis and progression of PD [42], there are also researchers who think that this relationship is not significant [43]. It is observed that the HYS remained stable in PD patients who were given vitamin D for 12 months and their daily activities were better [44]. Low vitamin D levels are another risk factor of arterial hypertension [45], as it can cause cardiac structural changes that can lead systolic and diastolic dysfunction [46]. In PD, low levels of vitamin D were found to be associated with AD [39], but the relationship between non-dipping and reverse dipping has not been researched before. In our study, when we compare, dipper, non-dipper, and reverse dipper patient groups showed no significant difference according to vitamin D levels. Therefore, vitamin D was not considered to be effective for nocturnal BP changes and its subtypes which is a marker of AD in PD.
As a result, NMSQ scores are high in advanced PD, and a limited number of patients report their symptoms subjectively in NMSQ. ABPM is a valuable tool for determining subclinic cardiac AD findings in PD. However, non-dipping and reverse dipping ratios are high in PD, this is independent of age, gender, disease duration, age at onset, the severity of the disease, the use of anti-PD drugs and vitamin D level. Prospective studies conducted on this subject with more number of patients are needed.
References
Antonini A, Barone P, Marconi R, Morgante L, Zapulla S, Pontieri FE, Ramat S, Ceravolo MG, Meco G, Cicarelli G, Pederzoli M, Manfredi M, Ceravolo R, Mucchiut M, Volpe G, Abbruzzese G, Bottacchi E, Bartolomei L, Ciacci G, Cannas A, Randisi MG, Petrone A, Baratti M, Toni V, Cossu G, Del Dotto P, Bentivoglio AR, Abrignani M, Scala R, Pennisi F, Quatrale R, Gaglio RM, Nicoletti A, Perini M, Avarello T, Pisani A, Scaglioni A, Martinelli PE, Iemolo F, Ferigo L, Simone P, Soliveri P, Troianiello B, Consoli D, Mauro A, Lopiano L, Nastasi G, Colosimo C (2012) The progression of non-motor symptoms in Parkinson’s disease and their contribution to motor disability and quality of lif. J Neurol 259:2621–2631
Ha AD, Brown CH, York MK, Jankovic J (2011) The prevalence of symptomatic orthostatic hypotension in patients with Parkinson’s disease and atypical parkinsonism. Parkinsonism Relat Disord 17(8):625–628. https://doi.org/10.1016/j.parkreldis.2011.05.02
O’Brien E, Sheridan J, Malley OK (1998) Dippers and non-dippers. Lancet 12:397
Di Daniele N, Fegatelli DA, Rovella V, Castagnola V, Gabriele M, Scuteri A (2017) Circadian blood pressure patterns and blood pressure control in patients with chronic kidney disease. Atherosclerosis 267:139–145
Ejaz AA, Sekhon IS, Munjal S (2006) Characteristics findings ies of patients with Parkinson’s disease. Er J Intern Med 17:417–420
Schmidt C, Berg D, Prieur S. Et al. Schmidt C, Berg D, Herting, Prieur S, Junghanns S, Schweitzer K, Globas C, Schöls L, Reichmann H, Ziemssen T (2009) Loss of nocturnal blood pressure fall in various extrapyramidal syndromes. Mov Disord 24:2136–2142
Senard JM, Chamontin B, Rascol A, Montastruc JL (1992) Neurogenic orthostatic hypotension as the initial feature of Parkinson disease. Clin Auton Res 2:99–104
Linden D, Diehl RR, Berlit P (1997) Sympathetic cardiovascular dysfunction in longstanding idiopathic Parkinson’s disease. Clin Auton Res 7:311–314
Goldstein DS (2006) Orthostatic hypotension as an early finding in Parkinson disease. Clin Auton Res 16:46–54
Milazzo V, Di Stefano C, Servo S, Zibetti M, Lopiano L, Maule S (2012) Neurogenic orthostatic hypotension as the initial feature of Parkinson disease. Clin Auton Res 22:203–206
