Abstract
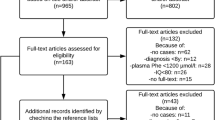
The aim of this study is to evaluate the neuropsychological status in a cohort of children with early and continuously treated phenylketonuria in Assiut, Upper Egypt. The study was implemented in seventy-eight phenylketonuria (PKU) children. Only 34 patients met the inclusion criteria. Investigated patients were evaluated according to detailed history, neurological examination, Childhood Autism Rating Scale, full scale Intelligence Quotient, attention deficit hyperactivity disorder, electroencephalography and magnetic resonance imaging (MRI). This study concluded that the prognosis for early diagnosed children with PKU treated from the first weeks of life is generally good. However, they are at increased risk for neurological complications and behavioral problems. So, neonatal screening for PKU is highly recommended in Egypt, for early detection and management. In addition, neuropsychological and MRI assessments in PKU children should be done.
Similar content being viewed by others
References
Følling A (1934) Über Ausscheidung von Phenylbrenztraubensäure im Harn als Stoffwechselanomalie in Verbindung mit Imbezillität. Hoppe Seylers Z Physiol Chem. 227:169–176
MacLeod EL, Ney DM (2010) Nutritional management of phenylketonuria. Ann Nestlé (Engl). 68:58–69
Blau N, van Spronsen FJ, Levy HL (2010) Phenylketonuria. Lancet 376:1417–1427
Brumm VL, Bilder D, Waisbren SE (2010) Psychiatric symptoms and disorders in phenylketonuria. Mol Genet Metab 99(Suppl 1):S59–S63
Brumm VL, Azen C, Moats RA, Stern AM, Broomand C, Nelson MD, Koch R (2004) Neuropsychological outcome of subjects participating in the PKU adult collaborative study: a preliminary review. J Inherit Metab Dis 27:549–566
Surtees R, Blau N (2000) The neurochemistry of phenylketonuria. Eur J Pediatr 159:S109–S113
Scarabino T, Popolizio T, Tosetti M, Montanaro D, Giannatempo GM, Terlizzi R, Pollice S, Maiorana A, Maggialetti N, Carriero A, Leuzzi V, Salvolini U (2009) Phenylketonuria: white-matter changes assessed by 3.0-T magnetic resonance (MR) imaging, MR spectroscopy and MR diffusion. Radiol Med. 114:461–474
Jones SJ, Turano G, Kriss A, Shawkat F, Kendall B, Thompson AJ (1995) Visual evoked potentials in phenylketonuria: association with brain MRI, dietary state, and IQ. J Neurol Neurosurg Psychiatry 59:260–265
Scriver CR, Kaufman S (2001) Hyperphenylalaninemia: phenylalanine hydroxylase deficiency. In: Scriver CR, Beaudet AL, Sly WS, Valle D (eds) The Metabolic and Molecular Bases of Inherited Disease, 8th edn. McGraw-Hill, New York, pp 1667–1724
Williams RA, Mamotte CD, Burnett JR (2008) Phenylketonuria: an inborn error of metabolism. Clin Biochem Rev 29:31–40
Autism and Developmental Disabilities Monitoring Network Surveillance Year 2008 Principal Investigators; Centers for Disease Control and Prevention (2012) Prevalence of autism spectrum disorders–Autism and Developmental Disabilities Monitoring Network, 14 sites, United States, 2008. MMWR Surveill Summ. 61(3):1–19
Kuehn BM (2007) CDC: autism spectrum disorders common. JAMA 297:940
Manzi B, Loizzo AL, Giana G, Curatolo P (2008) Autism and metabolic diseases. J Child Neurol 23:307–314
Saad K, Hammad E, Abdel-rahman AA, Sobhy KM (2013) Autistic symptoms in late diagnosed phenylketonuric children in Upper Egypt. J Neur Res 3(3–4):122–129
Chen CH, Hsiao KJ (1989) A Chinese classic phenylketonuria manifested as autism. Br J Psych 155:251–253
Steiner CE, Acosta AX, Guerreiro MM, Marques-de-Faria AP (2007) Genotype and natural history in unrelated individuals with phenylketonuria and autistic behavior. Arq Neuropsiquiatr 65(2A):202–205
Moss J, Howlin P (2009) Autism spectrum disorders in genetic syndromes: implications for diagnosis, intervention and understanding the wider autism spectrum disorder population. J Intel Dis Res. 53:852–873
Caglayan AO (2010) Genetic causes of syndromic and non-syndromic autism. Dev Med Child Neurol 52:130–138
Antshel KM, Waisbren SE (2003) Developmental timing of exposure to elevated levels of phenylalanine is associated with ADHD symptom expression. J Abnorm Child Psychol 31:565–574
Antshel KM (2010) ADHD, learning, and academic performance in phenylketonuria. Mol Genet Metab 99(S1):S52–S58
Melika LK (1998) The Stanford Binet Intelligence Scale. Arabic Examiner’s Handbook, 4th edn. Dar El-Maref Publishing, Cairo
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders: DSM-IV-TR. American Psychiatric Association, Washington, DC
