Abstract
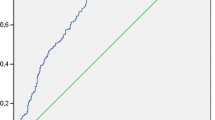
We aimed to compare the association of high-sensitivity C-reactive protein (CRP) and National Institutes of Health Stroke Scale (NIHSS) score with mortality risk and to determine the optimal threshold of CRP for prediction of mortality in ischemic-stroke patients. A series of 162 patients with first-ever ischemic-stroke admitted within 24 h after onset of symptoms was enrolled. CRP and NIHSS score were estimated on admission and their predictive abilities for mortality at 7 days were determined by logistic-regression analyses. Receiver-Operating Characteristic (ROC) curves were depicted to identify the optimal cut-off of CRP, using the maximum Youden-index and the shortest-distance methods. Deceased patients had higher levels of CRP and NIHSS on admission (8.87 ± 7.11 vs. 2.20 ± 4.71 mg/l for CRP, and 17.31 ± 6.36 vs. 8.70 ± 4.85 U for NIHSS, respectively, P < 0.01). CRP and NIHSS were correlated with each other (r 2 = 0.39, P < 0.001) and were also independently associated with increased risk of mortality [odds ratios (95 % confidence interval) of 1.16 (1.05–1.28) and 1.20 (1.07–1.35) for CRP and NIHSS, respectively, P < 0.01]. The areas under the ROC curves of CRP and NIHSS for mortality were 0.82 and 0.84, respectively. The CRP value of 2.2 mg/l was identified as the optimal cut-off value for prediction of mortality within 7 days (sensitivity: 0.81, specificity: 0.80). Thus, CRP as an independent predictor of mortality following ischemic-stroke is comparable with NIHSS and the value of 2.2 mg/l yields the optimum sensitivity and specificity for mortality prediction.

Similar content being viewed by others
References
Hansson GK (2005) Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med 352(16):1685–1695. doi:10.1056/NEJMra043430
Elias-Smale SE, Kardys I, Oudkerk M, Hofman A, Witteman JC (2007) C-reactive protein is related to extent and progression of coronary and extra-coronary atherosclerosis; results from the Rotterdam study. Atherosclerosis 195(2):e195–e202. doi:10.1016/j.atherosclerosis.2007.07.006
Hamer M, Chida Y, Stamatakis E (2010) Association of very highly elevated C-reactive protein concentration with cardiovascular events and all-cause mortality. Clin Chem 56(1):132–135. doi:10.1373/clinchem.2009.130740
Bova IY, Bornstein NM, Korczyn AD (1996) Acute infection as a risk factor for ischemic stroke. Stroke 27(12):2204–2206
Grau AJ (1997) Infection, inflammation, and cerebrovascular ischemia. Neurology 49(5 Suppl 4):S47–S51
Wang PY, Kao CH, Mui MY, Wang SJ (1993) Leukocyte infiltration in acute hemispheric ischemic stroke. Stroke 24(2):236–240
Fassbender K, Rossol S, Kammer T, Daffertshofer M, Wirth S, Dollman M, Hennerici M (1994) Proinflammatory cytokines in serum of patients with acute cerebral ischemia: kinetics of secretion and relation to the extent of brain damage and outcome of disease. J Neurol Sci 122(2):135–139
Dirnagl U, Iadecola C, Moskowitz MA (1999) Pathobiology of ischaemic stroke: an integrated view. Trends Neurosci 22(9):391–397. p[ii]:S0166-2236(99)01401-0
Nilupul Perera M, Ma HK, Arakawa S, Howells DW, Markus R, Rowe CC, Donnan GA (2006) Inflammation following stroke. J Clin Neurosci 13(1):1–8. doi:10.1016/j.jocn.2005.07.005
den Hertog HM, van Rossum JA, van der Worp HB, van Gemert HM, de Jonge R, Koudstaal PJ, Dippel DW (2009) C-reactive protein in the very early phase of acute ischemic stroke: association with poor outcome and death. J Neurol 256(12):2003–2008. doi:10.1007/s00415-009-5228-x
Gunstad J, Bausserman L, Paul RH, Tate DF, Hoth K, Poppas A, Jefferson AL, Cohen RA (2006) C-reactive protein, but not homocysteine, is related to cognitive dysfunction in older adults with cardiovascular disease. J Clin Neurosci 13(5):540–546. doi:10.1016/j.jocn.2005.08.010
Di Napoli M, Papa F, Bocola V (2001) C-reactive protein in ischemic stroke: an independent prognostic factor. Stroke 32(4):917–924
Lau AY, Wong EH, Leung TW, Mok VC, Wong KS (2012) Intravenous alteplase for Chinese patients with stroke and borderline eligibility. J Clin Neurosci 19(10):1383–1386. doi:10.1016/j.jocn.2012.01.027
Zandieh A, Kahaki ZZ, Sadeghian H, Fakhri M, Pourashraf M, Parviz S, Ghaffarpour M, Ghabaee M (2013) A simple risk score for early ischemic stroke mortality derived from National Institutes of Health Stroke Scale: a discriminant analysis. Clin Neurol Neurosurg 115(7):1036–1039. doi:10.1016/j.clineuro.2012.10.034
Kasner SE, Chalela JA, Luciano JM, Cucchiara BL, Raps EC, McGarvey ML, Conroy MB, Localio AR (1999) Reliability and validity of estimating the NIH stroke scale score from medical records. Stroke 30(8):1534–1537
Meyer BC, Lyden PD (2009) The modified National Institutes of Health Stroke Scale: its time has come. Int J Stroke 4(4):267–273. doi:10.1111/j.1747-4949.2009.00294.x
