Abstract
Familial amyloid polyneuropathy (FAP) is a most often length-dependent axonal neuropathy, often part of a multisystem disorder also affecting other organs, such as cardiac, gastrointestinal, genitourinary, renal, meningeal and eye tissue. It is most frequently the result of a mutation in the TTR gene, most commonly a p.Val50Met mutation. TTR-FAP is a rare autosomal dominant heritable disabling, heterogeneous disease in which early diagnosis is of pivotal importance when attempting treatment. This paper discusses the course of four Belgian FAP patients with different TTR mutations (p.Val48Met; p.Val52Ala; p.Ala59Val; p.Val50Met). We also review the diagnosis and differential diagnosis of TTR-FAP, diagnostic studies, follow-up, its current treatment and those in development, prognosis and the importance of genetic counseling. At first, TTR-FAP is often misdiagnosed as a chronic inflammatory demyelinating polyneuropathy or chronic idiopathic axonal polyneuropathy. Genetic testing is obligatory to confirm the diagnosis of TTR-FAP, except in familial cases. Biopsy samples are an asset in diagnosing TTR-FAP but can be falsely negative. At the moment, tafamidis meglumine is considered as first-line treatment in stage I neurological disease. Patients eligible for liver transplantation should be carefully selected when first-line therapy fails.

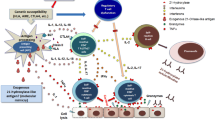
Modified from Adams [6]
Similar content being viewed by others
References
Plante-Bordeneuve V, Said G (2011) Familial amyloid polyneuropathy. Lancet Neurol. https://doi.org/10.1016/s1474-4422(11)70246-0
Rowczenio DM, Noor I, Gillmore JD, Lachmann HJ, Whelan C, Hawkins PN et al (2014) Online registry for mutations in hereditary amyloidosis including nomenclature recommendations. Hum Mutat. https://doi.org/10.1002/humu.22619
Adams D (2013) Recent advances in the treatment of familial amyloid polyneuropathy. Ther Adv Neurol Disord. https://doi.org/10.1177/1756285612470192
Ruberg FL, Berk JL (2012) Transthyretin (TTR) cardiac amyloidosis. Circulation. https://doi.org/10.1161/circulationaha.111.078915
Ikeda S, Nakazato M, Ando Y, Sobue G (2002) Familial transthyretin-type amyloid polyneuropathy in Japan: clinical and genetic heterogeneity. Neurology 58:1001–1007
Adams D, Suhr OB, Hund E, Obici L, Tournev I, Campistol JM et al (2016) First European consensus for diagnosis, management, and treatment of transthyretin familial amyloid polyneuropathy. Curr Opin Neurol. https://doi.org/10.1097/wco.0000000000000289
Chanoine JP, Alex S, Fang SL, Stone S, Leonard JL, Korhle J et al (1992) Role of transthyretin in the transport of thyroxine from the blood to the choroid plexus, the cerebrospinal fluid, and the brain. Endocrinology 130:933–938. https://doi.org/10.1210/endo/130.2.1733735
Schmidt HH-J, Barroso F, Gonzalez-Duarte A, Conceicao I, Obici L, Keohane D et al (2016) Management of asymptomatic gene carriers of transthyretin familial amyloid polyneuropathy. Muscle Nerve. https://doi.org/10.1002/mus.25210
Kollmer J, Sahm F, Hegenbart U, Purrucker JC, Kimmich C, Schonland SO et al (2017) Sural nerve injury in familial amyloid polyneuropathy: MR neurography vs clinicopathologic tools. Neurology. https://doi.org/10.1212/wnl.0000000000004178
Bulawa CE, Connelly S, DeVit M, Wang L, Weigel C, Fleming JA et al (2012) Tafamidis, a potent and selective transthyretin kinetic stabilizer that inhibits the amyloid cascade. Proc Natl Acad Sci. https://doi.org/10.1073/pnas.1121005109
Berk JL, Suhr OB, Obici L, Sekijima Y, Zeldenrust SR, Yamashita T et al (2013) Repurposing diflunisal for familial amyloid polyneuropathy: a randomized clinical trial. JAMA. https://doi.org/10.1001/jama.2013.283815
Cavallaro T, Klunk W (2016) PiB-PET detects transthyretin-related cerebral amyloid angiopathy. Neurology. https://doi.org/10.1212/wnl.0000000000003018
Sekijima Y, Yazaki M, Oguchi K, Ezawa N, Yoshinaga T, Yamada M (2016) Cerebral amyloid angiopathy in posttransplant patients with hereditary ATTR amyloidosis. Neurology. https://doi.org/10.1212/WNL.0000000000003001
Liepnieks JJ, Zhang LQ, Benson MD (2010) Progression of transthyretin amyloid neuropathy after liver transplantation. Neurology. https://doi.org/10.1212/wnl.0b013e3181ea15d4
Franz C, Hoffmann K, Hinz U, Singer R, Hund E, Gotthardt DN et al (2013) Modified body mass index and time interval between diagnosis and operation affect survival after liver transplantation for hereditary amyloidosis: a single-center analysis. Clin Transplant. https://doi.org/10.1111/ctr.12193
Wange N, Anan I, Ericzon B-G, Pennlert J, Pilebro B, Suhr OB et al (2017) Atrial fibrillation and central nervous complications in liver transplanted hereditary transthyretin amyloidosis patients. Transplantation. https://doi.org/10.1097/tp.0000000000001975
Eriksson M, Buttner J, Todorov T, Yumlu S, Schonland S, Hegenbart U et al (2009) Prevalence of germline mutations in the TTR gene in a consecutive series of surgical pathology specimens with ATTR amyloid. Am J Surg Pathol. https://doi.org/10.2353/jmoldx.2009.08016
Coelho T, Adams D, Silva A, Lozeron P, Hawkins PN, Mant T et al (2013) Safety and efficacy of RNAi therapy for transthyretin amyloidosis. N Engl J Med. https://doi.org/10.1056/nejmoa1208760
Obici L, Kuks JB, Buades J, Adams D, Suhr OB, Coelho T et al (2016) Recommendations for presymptomatic genetic testing and management of individuals at risk for hereditary transthyretin amyloidosis. Curr Opin Neurol. https://doi.org/10.1097/wco.0000000000000290
Funding
The authors declare that no funding was involved.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Martens, B., De Pauw, M. & De Bleecker, J.L. Single-centre experience on transthyretin familial amyloid polyneuropathy: case series and literature review. Acta Neurol Belg 118, 179–185 (2018). https://doi.org/10.1007/s13760-018-0906-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-018-0906-z