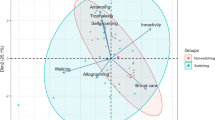
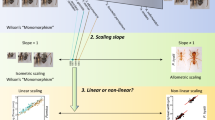
Abstract
In polymorphic ants, whose workers display continuous size distribution, each subcaste occupies a phenotypic space, usually with diffuse morphological boundaries. These morphological differences are closely associated to size by allometry although the environment also plays a key role that affects the fitness of the species. In Camponotus borellii Emery, the species selected as a study model, workers exhibit a continuous increase in size; geometric morphometric (GM) was used over four morphological traits: head capsule, clypeus, pronotum, and mesosoma, in order to assess (1) changes in shape, among the worker caste; (2) the influence of allometry on such changes; and (3) pronotum shape in respect to the head so as to infer which factors may influence the polymorphic development of the worker caste. The results indicated that the pronotum is organized into two highly integrated functional modules (neck and shield), corresponding to one developmental module. GM shows a similar pattern to that obtained for linear morphometry, though the worker ratio was different along continuous size distribution due to shape changes in two traits, with are also useful for delimiting modular units: (1) rounded shape of the posterior region of the head in minor workers; (2) shape of the pronotum, especially its anterior region, henceforth, neck, which widens as a consequence of the higher development of its central region, henceforth, shield, in major workers. The relevance of these results is discussed regarding functional morphology (pronotum in relation to the head), work division, and development of the worker caste.







Similar content being viewed by others
References
Abouheif E, Wray GA (2002) Evolution of the gene network underlying wing polyphenism in ants. Science 297:249–252. https://doi.org/10.1126/science.1071468
Adams DC, Rohlf FJ, Slice DE (2013) A field comes of age: geometric morphometrics in the 21st century. Hystrix, the Italian Journal of Mammalogy 24(1):1–8
Alvarado S, Rajakumar R, Abouheif E, Szyf M (2015) Epigenetic variation in the Egfr gene generates quantitative variation in a complex trait in ants. Nat Commun 6:6513. https://doi.org/10.1038/ncomms7513
Anderson P, Rivera M, Suarez A (2020) “Simple” biomechanical model for ants reveals how correlated evolution among body segments minimizes variation in center of mass as heads get larger. Integrative and Comparative Biology. icaa027 doi:https://doi.org/10.1093/icb/icaa027
Araujo MB, Tschinkel WR (2010) Worker allometry in relation to colony size and social form in the fire ant Solenopsis invicta. J Insect Sci 10(94):1–10. https://doi.org/10.1673/031.010.9401
Breuker CJ, Debat V, Klingenberg CP (2006) Functional evo-devo. Trends in Ecology and Evolution 21(9):488–492. https://doi.org/10.1016/j.tree.2006.06.003
Brian MV (1956) Studies of caste differentiation in Myrmica rubra L. 4. Controlled larval nutrition. Insect Soc 3:369–394. https://doi.org/10.1007/BF02225758
Brian MV (1974) Caste differentiation in Myrmica rubra: the role of hormones. J Insect Physiol 20(7):1351–1365. https://doi.org/10.1016/0022-1910(74)90238-8
Cheverud JM (1984) Quantitative genetics and developmental constraints on evolution by selection. J Theor Biol 110(2):155–171. https://doi.org/10.1016/S0022-5193(84)80050-8
Crispo E (2007) The Baldwin effect and genetic assimilation: revisiting two mechanisms of evolutionary change mediated by phenotypic plasticity. Evolution 61(11):2469–2479. https://doi.org/10.1111/j.1558-5646.2007.00203.x
Debat V, Debelle A, Dworkin I (2009) Plasticity, canalization, and developmental stability of the Drosophila wing: joint effects of mutation and developmental temperature. Evolution 63(11): 2864–2876. doi: 10.111/j.1558-5646.2009.00774.x
Diniz-Filho JA, Von Zuber CJ, Fowler HG, Schlindwein MN, Bueno OC (1994) Multivariate morphometrics and allometry in a polymorphic ant. Insect Soc 41(2):153–163. https://doi.org/10.1007/BF01240475
