Abstract
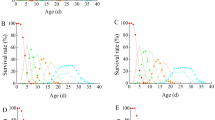
The peanut thrips, Enneothrips enigmaticus (Thysanoptera: Thrypidae), is an important pest of the peanut (Arachis hypogaea) in South America. Due to concerns about the environment and human health induced by the extensive use of pesticides in the management control of pests, environmentally and friendlier tactics must be targeted. Thus, this study investigates, for the first time, the behavior of Xylocoris sordidus (Hemiptera: Anthocoridae) as a biological control agent for E. enigmaticus. The methodology included no-choice tests to assess whether the predation rate varies according to the developmental stage of the prey, as well as the predator’s developmental stage with the highest predation capacity. Additionally, an analysis of the functional response of adult and 5th instar nymphs of X. sordidus exposed to different densities of E. enigmaticus nymphs (1, 2, 4, 8, 16, and 32) was conducted. The results confirm the predation of peanut thrips by X. sordidus, with a higher predation rate in the nymphal stages of the prey. There was no difference in predation capacity between predator nymphs and adults, and exhibiting a type II functional response. Therefore, the potential of X. sordidus as a biological control agent for E. enigmaticus is confirmed, showing the importance of adopting measures to preserve this predator in peanut crops.


Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the first author upon reasonable request.
References
Albashir A., Aljetlawi Erik, Sparrevik Kjell, Leonardsson (2004) Prey–predator size‐dependent functional response: derivation and rescaling to the real world. J Anim Ecol 73(2):239–252. https://doi.org/10.1111/jae.2004.73.issue-2https://doi.org/10.1111/j.0021-8790.2004.00800.x
Almeida PRD, Arruda HVD (1962) Controle do tripes causador do prateamento das fôlhas do amendoim, por meio de inseticidas. Bragantia 21:679–687. https://doi.org/10.1590/S0006-87051962000100039
Andow DA, Barratt BI, Pfannenstiel RS, Paula DP (2021) Exotic generalist arthropod biological control agents: need to improve environmental risk assessment to ensure safe use. Biol Control 66:1–8. https://doi.org/10.1007/s10526-020-10067-2
Arbogast RT, Flaherty BR, Press JW (1983) Demography of the predaceous bug Xylocoris sordidus (Reuter). Am Midl Nat 109:398–405. https://doi.org/10.2307/2425421
Arnó J, Roig J, Riudavets J (2008) Evaluation of Orius majusculus and O. laevigatus as predators of Bemisa tabaci and estimation of their prey preference. Biol Control 44:1–6. https://doi.org/10.1016/j.biocontrol.2007.10.009
Atakan E (2006) Associations between Frankliniella spp. and Orius niger populations in cotton. Phytoparasitica 34:221–234. https://doi.org/10.1007/BF02980949
Atakan E, Pehlivan S (2020) Influence of weed management on the abundance of thrips species (Thysanoptera) and the predatory bug, Orius niger (Hemiptera: Anthocoridae) in citrus mandarin. Appl Entomol Zool 55:71–81. https://doi.org/10.1007/s13355-019-00655-9
Baez I, Reitz SR, Funderburk JE (2004) Predation by Orius insidiosus (Heteroptera: Anthocoridae) on life stages and species of Frankliniella flower thrips (Thysanoptera: Thripidae) in pepper flowers. Environ Entomol 33:662–670. https://doi.org/10.1603/0046-225X-33.3.662
Baker BP, Green TA, Loker AJ (2020) Biological control and integrated pest management in organic and conventional systems. Biol Control 140:104095. https://doi.org/10.1016/j.biocontrol.2019.104095