Kalueff AV, Eremin KO, Tuohımaa P (2004) Mechanisms of neuropretective action of vitamin D3. Biochemistry 69:738–741
Jang W, Park J, Kim JS, Youn J, Oh E, Kwon KY, Jo KD, Lee MK, Kim H-T (2015) Vitamin D deficiency in Parkinson’s disease patients with orthostatic hypotension. Acta Neurol Scand 132:242–250
Pilz S, Tomaschitz A, Ritz E, Pieber TR (2009) Vitamin D status and arteriel hypertension: a systematic review. Nat rev Cardiol 6:621–630
Vinh Quoc Luong K, Thi Hoang Nguyen L (2012) Vitamin D and Parkinson’s disease. J Neurosci Res 90:2227–2236
Hughes AJ, Daniel SE, Kilford L, Lees AJ (1992) Accuracy of clinical diagnosis of idiopathic Parkinson’s disease: a clinico-pathological study of 100 cases. J Neurol Neurosurg Psychiatry 55:181–184
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE (2010) Systematic Review of Levodopa Dose Equivalency Reporting in Parkinson’s. Disease Mov Disord 25:2649–2653
Eigenbrodt ML, Rose KM, Couper DJ, Arnett DK, Smith R, Jones D (2000) Orthostatic hypotension as a risk factor for stroke: the atherosclerosis risk in communities (ARIC) study, 1987–1996. Stroke 31:2307–2313
Schapira AH (2013) Recent developments in biomarkers in Parkinson disease. Curr Opin Neurol 26:395–400
Chaudhuri KR, Healy DG, Schapira AH, National Institute for Clinical Excellence (2006) Non-motor symptoms of Parkinson’s disease: diagnosis andmanagement. Lancet Neurol 5:235–245
Barone P, Antonini A, Colosimo C, Marconi R, Morgante L, Avarello TP, Bottacchi E, Cannas A, Ceravolo G, Ceravolo R, Cicarelli G, Gaglio RM, Giglia RM, Iemolo F, Manfredi M, Meco G, Nicoletti A, Pederzoli M, Petrone A, Pisani A, Pontieri FE, Quatrale R, Ramat S, Scala R, Volpe G, Zappulla S, Bentivoglio AR, Stocchi F, Trianni G, Dotto PD, PRIAMO study group (2009) The PRIAMO study: a multicenter assessment of nonmotor symptoms and their impact on quality of life in Parkinson’s disease. Mov Disord 24:1641–1649
Crosiers D, Pickut B, Theuns J, Deyn PP, Van Broeckhoven C, Martinez-Martin P, Chaudhuri KR, Cras P (2012) Non-motor symptoms in a Flanders-Belgian population of 215 Parkinson’s disease patients as assessed by the Non-motor Symptoms Questionnaire. Am J Neurodegener Dis 1:160–167
Gan J, Zhou M, Chen W, Liu Z (2014) Non-motor symptoms in Parkinson’s disease patients. J Clin Neurosci 21:751–754
Hermida RC (2007) Ambulatory blood pressure monitoring in the prediction of cardiovascular events and effects of chronotherapy: rationale and design of the MAPEC study. Chronobiol Int 24:749–775
Saladini F, Di Marco A, Palatini P (2016) Autonomic dysfunction: how to identify and when to treat? High Blood Press Cardiovasc Prev 23:237–243
Park JH, Lee HS, Kim JH, Lee JH, Kim J, Choi SW (2014) J Stroke Cerebrovasc Dis 23:1171–1176
Yan B, Peng L, Dong Q, Zheng F, Yang P, Sun L, Gong S, Zeng L, Wang G (2015) Reverse-dipper pattern of blood pressure may predict lacunar infarction in patients with essential hypertension. Reverse-dipper pattern of blood pressure may predict lacunar infarction in patients with essential hypertension. Eur J Neurol 22:1022–1025
Wang C, Ye Z, Li Y, Zhang J, Zhang Q, Ma X, Peng H, Lou T (2016) Prognostic value of reverse dipper blood pressure pattern in chronic kidney disease patients not undergoing dialysis: prospective cohort study. Sci Rep 6:34932
Sommer S, Aral-Becher B, Jost W (2011) Nondipping in Parkinson’s disease. Parkinsons Dis 2011:897586
Oh YS, Kim JS, Chung YA, You IeR, Yang DW, Chung SW, Park JW, Kim YI, Lee KS (2013) Orthostatic hypotension, non-dipping and striatal dopamine in Parkinson disease. Neurol Sci 34:557–560