Espadaler X, Retana J, Cerdá X (1990) The caste system of Camponotus foreli Emery (Hymenoptera: Formicidae). Sociobiology 17(2):299–312
Friedman NR, Lecroq Bennet B, Fischer G, Sarnat EM, Huang J, Lacey Knowles L, Economo EP (2020) Macroevolutionary integration of phenotypes within and across ant worker castes. Ecology and Evolution 10:9371–9383. https://doi.org/10.1002/ece3.6623
Gallardo MH (2017) Evolución: el curso de la vida. Primera Edición Electrónica. http://sitiosciencias.uach.cl/EvolucionElCursodelaVida2017.pdf Accessed June, 5 2019
Hammer Ø, Harper DA, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):9pp
Hölldobler B, Wilson EO (1990) The ants. The Belknap Press of Harvard University Press, Cambridge, Massachusetts, p 732
Kaspari M, Weiser MD (1999) The size-grain hypothesis and interspecific scaling. Funct Ecol 13(4):530–538. https://doi.org/10.1046/j.1365-2435.1999.00343.x
Keller R, Peeters C, Beldade P (2014) Evolution of thorax architecture in ant castes highlights trade-off between flight and ground behaviors. eLIFE 3:e01539. https://doi.org/10.7554/eLife.01539
Klingenberg CP (2009) Morphometric integration and modularity in configurations of landmarks: tools for evaluating a priori hypotheses. Evolution and Developmental 11(4):405–421. https://doi.org/10.1111/j.1525-142x.2009.00347.x
Klingenberg CP (2011) MorphoJ: an integrated software package for geometric morphometrics. Mol Ecol Resour 11:353–357. https://doi.org/10.1111/j.1755-0998.2010.02924.x
Kusnezov N (1951) El género Camponotus (Hymenoptera: Formicidae) en la Argentina. Acta Zoológica Lilloana XII:183–252
Laciny A, Zettel H, Druzhinina I (2016) Workers, soldiers, and gynes – morphometric characterization and description of the female castes of Camponotus singularis (Smith, 1858) (Hymenoptera: Formicidae). Deutsche Entomologische Zeitschrift 63(2):183–193. https://doi.org/10.3897/dez.63.9435
Liu S, Richter A, Stoessel A, Beutel RG (2019) The mesosomal anatomy of Myrmecia nigrocincta workers and evolutionary transformations in Formicidae (Hymenoptera). Arthropod Systematics and Phylogeny 77: 1–19 doi: https://doi.org/10.26049/ASP77-1-2019-01
Londe S, Monnin T, Cornette R, Debat V, Fisher B, Molet M (2015) Phenotypic plasticity and modularity allow for the production of novel mosaic phenotypes in ants. EvoDevo 6:36. https://doi.org/10.1186/s13227-015-0031-5
Molet M, Wheeler DE, Peeters C (2012) Evolution of novel mosaic in ants: modularity, phenotypic plasticity, and colonial buffering. Am Nat 180(3):328–341. https://doi.org/10.1086/667368
Molet M, Maicher V, Peeters C (2014) Bigger helpers in the ant Cataglyphis bombicina: increased worker polymorphism or novel soldier caste? PLoS One 9(1):e84929. https://doi.org/10.1371/journal.pone.0084929
Morello J, Matteucci S, Rodríguez A (2012) Ecorregiones y complejos ecosistémicos argentinos. 1° edición. Orientación Gráfica Editora, Buenos Aires, p 752
Nijhout HF, Emlen DJ (1998) Competition among body parts in the developmental and evolution of insect morphology. Proc Natl Acad Sci U S A 95:3685–3689. https://doi.org/10.1073/pnas.95.7.3685
Paul J (2001) Mandible movement in ants. Comp Biochem Physiol A Mol Integr Physiol 131(1):7–20. https://doi.org/10.1016/S1095-6433(01)00458-5
Paul J, Gronenberg W (1999) Optimizing force and velocity: mandible muscle fibre attachments in ants. J Exp Biol 202:797–808
Peeters C, Molet M, Lin C, Billen J (2017a) Evolution of cheaper workers in ants: a comparative study of exoskeleton thickness. Biol J Linn Soc 121(3):556–563. https://doi.org/10.1093/biolinnean/blx011
Peeters C, Ito F, Wiwatwitaya D, Keller R, Hashim R, Molet M (2017b) Striking polymorphism among infertile helpers in the arboreal ant Gesomyrmex. Asian Myrmecology 9: e009015. https://doi.org/10.20362/am.009015
Peeters C, Keller R, Khalife A, Fischer G, Katzke J, Blanke A, Economo EP (2020) The loss of flight in ant workers enabled an evolutionary redesign of the thorax for ground labour. Front Zool 17:33. https://doi.org/10.1186/s12983-020-00375-9