Ballal CR, Yamada K (2016) Anthocorid predators. In: Omkar (Ed) Ecofriendly pest management for food security, 1st edn. Elsevier, New York, pp 183–216
Barzman M, Bàrberi P, Birch ANE, Boonekamp P, Dachbrodt-Saaydeh S, Graf B, Sattin M (2015) Eight principles of integrated pest management. Agron Sustain Dev 35:1199–1215. https://doi.org/10.1007/s13593-015-0327-9
Calore RA, Ferreira MC, Galli JC (2015) Efeitos de adjuvantes no controle de Enneothrips flavens Moulton, 1941 (Thysanoptera: Thripidae) na cultura do amendoim. Bras Cienc Agrar 10:74–81. https://doi.org/10.5039/agraria.v10i1a5043
Clercq PD (2002) Dark clouds and their silver linings: exotic generalist predators in augmentative biological control. Neotrop Entomol 31:169–176. https://doi.org/10.1590/S1519-566X2002000200001
Dawes JHP, Souza M (2013) A derivation of Holling’s type I, II and III functional responses in predator–prey systems. J Theor Biol 327:11–22. https://doi.org/10.1016/j.jtbi.2013.02.017
Deguine JP, Aubertot JN, Flor RJ, Lescourret F, Wyckhuys KA, Ratnadass A (2021) Integrated pest management: good intentions, hard realities. A Review Agron Sustain Dev 41:38. https://doi.org/10.1007/s13593-021-00689-w
Di Stefano J (2005) Effect size estimates and confidence intervals: an alternative focus for the presentation and interpretation of ecological data. In: Burk AR (ed) New Trends in ecology research. Nova Science, New York, pp 71–102
Diehl E, Sereda E, Wolters V, Birkhofer K (2013) Effects of predator specialization, host plant and climate on biological control of aphids by natural enemies: a meta-analysis. J Appl Ecol 50:262–270. https://doi.org/10.1111/1365-2664.12032
Doğramaci M, Arthurs SP, Chen J, McKenzie C, Irrizary F, Osborne L (2011) Management of chilli thrips Scirtothrips dorsalis (Thysanoptera: Thripidae) on peppers by Amblyseius swirskii (Acari: Phytoseiidae) and Orius insidiosus (Hemiptera: Anthocoridae). Biol Control 59:340–347. https://doi.org/10.1016/j.biocontrol.2011.09.008
Donnelly BE, Phillips TW (2001) Functional response of Xylocoris flavipes (Hemiptera: Anthocoridae)-effects of prey species and habitat. Environ Entomol 30:617–624. https://doi.org/10.1603/0046-225X-30.3.617
Farinelli JBM, Horita K, Santos DFL (2018) Analysis of the economic viability of the peanut crop in the region of Jaboticabal, São Paulo. Científica 46:215–220. https://doi.org/10.15361/1984-5529.2018v46n3p215-220
Fernandes OA, Michelotto MD (2022) Pragas do amendoim. In: Rada A et al (eds) Manual de entomologia: pragas das culturas, 1st edn. Ouro fino, MG, Agronômica Ceres, pp 41–56
Ge Y, Camara I, Wang Y, Liu P, Zhang L, Xing Y, Shi W (2018) Predation of Aphis craccivora (Hemiptera: Aphididae) by Orius sauteri (Hemiptera: Anthocoridae) under different temperatures. J Econ Entomol 111:2599–2604. https://doi.org/10.1093/jee/toy255
Gitonga LM, Overholt WA, Löhr B, Magambo JK, Mueke JM (2002) Functional response of Orius albidipennis (Hemiptera: Anthocoridae) to Megalurothrips sjostedti (Thysanoptera: Thripidae). Biol Control 24:1–6. https://doi.org/10.1016/S1049-9644(02)00001-4
Hassanzadeh-Avval M, Sadeghi-Namaghi H, Fekrat L (2019) Factors influencing functional response, handling time and searching efficiency of Anthocoris minki Dohrn (Hem.: Anthocoridae) as predator of Psyllopsis repens Loginova (Hem.: Psyllidae). Phytoparasitica 47:341–350. https://doi.org/10.1007/s12600-019-00739-w
Holling CS (1959) Some characteristics of simple types of predation and parasitism. Can Entomol 91:385–398. https://doi.org/10.4039/Ent91385-7