Milazzo V, Di Stefano C, Vallelonga F, Sobrero G, Zibetti M, Romagnolo A, Merola A, Milan A, Espay AJ, Lopiano L, Veglio F, Maule S (2018) Reverse blood pressure dipping as marker of dysautonomia in Parkinson disease. Parkinsonism Relat Disord
Noack C, Schroeder C, Heusser K, Lipp A (2014) Cardiovascular effects of levodopa in Parkinson’s disease. Parkinsonism Relat Disord 20:815–818
Mehagnoul-Schipper DJ, Boerman RH, Hoefnagels WH, Jansen RW (2001) Effect of levodopa on orthostatic and postprandial hypotension in elderly Parkinsonian patients. J Gerontol A Biol Sci Med Sci 56:M749–M755
Goldstein DS, Eldadah BA, Holmes C, Pechnik S, Moak J, Saleem A, Sharabi Y (2005) Neurocirculatory abnormalities in Parkinson disease with orthostatic hypotension: independence from levodopa treatment. Hypertension 46:1333–1339
Kujawa K, Leurgans S, Raman R, Blasucci L, Goetz CG (2000) Acute orthostatic hypotension when starting dopamine agonists in Parkinson’s disease. Arch Neurol 57:1461–1463
Sánchez-Ferro A, Benito-León J, Gómez-Esteban JC (2013) The management of orthostatic hypotension in Parkinson’s disease. Front Neurol 4:64
Resta O, Rana L, Procacci V, Guido P, Picca V, Scarpelli F (1998) Autonomic dysfunction in normotensive awake subjects with obstructive sleep apnoea syndrome. Monaldi Arch Chest Dis 53(1):23–29
Cortelli P, Parchi P, Sforza E, Contin M, Pierangeli G, Barletta G, Lugaresi E (1994) Cardiovascular autonomic dysfunction in normotensive awake subjectswith obstructive sleep apnoea syndrome. Clin Auton Res 4(1–2):57–62
Jain S, Goldstein DS (2012) Cardiovascular dysautonomia in Parkinson disease: from pathophysiology to pathogenesis. Neurobiol Dis 46:572–580
Jang W, Park HH, Lee KY, Lee YJ, Kim HT, Koh SH (2015) 1,25-dyhydroxyvitamin D3 attenuates L-DOPA-induced neurotoxicity in neural stem cells. Mol Neurobiol 51:558–570
Evatt ML, DeLong MR, Kumari M, Auinger P, McDermott MP, Tangpricha V (2011) High prevalence of hypovitaminosis D status in patients with early Parkinson disease. Arch Neurol 68:314–319
Ding H, Dhima K, Lockhart KC, Locascio JJ, Hoesing AN, Duong K, Trisini-Lipsanopoulos A, Hayes MT, Sohur US, Wills AM, Mollenhauer B, Flaherty AW, Hung AY, Mejia N, Khurana V, Gomperts SN, Selkoe DJ, Schwarzschild MA, Schlossmacher MG, Hyman BT, Sudarsky LR, Growdon JH, Scherzer CR (2013) Unrecognized vitamin D3 deficiency is common in Parkinson disease: Harvard Biomarker Study. Neurology 81:1531–1537
Newmark HL, Newmark J (2007) Vitamin D and Parkinson’s disease-a hypothesis. Mov Disord 22:461–468
Petersen MS, Bech S, Christiansen DH, Schmedes AV, Halling J (2014) The role of vitamin D levels and vitamin D receptor polymorphism on Parkinson’s disease in the Faroe Islands. Neurosci Lett 561:74–79
Suzuki M, Yoshioka M, Hashimoto M, Murakami M, Noya M, Takahashi D, Urashima M (2013) Randomized, double-blind, placebo-controlled trial of vitamin D supplementation in Parkinson disease. Am J Clin Nutr 97:1004–1013
Kunutsor SK, Apekey TA, Steur M (2013) Vitamin D and risk of future hypertension: meta-analysis of 283,537 participants. Eur J Epidemiol 28:205–221
Pekkanen MP, Ukkola O, Hedberg P, Piira OP, Lepojärvi S, Lumme J, Tulppo MP, Huikuri HV (2015) Serum 25-hydroxyvitamin D is associated with major cardiovascular risk factors and cardiac structure and function in patients with coronary artery disease. Nutr Metab Cardiovasc Dis 25:471–478
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Arici Duz, O., Helvaci Yilmaz, N. Nocturnal blood pressure changes in Parkinson’s disease: correlation with autonomic dysfunction and vitamin D levels. Acta Neurol Belg 120, 915–920 (2020). https://doi.org/10.1007/s13760-019-01113-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-019-01113-7