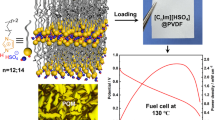
Abstract
A new decavanadate hybrid material, (C4H7N2S)4[H2V10O28]·4H2O, (2A4MT V10), was successfully grown in aqueous solution as single crystals with acidic decavanadate (V10) cluster and 2-amino-4-methyltiazolium (2A4MT) cation. It was thoroughly characterized by XRD, IR, DTA-TGA and alternating current impedance spectroscopy measurements. Crystal structure of (2A4MT V10) exhibits a typical 3D-porous supramolecular topology of diprotonated V10 clusters and water molecules featuring tunnel-voids filled with 2A4MT cations mainly connected through O···H/H···O, H bonding with considerable contributions of 65.9% as indicated by Hirshfeld surface analysis. The conductivity study shows that the hybrid crystal possesses significant conductivities above 10−4 S cm−1 in the temperature range of 130–180 °C to rich 9 × 10−4 S cm−1 at 423 K (150 °C), which can be considered as promising proton conductor for fuel cell applications.
















Similar content being viewed by others
Data availability
All relevant data generated or analyzed during this study are included in this published article and its supplementary information files.
References
K.-D. Kreuer, S.J. Paddison, E. Spohr, M. Schuster, Transport in proton conductors for fuel-cell applications: simulations, elementary reactions, and phenomenology. Chem. Rev. 104, 4637 (2004). https://doi.org/10.1021/cr020715f
B.C.H. Steele, A. Heinzel, Materials for fuel-cell technologies. Mater. Renew. Sustain. Energy (2010). https://doi.org/10.1142/9789814317665_0031
G. Alberti, M. Casciola, Solid state protonic conductors, present main applications and future prospects. Solid State Ion. 145, 3 (2001). https://doi.org/10.1016/S0167-2738(01)00911-0
N. Ogiwara, T. Iwano, T. Ito, S. Uchida, Proton conduction in ionic crystals based on polyoxometalates. Coord. Chem. Rev. 462, 214524 (2022). https://doi.org/10.1016/j.ccr.2022.214524
G. Wang, J. Li, L. Zhai, X. Li, H. He, H. Guo, H. Li, C. Zhao, L. Wu, H. Li, Polyoxometalate-polymer nanocomposites with multiplex proton transport channels for high-performance proton exchange membranes. Compos. Sci. Technol. 232, 109842 (2023). https://doi.org/10.1016/j.compscitech.2022.109842
D. Bose, A. Mukherjee, G. Mitra, Energy recovery prospects of fuel cell technologies: sustainability and bioremediation. Aust. J. Mech. Eng. 20, 736 (2022). https://doi.org/10.1080/14484846.2020.1747152
L. Zhang, T. Cui, X. Cao, C. Zhao, Q. Chen, L. Wu, H. Li, Polyoxometalate-polymer hybrid materials as proton exchange membranes for fuel cell applications. Angew. Chem. Int. Ed. 56, 9013 (2017). https://doi.org/10.3390/molecules24193425
B.C.H. Steele, A. Heinzel, Materials for fuel-cell technologies. Nature 414, 345 (2001). https://doi.org/10.1038/35104620
A. Martinelli, J.M.O. Mato, M.N. Garaga, K. Elamin, S.M.H. Rahman, J.W. Zwanziger, U.W. Zwanziger, L.M. Varela, A new solid-state proton conductor: the salt hydrate based on imidazolium and 12-tungstophosphate. J. Am. Chem. Soc. 143, 13895 (2021). https://doi.org/10.1021/jacs.1c06656
X.Y. Qian, X. Tong, Q.Y. Wu, Z.Q. He, F.H. Cao, W.F. Yan, Synthesis and electrochemical properties of substituted heteropoly acid with Dawson structure H7[In(H2O)P2W17O61]·23H2O. Dalton Trans. 41, 9897 (2012). https://doi.org/10.1007/s11434-011-4445-8
P.G. Romero, J.A. Asensio, S. Borros, Hybrid proton conducting membranes for polymer electrolyte fuel cells phosphomolybdic acid doped poly(2,5-benzimidazole)-(ABPBIH3PMo12O40). Electrochim. Acta 50, 4715 (2005). https://doi.org/10.1016/j.electacta.2005.02.029
M. Daraie, M. Mirzaei, M. Bazargan, V.S. Amiri, B.A. Sanati, M.M. Heravi, Lanthanoid-containing polyoxometalate nanocatalysts in the synthesis of bioactive isatin-based compounds. Sci. Rep. 12(1), 12004 (2022). https://doi.org/10.1038/s41598-022-16384-z