Holling CS (1966) The functional response of invertebrate predators to prey density. Mem Entomol Soc Can 98:5–86. https://doi.org/10.4039/entm9848fv
Juliano SA (2001) Non-linear curve fitting: predation and functional response curves. In: Scheiner SM, Gurevitch J (eds) Design and analysis of ecological experiments, 2nd edn. Chapman & Hall, New York, pp 178–196
Kheirodin A, Simmons AM, Legaspi JC, Grabarczyk EE, Toews MD, Roberts PM, Schmidt JM (2020) Can generalist predators control Bemisia tabaci? Insects 11:823. https://doi.org/10.3390/insects11110823
Koss AM, Snyder WE (2005) Alternative prey disrupt biocontrol by a guild of generalist predators. Biol Control 32(2):243–251. https://doi.org/10.1016/j.biocontrol.2004.10.002
Krey KL, Blubaugh CK, Chapman EG, Lynch CA, Snyder GB, Jensen AS, Snyder WE (2017) Generalist predators consume spider mites despite the presence of alternative prey. Biol Control 115:157–164. https://doi.org/10.1007/BF02371908
LeCato GL (1976) Predation by Xylocoris flavipes [Hem.: Anthocoridae]: Influence of stage, species and density of prey and of starvation and density of predator. Entomophaga 21:217–221. https://doi.org/10.1007/BF02371908
Lima ÉFB, Alencar ÁRSD, Nanini F, Michelotto MD, Corrêa AS (2022) “Unmasking the Villain”: integrative taxonomy reveals the real identity of the key pest (Thysanoptera: Thripidae) of peanuts (Arachis hypogaea L.) in South America. Insects 13:120. https://doi.org/10.3390/insects13020120
Lin T, Zeng Z, Chen Y, You Y, Hu J, Yang F, Wei H (2021) Compatibility of six reduced-risk insecticides with Orius strigicollis (Heteroptera: Anthocoridae) predators for controlling Thrips hawaiiensis (Thysanoptera: Thripidae) pests. Ecotoxicol Environ Saf 226:112812. https://doi.org/10.1016/j.ecoenv.2021.112812
Liu P, Jia W, Zheng X, Zhang L, Sangbaramou R, Tan S, Shi W (2018) Predation functional response and life table parameters of Orius sauteri (Hemiptera: Anthocoridae) feeding on Megalurothrips usitatus (Thysanoptera: Thripidae). Fla Entomol 101:254–259. https://doi.org/10.1653/024.101.0216
Loomans AJ (2021) Every generalist biological control agent requires a special risk assessment. Biocontrol 66:23–35. https://doi.org/10.1007/s10526-020-10022-1
Messelink GJ, Janssen A (2014) Increased control of thrips and aphids in greenhouses with two species of generalist predatory bugs involved in intraguild predation. Biol Control 79:1–7. https://doi.org/10.1016/j.biocontrol.2014.07.009
Michelotto MD, De Godoy IJ, Pirotta MZ, Santos JF, Finoto EL, Pereira Fávero A (2017) Resistance to thrips (Enneothrips flavens) in wild and amphidiploid Arachis species. PLoS ONE 12:e0176811. https://doi.org/10.1371/journal.pone.0176811
Miranda MMM, Picanço MC, Zanuncio JC, Guedes RNC (1998) Ecological life table of Tuta absoluta (Meyrick) (Lepidoptera: Gelechiidae). Biocontrol Sci Technol 8:597–606. https://doi.org/10.1080/09583159830117
Moraes ARAD, Lourenção AL, Godoy IJD, Teixeira GDC (2005) Infestation by Enneothrips flavens Moulton and yield of peanut cultivars. Sci Agric 62:469–472. https://doi.org/10.1590/S0103-90162005000500010
Naranjo SE, Ellsworth PC, Frisvold GB (2015) Economic value of biological control in integrated pest management of managed plant systems. Annu Rev Entomol 60:621–645. https://doi.org/10.1146/annurev-ento-010814-021005
Peñalver-Cruz A, Alvarez-Baca JK, Alfaro-Tapia A, Gontijo L, Lavandero B (2019) Manipulation of agricultural habitats to improve conservation biological control in South America. Neotrop Entomol 48:875–898. https://doi.org/10.1007/s13744-019-00725-1
