Abstract
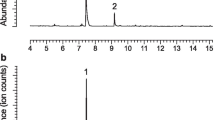
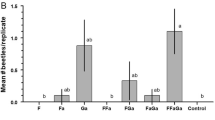
The subfamily Cerambycinae, one of the most diverse in longhorn beetles, is well known for its remarkable chemical parsimony in male-emitted pheromones. Conserved shared structural motifs have been reported in numerous species, sometimes working in combination with plant volatile kairomones. Among other compounds, the most ubiquitous male pheromone in cerambycine species is 3-hydroxyhexan-2-one. We conducted field trials using intercept traps baited with 3-hydroxyhexan-2-one and observed abundant captures of several Neotropical cerambycine species. These were Retrachydes thoracicus thoracicus (Olivier), Megacyllene acuta (Germar), Compsocerus violaceus (White), and Cotyclytus curvatus (Germar) in high numbers, as well as Chydarteres striatus striatus (Fabricius) and Odontocroton flavicauda (Bates) in smaller numbers. When ethanol was added to the traps, a remarkable increase in the attractiveness of 3-hydroxyhexan-2-one was observed for R. thoracicus thoracicus and M. acuta. Adding ethanol also resulted in the capture of Chrysoprasis aurigena (Germar). Finally, incidental catches in pheromone-baited traps of Trachelissa maculicollis (Audinet-Serville), Neoclytus pusillus (Laporte & Gory), Achryson unicolor (Bruch, 1908) and Achryson surinamum (Linnaeus), Megacyllene mellyi (Chevrolat) and Thelgetra adustus (Burmeister) were also observed. Pheromone chemistry has been reported for C. curvatus, M. acuta and N. pusillus, all three producing 3-hydroxyhexan-2-one, and for C. aurigena and A. surinamum, which produce other compounds. Our findings suggest that the captured species probably produce 3-hydroxyhexan-2-one for their pheromone communication system. Alternatively, they might be “eavesdropping” on the pheromones of other cerambycine species. The probable synergistic effect of ethanol is likely explained from its kairomonal role as a volatile cue for plant stress or ripeness.



Similar content being viewed by others
References
Aguirre Gil OJ et al (2021) Screening known Cerambycidae pheromones for activity with the Peruvian fauna. Agr Forest Entomol. https://doi.org/10.1111/afe.12454
Amorós ME, Lagarde L, Do Carmo H, Heguaburu V, González A (2020) Pheromone chemistry of the citrus borer, Diploschema rotundicolle (Coleoptera: Cerambycidae). J Chem Ecol 46:809–819. https://doi.org/10.1007/s10886-020-01203-4
Barriga-Tuñón JE (2009) Coleoptera Neotropical. http://www.coleoptera-neotropical.org/paginas/2_PAISES/Uruguay/Cerambycidae/cerambycinae-uru.html. Accessed June 2019
Bentancourt CM, Scatoni IB (2010) Guía de insectos y ácaros de importancia agrícola y forestal en el Uruguay. 3 edn. Facultad de Agronomía, Montevideo
Bezark LG, Monné MA (2019) Checklist of the Oxypeltidae, Vesperidae, Disteniidae and Cerambycidae, (Coleoptera) of the Western Hemisphere 2019 Edition (updated through 31 December 2018).