Z.Y. Ghasemi, M.M. Heravi, M. Malmir, G. Jahani, M.B. Bisafar, M. Mirzaei, Fabrication of heterogeneous-based lacunary polyoxometalates as efficient catalysts for the multicomponent and clean synthesis of pyrazolopyranopyrimidines. Inorg. Chem. Commun. 140, 109456 (2022). https://doi.org/10.1016/j.inoche.2022.109456
M.M. Heravi, T. Momeni, M. Mirzaei, V. Zadsirjan, M. Tahmasebi, An amino acid@isopolyoxometalate nanoparticles catalyst containing aspartic acid and octamolybdate for the synthesis of functionalized spirochromenes. Inorg. Nano-Met. Chem. 51, 896 (2021). https://doi.org/10.1080/24701556.2020.1813172
M. Ghanbarian, S.Y.S. Beheshtiha, M.M. Heravi, M. Mirzaei, V. Zadsirjan, N. Lotfian, A nano-sized Nd–Ag@polyoxometalate catalyst for catalyzing the multicomponent Hantzsch and Biginelli reactions. J. Clust. Sci. 31(6), 1295 (2020). https://doi.org/10.1007/s10876-019-01739-w
M.M. Heravi, T. Hosseinnejad, M. Tamimi, V. Zadsirjana, M. Mirzaei, 12-Tungstoboric acid (H5BW12O40) as an efficient Lewis acid catalyst for the synthesis of chromenopyrimidine-2,5-diones and thioxochromenopyrimidin-5-ones: joint experimental and computational study. J. Mol. Struct. 1205, 127598 (2020)
S.H. Baghan, M. Mirzaei, H.E. Hosseini, V. Zadsirjan, M.M. Heravi, J.T. Mague, An inorganic–organic hybrid material based on a Keggin-type polyoxometalate@Dysprosium as an effective and green catalyst in the synthesis of 2-amino-4H-chromenes via multicomponent reactions. Appl. Organomet. Chem. 34(9), e5793 (2020). https://doi.org/10.1002/aoc.5793
N. Lotfian, M.M. Heravi, M. Mirzaei, M. Daraie, Investigation of the uncommon basic properties of [Ln(W5O18)2]9– (Ln = La, Ce, Nd, Gd, Tb) by changing central lanthanoids in the syntheses of pyrazolopyranopyrimidines. J. Mol. Struct 1199, 126953 (2020). https://doi.org/10.1016/j.molstruc.2019.126953
N. Ogiwara, M. Tomoda, S. Miyazaki, Z. Weng, H. Takatsu, H. Kageyama, T. Misawa, T. Ito, S. Uchida, Integrating molecular design and crystal engineering approaches in non-humidified intermediate-temperature proton conductors based on a Dawson-type polyoxometalate and poly(ethylene glycol) derivatives. Nanoscale 13, 8049 (2021). https://doi.org/10.1039/D1NR01220G
S. Otobe, Y. Kiyota, S. Magira, T. Misawa, K. Fujio, H. Naruke, S. Uchida, T. Ito, Conductive inorganic-organic hybrid layered crystals composed of Keggin-type polyoxotungstates and a heterocyclic surfactant. Eur. Inorg. Chem. 3, 442 (2019). https://doi.org/10.1002/ejic.201800535
M. Taira, H. Sato, K. Fukumoto, T. Misawa, H. Naruke, T. Ito, Polyoxovanadate-surfactant hybrid layered crystals toward anhydrous proton conductors. J. Mol. Struct. 1226, 129355 (2021). https://doi.org/10.1016/j.molstruc.2020.129355
M. Yamada, I. Honma, Heteropolyacid-encapsulated self-assembled materials for anhydrous proton-conducting electrolytes. J. Phys. Chem. B 110, 20486 (2006). https://doi.org/10.1021/jp063488k
T. Ito, M. Taira, K. Fukumoto, K. Yamamoto, H. Naruke, K. Tomita, K. Bull, Polyoxovanadate-surfactant hybrid layered crystal containing one-dimensional hydrogen-bonded cluster chain. Chem. Soc. Jpn. 85, 1222 (2012). https://doi.org/10.1246/bcsj.20120157
G.M. Sheldrick, Acta Cryst. A64, 112 (2008). https://doi.org/10.1107/S0108767307043930
L.J. Farrugia, WINGX (University of Glasgow, Glasgow, 2005)
K. Brandenburg, H. Putz, DIAMOND (Crystal Impact GbR, Bonn, 1999)
D. Kobashi, S. Kohara, J. Yamakawa, A. Kawahara, Structure d’un diphosphate synthétique de cobalt: Co2P2O7. Acta Crystallogr. 53, 1523 (1997). https://doi.org/10.1107/S0108270197006689
M.T. Pope, Heteropoly and IsopolyOxometalates, vol. 18 (Springer, New York, 1983)
I.D. Brown, M. O’Keefe, A. Navrotsky (eds.), Structure and Bonding in Crystals (Academic Press, New York, 2012). https://doi.org/10.1007/BF00646987