Perdikis D, Fantinou A, Lykouressis D (2011) Enhancing pest control in annual crops by conservation of predatory Heteroptera. Biol Control 59:13–21. https://doi.org/10.1016/j.biocontrol.2011.03.014
Pervez A, Omkar A (2005) Functional responses of coccinellid predators: an illustration of a logistic approach. J Insect Sci 5:1–6. https://doi.org/10.1093/jis/5.1.5
Pijnakker J, Vangansbeke D, Duarte M, Moerkens R, Wäckers FL (2020) Predators and parasitoids-in-first: From inundative releases to preventative biological control in greenhouse crops. Front Sustain Food Syst 4:595630. https://doi.org/10.3389/fsufs.2020.595630
Pilkington LJ, Messelink G, van Lenteren JC, Le Mottee K (2010) “Protected Biological Control”–biological pest management in the greenhouse industry. Biol Control 52:216–220. https://doi.org/10.1016/j.biocontrol.2009.05.022
Queiroz OS, Ramos RS, Gontijo LM, Picanço MC (2015) Functional response of three species of predatory pirate bugs attacking eggs of Tuta absoluta (Lepidoptera: Gelechiidae). Environ Entomol 44:246–251. https://doi.org/10.1093/ee/nvu026
Rahman MA, Sarker S, Ham E, Lee JS, Lim UT (2022) Prey preference of Orius minutus and its functional response in comparison that of O. laevigatus, on Tetranychus urticae. J Asia Pac Entomol 25:101912. https://doi.org/10.1016/j.aspen.2022.101912
Rashedi A, Rajabpour A, Sohani NZ, Rasekh A (2020) Prey stage preference and functional response of Orius albidipennis (Hetetroptera, Anthocoridae) to Aphis fabae (Homomoptera, Aphididae). Int J Trop Insect Sci 40:13–19. https://doi.org/10.1007/s42690-019-00045-2
Rogers DJ (1972) Random search and insect population models. J Animal Ecol 41:369–383. https://doi.org/10.2307/3474
Salehi Z, Yarahmadi F, Rasekh A, Sohani NZ (2016) Functional responses of Orius albidipennis Reuter (Hemiptera, Anthocoridae) to Tuta absoluta Meyrick (Lepidoptera, Gelechiidae) on two tomato cultivars with different leaf morphological characteristics. Entomol Gen 36:127–136
Santos NAD, Bortoli SAD (2018) Xylocoris sordidus (Reuter) (Hemiptera: Anthocoridae): possível agente de controle biológico aplicado. Universidade de São Paulo, Thesis
Santos DFL, Silva BL, MoraesFarinelli JB, Horita K, Souza CAF, Montoro SB (2019) Economic viability of peanut production on leased land in the Jaboticabal region of São Paulo state, Brazil. Revista Agro@mbiente On-line 13:142–154. https://doi.org/10.18227/1982-8470ragro.v13i0.5342
Santos NA, Ramalho DG, Marques HM, Godoy JDS, De Bortoli CP, Magalhães GO, De Bortoli SA (2020) Interaction between the predator Xylocoris sordidus and Bacillus thuringiensis bioinsecticides. Entomol Exp Appl 168:371–380. https://doi.org/10.1111/eea.12896
Sarker AC, Islam W, Parween S (2016) Host-stage specific effects on the biological parameters of Xylocoris flavipes (reuter) preyed on Cryptolestes pussilus (SCHON.). Trop Agric Res 19:305–312
SAS Institute (2022) SAS/Graph Software: Reference, Vol. 1, Version 8. SAS Institute, Cary, NC, USA
Saulich AK, Musolin DL (2009) Seasonal development and ecology of anthocorids (Heteroptera, Anthocoridae). Entomol Rev 89:501–528. https://doi.org/10.1134/S0013873809050017
Seko T, Miura K (2008) Functional response of the lady beetle Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) on the aphid Myzus persicae (Sulzer) (Homoptera: Aphididae). Appl Entomol Zool 43:341–345. https://doi.org/10.1303/aez.2008.341
Sherratt TN, Harvey IF (1993) Frequency-dependent food selection by arthropods: a review. Biol J Linn Soc 48:167–186. https://doi.org/10.1111/j.1095-8312.1993.tb00885.x