Schopler E, Reichller R, Renner BR (1994) The Childhood Autism Rating Scale CARS. Western Psychological Services, Los Angeles
Rellini E, Tortolani D, Trillo S, Carbone S, Montecchi F (2004) Childhood Autism Rating Scale CARS, and Autism Behavior Checklist ABC, correspondence and conflicts with DSM-IV criteria in diagnosis of autism. J Autism Dev Disord 34:703–708
Perry A, Condillac RA, Freeman NL, Dunn-Geier J, Belair J (2005) Multi-site study of the Childhood Autism Rating Scale (CARS) in five clinical groups of young children. J Autism Dev Disord 355:625–634
Pietz J, Dunckelmann R, Rupp A, Rating D, Meinck HM, Schmidt H, Bremer HJ (1998) Neurological outcome in adult patients with early-treated phenylketonuria. Eur J Pediatr 157:824–830
Albrecht J, Garbade SF, Burgard P (2009) Neuropsychological speed tests and blood phenylalanine levels in patients with phenylketonuria: a meta-analysis. Neurosci Biobehav Rev 33:414–421
Crossley LH, Anderson PJ (2010) Neuropsychological functioning in early-treated phenylketonuria—a review. Ann Nestlé (Engl). 68:78–88
McCandless DW (2012) Epilepsy: animal and human correlations. Springer, New York
González MJ, Gutiérrez AP, Gassió R, Fusté ME, Vilaseca MA, Campistol J (2011) Neurological complications and behavioral problems in patients with phenylketonuria in a follow-up unit. Mol Genet Metab 104:S73–S79
Yalaz K, Vanli L, Yilmaz E, Tokatli A, Anlar B (2006) Phenylketonuria in pediatric neurology practice: a series of 146 cases. J Child Neurol 21:987–990
Thompson AJ, Tillotson AJ, Smith I et al (1993) Brain MRI changes in phenylketonuria; associations with dietary status. Brain. 116:811–821
Cleary MA, Walter JH, Wraith JE, Jenkins JP, Alani SM, Tyler K, Whittle D (1994) Magnetic resonance imaging of the brain in phenylketonuria. Lancet 344:87–90
Toft PB, Lou HC, Krägeloh-Mann I, Andresen J, Güttler F, Guldberg P, Henriksen O (1994) Brain magnetic resonance imaging in children with optimally controlled hyperphenylalaninaemia. J Inherit Metab Dis 17:575–583
Weglage J, Pietsch M, Fünders B, Koch HG, Ullrich K (1995) Neurological findings in early treated phenylketonuria. Acta Paediatr 84:411–415
Landvogt C, Mengel E, Bartenstein P, Buchholz HG, Schreckenberger M, Siessmeier T, Scheurich A, Feldmann R, Weglage J, Cumming P, Zepp F, Ullrich K (2008) Reduced cerebral fluoro-l-dopamine uptake in adult patients suffering from phenylketonuria. J Cereb Blood Flow Metab 28:824–831
Goldman-Rakic PS (1998) The cortical dopamine system: role in memory and cognition. Adv Pharmacol 42:707–711
Arnold GL, Vladutiu CJ, Orlowski CC, Blakely EM, DeLuca J (2004) Prevalence of stimulant use for attentional dysfunction in children with phenylketonuria. J Inherit Metab Dis 27(2):137–143
Posner J, Gorman D, Nagel BJ (2009) Tyrosine supplements for ADHD symptoms with comorbid phenylketonuria. J Neuropsychiatry Clin Neurosci 21:228–230
Baieli S, Pavone L, Meli C, Fiumara A, Coleman M (2003) Autism and phenylketonuria. J Autism Dev Disord 33:201–204
Zafeiriou DI, Ververi A, Vargiami E (2007) Childhood autism and associated comorbidities. Brain Dev 29:257–272
Okado N (1999) Mechanisms for formation and maintenance of synapses mediated by biogenic amines: pathogenesis and therapy of mental retardation and developmental disabilities by genetic and epigenetic factors. Kaibogaku Zasshi. 74:351–362
Anderson PJ, Wood SJ, Francis DE, Coleman L, Warwick L, Casanelia S, Anderson VA, Boneh A (2004) Neuropsychological functioning in children with early-treated phenylketonuria: impact of white matter abnormalities. Dev Med Child Neurol 46:230–238
Anderson PJ, Leuzzi V (2010) White matter pathology in phenylketonuria. Mol Genet Metab 99:3–9
Christ SE, Steiner RD, Grange DK, Abrams RA, White DA (2006) Inhibitory control in children with phenylketonuria. Dev Neuropsychol. 30:845–864
Anderson PJ, Wood SJ, Francis DE, Coleman L, Anderson V, Boneh A (2007) Are neuropsychological impairments in children with early-treated phenylketonuria (PKU) related to white matter abnormalities or elevated phenylalanine levels? Dev Neuropsychol. 32(2):645–668
Conflict of interest
All authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Saad, K., Elserogy, Y., Abdel rahman, A.A. et al. ADHD, autism and neuroradiological complications among phenylketonuric children in Upper Egypt. Acta Neurol Belg 115, 657–663 (2015). https://doi.org/10.1007/s13760-014-0422-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-014-0422-8