Brockerhoff EG, Jones DC, Kimberley MO, Suckling DM, Donaldson T (2006) Nationwide survey for invasive wood-boring and bark beetles (Coleoptera) using traps baited with pheromones and kairomones. For Ecol Manag 228:234–240
Bruhn JC, Beltrame JBI (1980) Los taladros Praxithea derourei Chabrilac, Trachyderes thoracicus (Olivier) y T. striatus (Fabricius) (Coleoptera: Cerambycidae) y su relación con los cultivos de manzanas en Uruguay. Investigaciones Agronomicas 1:11–14
Costa MKC, Diodato MA, Fernandes JPP, Santos JPS (2019) Insetos nocivos a Prosopis sp. no Rio Grande do Norte (Brasil) e Piura (Peru). Agrop Cient Semiárido 15:158–116
Di Lorio ORR (1997) Plantas hospedadoras de Cerambycidae (Coleoptera) en el Espinal periestépico y en la provincia de Buenos Aires, Argentina. Revista de Biología Tropical 44(3)/45(1):159–165
Duffy EAJ (1953) A monograph of the immature stages of British and imported timber beetles (Cerambycidae). Brit Mus (Nat Hist), London
Fan J et al (2018) Multi-component blends for trapping native and exotic longhorn beetles at potential points-of-entry and in forests. J Pest Sci. https://doi.org/10.1007/s10340-018-0997-6
Graham EE, Mitchell RF, Reagel PF, Barbour JD, Millar JG, Hanks LM (2010) Treating panel traps with a fluoropolymer enhances their efficiency in capturing cerambycid beetles. J Econ Entomol 103:641–647
Hanks LM, Millar JG (2013) Field bioassays of cerambycid pheromones reveal widespread parsimony of pheromone structures, enhancement by host plant volatiles, and antagonism by components from heterospecifics. Chemoecology 23:21–44
Hanks LM, Millar JG (2016) Sex and aggregation-sex pheromones of cerambycid beetles: basic science and practical applications. J Chem Ecol 42:631–654. https://doi.org/10.1007/s10886-016-0733-8
Hanks LM, Millar JG, Mongold-Diers JA, Wong JCH, Meier LR, Reagel PF, Mitchell RF (2012) Using blends of cerambycid beetle pheromones and host plant volatiles to simultaneously attract a diversity of cerambycid species. Can J for Res 42:1050–1059
Heguaburu V, de Carmo H, Parpal F, Amorós ME, González A (2017) Synthesis of aggregation pheromone components of cerambycid species through α-hydroxylation of alkylketones. Tetrahedron Letters 58:1738–1741
Holdefer Woldan DR (2007) Análise faunística de cerambycidae (Coleoptera) em duas situações florísticas no município de União da Vitória – Paraná. UNIVERSIDADE COMUNITÁRIA REGIONAL DE CHAPECÓ
Hothorn T, Bretz F, Westfall P (2008) Simultaneous Inference in General Parametric Models. Biometrical J 50:346–363
Lindemberg Martins Mesquita A, Teles Portela Policarpo G, Emilson Cardoso J, Cavalcante de Souza Mota M (2017) Novas Ocorrências de Cerambycidae (Insecta:Coleoptera) em cajueiro no Brasil e recomendações de manejo vol Comunicado Técnico, 231. Embrapa Agroindústria Tropical
Machado VS, Botero JP, Carelli A, Cupello M, Quintino HY, Simões MVP (2012) Host plants of Cerambycidae and Vesperidae (Coleoptera, Chrysomeloidea) from South America. Revista Brasileira De Entomolgia 56:186–198
Martins UR, Galileo MHM (2011) Tribo Clytini Mulsant, 1839. In: Martins UR GM (ed) Cerambycidae Sul-Americanos (Coleoptera): taxonomia, vol 12. Sociedade Brasileira de Entomologia, São Paulo, pp 8–64
Millar JG et al (2018a) Identifying possible pheromones of cerambycid beetles by field testing known pheromone components in four widely separated regions of the United States. J Econ Entomol 111:252–259. https://doi.org/10.1093/jee/tox31
Millar JG et al (2018b) (2017) Identifying possible pheromones of cerambycid beetles by field testing known pheromone components in four widely separated regions of the United States. J Econ Entomol 111(1):252–259
Millar JG, Richards AB, Halloran S, Zou Y, Boyd EA, Quigley KN, Hanks LM (2019) Pheromone identification by proxy: identification of aggregation-sex pheromones of North American cerambycid beetles as a strategy to identify pheromones of invasive Asian congeners. J Pest Sci 92:213–220. https://doi.org/10.1007/s10340-018-0962-4
Miller DR, Crowe CM, Mayo PD, Reid LS, Silk PJ, Sweeney JD (2017) Interactions between ethanol, syn-2,3-hexanediol, 3-hydroxyhexan-2-one, and 3-hydroxyoctan-2-one lures on trap catches of hardwood longhorn beetles in Southeastern United States. J Econ Entomol 1–10. https://doi.org/10.1093/jee/tox188
Mitchell RF, Millar JG, Hanks LM (2013) Blends of (R)-3-hydroxyhexan-2-one and alkan-2-ones identified as potential pheromones produced by three species of cerambycid beetles. Chemoecology 23:121–127
Monné M (2021) Catalogue of the Cerambycidae (Coleoptera) of the Neotropical Region. Part I. Subfamily Cerambycinae. Available from cerambyxcat@com/Part 1 Cerambycinae. pdf. Accessed August 2021
Monné MA, Bezark L (2009) Checklist of the Cerambycidae, or longhorned beetles (Coleoptera) of the Western Hemisphere. https://www.cerambycoidea.com/titles/monnebezark2009.pdf.