M. Louati, R. Ksiksi, L. Jouffret, M.F. Zid, Preparation, crystal structure and spectroscopy characterization of vanadium (V) complex, 2-amino-4-picolinium,2-methylimidazolium decavanadate hydrate (C4N2H7)4(C6N2H10)V10O28·2H2O. CSTA 8, 1 (2019). https://doi.org/10.4236/csta.2019.81001
V. Arjunan, S. Sakiladevi, T. Rani, C.V. Mythili, S. Mohan, FTIR, FT-Raman, FT-NMR, UV–visible and quantum chemical investigations of 2-amino-4-methylbenzothiazole. Spectrochim-Part A 88, 220 (2012). https://doi.org/10.1016/j.saa.2011.12.037
S. Toumi, N.R. Ramond, S. Akriche, Decavanadate cage-like cluster templated by organic counter cation: synthesis, characterization and its antimicrobial effect against gram positive E. Feacium. J. Clust. Sci. 26, 1821 (2015). https://doi.org/10.1007/s10876-015-0881-y
J.J. McKinnon, M.A. Spackman, A.S. Mitchell, Novel tools for visualizing and exploring intermolecular interactions in molecular crystals. Science 60, 627 (2004). https://doi.org/10.1107/S0108768104020300
S.K. Wolff, D.J. Grimwood, J.J. McKinnon, M.J. Turner, D. Jayatilaka, M.A. Spackman, Crystal Explorer (University of Western Australia, Perth, 2013). https://doi.org/10.4236/jtts.2013.32A005
M. Taira, H. Sato, K. Fukumoto, T. Misawa, H. Naruke, T. Ito, J. Mol. Struct. 1226, 129355 (2021)
S.Ü. Çelik, A. Bozkurt, S.S. Hosseini, Alternatives toward proton conductive anhydrous membranes for fuel cells: heterocyclic protogenic solvents comprising polymer electrolytes. Prog. Polym. Sci. 37, 1265 (2012). https://doi.org/10.1016/j.progpolymsci.2011.11.006
H. Ma, B. Liu, B. Li, L. Zhang, Y.G. Li, H.Q. Tan, H.Y. Zang, G. Zhu, Cationic covalent organic frameworks: a simple platform of anionic exchange for porosity tuning and proton conduction. J. Am. Chem. Soc. 138, 5897 (2016). https://doi.org/10.1021/jacs.5b13490
A. Maalaoui, O. Pérez, M. Rzaigui, S.T. Akriche, Enhancement with Hirshfeld surface analysis of structural, electrical, dielectric and luminescent performance of two bioactive V-substituted polytungstates. J. Alloys Comp 95, 1061 (2016). https://doi.org/10.1016/j.jallcom.2016.10.231
A. Boukhachem, A. Ziouche, M. Ben Amor, O. Kamoun, M. Zergoug, H. Maghraoui-Meherzi, A. Yumak, K. Boubaker, M. Amlouk, Physical investigations on perovskite LaMnO3-δ sprayed thin films for spintronic applications. Mater. Res. Bull. 74, 202 (2016). https://doi.org/10.1016/j.materresbull.2015.10.003
A. Mater, A. Othmani, A. Boukhachem, A. Madani, Cathode performance study of La0.6Sr0.4Co0.8Fe0.2O3-δ with various electrolyte-doped ceria Ce0.8Sm0.17Ln0.03O1.9 for IT-solid oxide fuel cell. J. Electron. Mater. 49, 4123 (2020). https://doi.org/10.1007/s11664-020-08167-x
A.K. Jonscher, The electrical properties of silicon monoxide. Nature 29, 7 (1975). https://doi.org/10.1016/0040-6090(75)90211-4
M. Ben Amor, A. Boukhachem, K. Boubaker, M. Amlouk, Structural, optical and electrical studies on Mg-doped NiO thin films for sensitivity applications. Mater. Sci. Semicond. Process. 27, 994 (2014). https://doi.org/10.1016/j.mssp.2014.08.008
M. Abbassi, R. Ternane, I. Sobrados, A. Madani, M. Trabelsi-Ayadi, Ionic conductivity of apatite-type solid electrolyte ceramics Ca2−xBaxLa4Bi4(SiO4)6O2 (0≤x≤2). J. Sanz. Ceram. Int. 39, 9215 (2013). https://doi.org/10.1016/j.ceramint.2013.05.026
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by TOTA, AM, MR, STA, AB and JA. The first draft of the manuscript was written by TOTA and AM, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Amar, T.O.T., Maalaoui, A., Boukhachem, A. et al. Hybrid proton-containing decavanadate-organic crystal: synthesis, structure and enhancement of proton-conducting performance. J IRAN CHEM SOC 20, 1345–1356 (2023). https://doi.org/10.1007/s13738-023-02759-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-023-02759-0