Silva LP, Souza IL, Marucci RC, Guzman-Martinez M (2023) Doru luteipes (Dermaptera: Forficulidae) and Orius insidiosus (Hemiptera: Anthocoridae) as nocturnal and diurnal predators of thrips. Neotrop Entomol 52:263–272. https://doi.org/10.1007/s13744-022-00982-7
Stiling P, Cornelissen T (2005) What makes a successful biocontrol agent? A meta-analysis of biological control agent performance. Biol Control 34:236–246. https://doi.org/10.1016/j.biocontrol.2005.02.017
Tavares AM, Torres JB, Silva-Torres CS, Vacari AM (2013) Behavior of Montandoniola confusa Streito & Matocq (Hemiptera: Anthocoridae) preying upon gall-forming thrips Gynaikothrips ficorum Marchal (Thysanoptera: Phlaeothripidae). Biol Control 67:328–336. https://doi.org/10.1016/j.biocontrol.2013.09.004
Torres JB, Bueno ADF (2018) Conservation biological control using selective insecticides–a valuable tool for IPM. Biol Control 126:53–64. https://doi.org/10.1016/j.biocontrol.2018.07.012
Tuan SJ, Lin YH, Peng SC, Lai WH (2016) Predatory efficacy of Orius strigicollis (Hemiptera: Anthocoridae) against Tetranychus urticae (Acarina: Tetranychidae) on strawberry. J Asia Pac Entomol 19:109–114. https://doi.org/10.1016/j.aspen.2015.12.007
Uiterwaal SF, DeLong JP (2018) Multiple factors, including arena size, shape the functional responses of ladybird beetles. J Appl Ecol 55:2429–2438. https://doi.org/10.1111/1365-2664.13159
van Lenteren JC, Hemerik L, Lins JC Jr, Bueno VH (2016) Functional responses of three neotropical mirid predators to eggs of Tuta absoluta on tomato. Insects 7:34. https://doi.org/10.3390/insects7030034
van Lenteren JC, Bolckmans K, Köhl J, Ravensberg WJ, Urbaneja A (2018) Biological control using invertebrates and microorganisms: plenty of new opportunities. Biocontrol 63:39–59. https://doi.org/10.1007/s10526-017-9801-4
Yang NW, Zang LS, Wang S, Guo JY, Xu HX, Zhang F, Wan FH (2014) Biological pest management by predators and parasitoids in the greenhouse vegetables in China. Biol Control 68:92–102. https://doi.org/10.1016/j.biocontrol.2013.06.012
Zeng G, Zhi JR, Zhang CR, Zhang T, Ye JQ, Zhou L, Ye M (2021) Orius similis (Hemiptera: Anthocoridae): a promising candidate predator of Spodoptera frugiperda (Lepidoptera: Noctuidae). J Econ Entomol 114:582–589. https://doi.org/10.1093/jee/toaa318
Zhang Q, Zhang R, Zhang Q, Ji D, Zhou X, Jin L (2021) Functional response and control potential of Orius sauteri (Hemiptera: Anthocoridae) on tea thrips (Dendrothrips minowai Priesner). Insects 12:1132. https://doi.org/10.3390/insects12121132
Funding
CNPq (National Council for Scientific and Technological Development) and Capes (Coordination for the Improvement of Higher Education Personnel).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. All authors contributed to material preparation and data collection. Analysis was performed by D. G. R. All authors conducted an extensive literature review and contributed to the article's writing and result discussion. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no competing interests.
Additional information
Edited by Lessando Moreira Gontijo
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Oliveira, S.J., Nascimento, V.F., de Lacerda, L.B. et al. Predator–Prey Interaction Between Xylocoris sordidus (Hemiptera: Anthocoridae) and Enneothrips enigmaticus (Thysanoptera: Thripidae). Neotrop Entomol 53, 391–399 (2024). https://doi.org/10.1007/s13744-023-01126-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-023-01126-1