Monné MM, Bianchi M, Sánchez A, Escudero R (2002) Cerambícidos (Coleoptera) que atacan Eucalyptus globulus y Eucalyptus grandis en Uruguay. Agrociencia 6:63–68
RStudioTeam (2015) RStudio: Integrated Development for R. RStudio. Boston, MA Patent
Silva WD, Hanks LM, Alvarez JCS, Madalon FZ, Bento JMS, Bello JE, Millar JG (2020) Variations on a theme: two structural motifs create species-specific pheromone channels for multiple species of South American cerambycid beetles Insects: Special Issue “Ecology and Management of Invasive Insects in Forest Ecosystems” 11 https://doi.org/10.3390/insects11040222
Silva WD, Millar JG, Hanks LM, Bento JMS (2016) 10-methyldodecanal, a novel attractant pheromone produced by males of the south american cerambycid beetle. Eburodacrys Vittata 11. https://doi.org/10.1371/journal.pone.0160727
Silva WD, Millar JG, Hanks LM, Costa CM, Leite MOG, Tonelli M, Bento JMS (2018) Interspecific cross-attraction between the south american cerambycid beetles Cotyclytus curvatus and Megacyllene acuta is averted by minor pheromone components. J Chem Ecol 44:268–275. https://doi.org/10.1007/s10886-018-0933-5
Silva WD, Zou Y, Bento JMS, Hanks LM, millar JG, (2017) Aggregation-sex pheromones and likely pheromones of 11 South American Cerambycid beetles, and partitioning of pheromone channels. Front Ecol Evol 5:1–9
Wang Q (2017) Cerambycidae of the world: biology and pest management Cerambycidae of the world: biology and pest management. CRC Press, Boca Raton
Acknowledgements
Technical advice and field assistance was kindly provided by Yamandú Pochintesta, Martín Lanfranco, Ramiro Vacca, Gabriel Bueno, Alejandro Borges, José Berreta, Johana Dalla Valle, Gustavo Rodríguez, Roy Mazondo, Anna Paula Burgueño, Diana Valle, and Federico Rodrigo.
Funding
The authors wish to thank financial support from CSIC (Comisión Sectorial de Investigación Científica) Universidad de la República, PEDECIBA (Programa para el Desarrollo de las Ciencias Básicas, Uruguay), and INIA (Instituto Nacional de Investigación Agropecuaria).
Author information
Authors and Affiliations
Contributions
María Eugenia Amorós and Andrés González contributed to the study conception and design, and wrote the manuscript. Lautaro Lagarde and María Eugenia Amorós performed material preparation, data collection, and analysis. Hugo Do Carmo, Vivivana Heguaburu, and José Buenahora contributed with the synthetic compounds. Marcela Monné identified the insects.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Edited by Gabriel Manrique
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Amorós, M.E., Lagarde, L., Do Carmo, H. et al. Trapping of Retrachydes thoracicus thoracicus (Olivier) and Other Neotropical Cerambycid Beetles in Pheromone- and Kairomone-Baited Traps. Neotrop Entomol 51, 386–396 (2022). https://doi.org/10.1007/s13744-022-00955-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-022-00955-w