Powell S, Franks NR (2006) Ecology and the evolution of worker morphological diversity: a comparative analysis with Eciton army ants. Funct Ecol 20(6):1105–1114. https://doi.org/10.1111/j.1365-2435.2006.01184.x
Rajakumar R, San Mauro D, Dijkstra M, Huang M, Wheeler D, Hiou-Tim F, Khila A, Cournoyea M, Abouhief E (2012) Ancestral developmental potential facilitates parallel evolution in ants. Science 335:79–82. https://doi.org/10.1126/science.1211451
Rasband W (1997) ImageJ 1.52c. U.S. National Institutes of Health, Bethesda, Maryland, USA. http://imagej.nih.gov/ij/, 1997–2018
Rohlf FJ (2010a) tpsDig-Thin Plate Spline Digitizer, Version 2.16. Department of Ecology and Evolution, State University of New York at Stony Brook, New York, USA. https://life.bio.sunysbe.edu/morph
Rohlf FJ (2010b) tpsUtil-Thin Plate Spline Utility, Version 1.46. Department of Ecology and Evolution, State University of New York at Stony Brook, New York, USA. https://life.bio.sunysbe.edu/morph
Sameshima S, Miura T, Matsumoto T (2004) Wing disc developmental during caste differentiation in the ant Pheidole megacephala (Hymenoptera: Formicidae). Evolution and Developmental 6(5):336–341. https://doi.org/10.1111/j.1525-142X.2004.04041.x
Schöning C, Kinuthia W, Franks N (2005) Evolution of allometries in the worker caste of Dorylus army ants. Oikos 110(2):231–240. https://doi.org/10.1111/j.0030-1299.2005.13672.x
Shbailat SJ, Khila A, Abouheif E (2010) Correlations between spatiotemporal changes in gene expression and apoptosis underlie wing polyphenism in the ant Pheidole morrisi. Evolution and Developmental 12(6):580–591. https://doi.org/10.1111/j.1525-142X.2010.00443.x
Simpson GG (1953) The Baldwin effect. Evolution 7(2):110–117. https://doi.org/10.2307/2405746
Trible W, Kronauer DJ (2017) Caste developmental and evolution in ants: it’s all about size. J Exp Biol 220:53–62. https://doi.org/10.1242/jeb.145292
Wheeler DE (1991) The developmental basis of worker caste polymorphism in ants. Am Nat 138(5):1218–1238
Wills BD, Powell S, Rivera MD, Suarez AV (2018) Correlates and consequences of worker polymorphism in ants. Annu Rev Entomol 63:575–598. https://doi.org/10.1146/annurev-ento-020117-043357
Wilson EO (1953) The origin and evolution of polymorphism in ants. Q Rev Biol 28(2):136–156. https://doi.org/10.1086/399512
Wilson EO (1976) A social ethogram of the Neotropical arboreal ant Zacryptocerus varians (Fr. Smith). Anim Behav 24:354–363. https://doi.org/10.1016/S0003-3472(76)80043-7
Zelditch ML, Swiderdki DL, Sheets HD (2012) Geometric morphometrics for biologists: a primer. Second edition. Academic Press, p 477
Acknowledgments
We thank Dra. Florencia Vera Candioti and Dr. Hugo Benítez for their suggestion in the analysis of data. We also wish to thank our reviewers for their valuable feedback.
Authors’ Contribution Statement
A Galbán conceived and design of the study; performed the figures and measurements, collection and analysis of data; drafted the first draft; and corrected the manuscript. F Cuezzo participated in the original idea, conceptualization, and design of the study and corrected the manuscript. J Torréns took the photographs, performed the figures, and corrected the manuscript. All authors read and approved the final manuscript.
Funding
This work was made possible by funds provided by Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET), Agencia Nacional de Promoción Científica y Tecnológica, PICT 2013-2324, CIUNT: G638/2- PIUNT 2918 (SCAIT) and Instituto Superior de Entomología “Dr. Abraham Willink” (INSUE).
Author information
Authors and Affiliations
Corresponding author
Additional information
Edited by Heraldo Vasconcelos
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Galbán, A., Cuezzo, F. & Torréns, J. The Pronotum of Worker of Camponotus borellii Emery (Hymenoptera: Formicidae): How Can It Affect Performance of the Head, Work Division, and Development of the Worker Caste?. Neotrop Entomol 50, 78–89 (2021). https://doi.org/10.1007/s13744-020-00828-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-020-00828-0