Mittal M, Rymer M, Lai SM (2012) Should all patients with mild ischemic stroke be excluded from therapeutic stroke trials? J Clin Neurosci 19(11):1486–1489. doi:10.1016/j.jocn.2012.03.008
Gu Y, Ni W, Jiang H, Ning G, Xu B, Tian Y, Xu F, Liao Y, Song D, Mao Y (2012) Efficacy of extracranial–intracranial revascularization for non-moyamoya steno-occlusive cerebrovascular disease in a series of 66 patients. J Clin Neurosci 19(10):1408–1415. doi:10.1016/j.jocn.2011.11.037
Adams HP Jr, Bendixen BH, Leira E, Chang KC, Davis PH, Woolson RF, Clarke WR, Hansen MD (1999) Antithrombotic treatment of ischemic stroke among patients with occlusion or severe stenosis of the internal carotid artery: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology 53(1):122–125
Nogueira RG, Liebeskind DS, Sung G, Duckwiler G, Smith WS (2009) Predictors of good clinical outcomes, mortality, and successful revascularization in patients with acute ischemic stroke undergoing thrombectomy: pooled analysis of the Mechanical Embolus Removal in Cerebral Ischemia (MERCI) and Multi MERCI Trials. Stroke 40(12):3777–3783. doi:10.1161/STROKEAHA.109.561431
Idicula TT, Brogger J, Naess H, Waje-Andreassen U, Thomassen L (2009) Admission C-reactive protein after acute ischemic stroke is associated with stroke severity and mortality: the ‘Bergen stroke study’. BMC Neurol 9:18. doi:10.1186/1471-2377-9-18
Aho K, Harmsen P, Hatano S, Marquardsen J, Smirnov VE, Strasser T (1980) Cerebrovascular disease in the community: results of a WHO collaborative study. Bull World Health Organ 58(1):113–130
Adams HP Jr, Bendixen BH, Kappelle LJ, Biller J, Love BB, Gordon DL, Marsh EE 3rd (1993) Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of Org 10172 in Acute Stroke Treatment. Stroke 24(1):35–41
Ormstad H, Aass HC, Lund-Sorensen N, Amthor KF, Sandvik L (2011) Serum levels of cytokines and C-reactive protein in acute ischemic stroke patients, and their relationship to stroke lateralization, type, and infarct volume. J Neurol 258(4):677–685. doi:10.1007/s00415-011-6006-0
Cho KH, Kang DW, Kwon SU, Kim JS (2009) Lesion volume increase is related to neurologic progression in patients with subcortical infarction. J Neurol Sci 284(1–2):163–167. doi:10.1016/j.jns.2009.05.013
Tei H, Uchiyama S, Usui T, Ohara K (2011) Diffusion-weighted ASPECTS as an independent marker for predicting functional outcome. J Neurol 258(4):559–565. doi:10.1007/s00415-010-5787-x
Saver JL, Johnston KC, Homer D, Wityk R, Koroshetz W, Truskowski LL, Haley EC (1999) Infarct volume as a surrogate or auxiliary outcome measure in ischemic stroke clinical trials. The RANTTAS investigators. Stroke 30(2):293–298
Muir KW, Weir CJ, Alwan W, Squire IB, Lees KR (1999) C-reactive protein and outcome after ischemic stroke. Stroke 30(5):981–985
Saposnik G, Hill MD, O’Donnell M, Fang J, Hachinski V, Kapral MK (2008) Variables associated with 7-day, 30-day, and 1-year fatality after ischemic stroke. Stroke 39(8):2318–2324. doi:10.1161/STROKEAHA.107.510362
Finlayson O, Kapral M, Hall R, Asllani E, Selchen D, Saposnik G (2011) Risk factors, inpatient care, and outcomes of pneumonia after ischemic stroke. Neurology 77(14):1338–1345. doi:10.1212/WNL.0b013e31823152b1
Esteghamati A, Zandieh A, Zandieh B, Khalilzadeh O, Esteghamati S, Meysamie A, Nakhjavani M (2012) Comparing abilities of different lipid measures in diagnosis of insulin resistance: a survey of risk factors of non-communicable diseases (SuRFNCD-2007) study. Metab Syndr Relat Disord 10(1):63–69. doi:10.1089/met.2011.0002
Esteghamati A, Zandieh A, Zandieh B, Khalilzadeh O, Meysamie A, Nakhjavani M, Gouya MM (2011) Leptin cut-off values for determination of metabolic syndrome: third national surveillance of risk factors of non-communicable diseases in Iran (SuRFNCD-2007). Endocrine 40(1):117–123. doi:10.1007/s12020-011-9447-4
Zandieh A, Esteghamati A, Morteza A, Noshad S, Khalilzadeh O, Gouya MM, Nakhjavani M (2012) Appropriate BMI cut-off values for identification of metabolic risk factors: third national surveillance of risk factors of non-communicable diseases in Iran (SuRFNCD-2007). Ann Hum Biol 39(6):484–489. doi:10.3109/03014460.2012.716860
Acknowledgments
This study was performed in partial fulfillment of the requirements for completing residency training of Dr. Shahrzad Mohebbi in neurology and received Grant No. 89-01-54-10354 from vice-chancellor for research of Tehran University of Medical Sciences (TUMS).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghabaee, M., Zandieh, A., Mohebbi, S. et al. Predictive ability of C-reactive protein for early mortality after ischemic stroke: comparison with NIHSS score. Acta Neurol Belg 114, 41–45 (2014). https://doi.org/10.1007/s13760-013-0238-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13760-013-0238-y