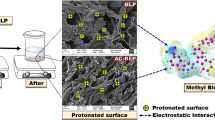
Abstract
In this work, the effect of acid activation conditions on the textural, structural and adsorptive characteristics of phosphoric acid-activated carbon (AC) prepared from pentaclethra macrophylla pods (PMAC) was examined for the removal of hexavalent chromium from aqueous solution. The precursor and prepared AC were characterized by TGA, FTIR, SEM–EDX, and iodine and methylene blue indices. The TGA curves depicted the presence of hemicellulose, cellulose and lignin confirming the lignocellulosic structure of the precursor. The iodine index (908.60, 911.14 and 955.31 mg/g) and methylene blue index (49.78, 106.36 and 204.12 mg/g) increased with increasing acid:solid ratios (1:1, 2:1 and 3:1) for PMAC1, PMAC2 and PMAC3, denoting increase in microporosity and mesoporosity, respectively. The experimental data were best explained by the Elovich kinetic model and Sips isotherm with maximum adsorption capacities of 438.71, 265.911 and 406.593 mg/g for PMAC1, PMAC2 and PMAC3, respectively. The calculated mean adsorption energies were less than 8 kJ/mol, indicating that the adsorption of Cr(VI) was a physisorption process. Activation ratio of 1:1 had the fastest adsorption rate and largest adsorption capacity for Cr(VI) and is recommended as the optimal synthesis condition.









Similar content being viewed by others
References
G. Agbor Tabi, L. Ngouateu Rene Blaise, K. Daouda et al., Non-linear modelling of the adsorption of Indigo Carmine dye from wastewater onto characterized activated carbon/volcanic ash composite. Arab. J. Chem. 15, 103515 (2022). https://doi.org/10.1016/j.arabjc.2021.103515
E.O. Akperov, O.H. Akperov, The wastage of the cotton stalks (Gossypium hirsutum L.) as low-cost adsorbent for removal of the Basic Green 5 dye from aqueous solutions. Appl. Water Sci. 9, 1–9 (2019). https://doi.org/10.1007/s13201-019-1071-0
M.A. Al-Ghouti, D,A.Da’ana, Guidelines for the use and interpretation of adsorption isotherm models: a review. J. Hazard. Mater. 393, 122383 (2020)
N. Ayawei, A.N. Ebelegi, D. Wankasi, Modelling and interpretation of adsorption isotherms. J. Chem. 2017, 1 (2017)
Y. Birhanu, S. Leta, G. Adam, Removal of chromium from synthetic wastewater by adsorption onto Ethiopian low-cost Odaracha adsorbent. Appl. Water Sci. 10, 1–11 (2020). https://doi.org/10.1007/s13201-020-01310-3
Y. Deng, M. Wang, T. Tian et al., The effect of hexavalent chromium on the incidence and mortality of human cancers: a meta-analysis based on published epidemiological cohort studies. Front. Oncol. (2019). https://doi.org/10.3389/fonc.2019.00024
M.B. Desta, Batch Sorption Experiments: Langmuir and Freundlich Isotherm Studies for the Adsorption of Textile Metal Ions onto Teff Straw ( Eragrostis tef ) Agricultural Waste (2013)
T. Dula, K. Siraj, S.A. Kitte, Adsorption of hexavalent chromium from aqueous solution using chemically activated carbon prepared from locally available waste of bamboo (Oxytenanthera abyssinica). ISRN Environ. Chem. 2014, 1–9 (2014). https://doi.org/10.1155/2014/438245
T. Eliku, S. Leta, Spatial and seasonal variation in physicochemical parameters and heavy metals in Awash River, Ethiopia. Appl. Water Sci. (2018). https://doi.org/10.1007/s13201-018-0803-x
X. Guo, A. Liu, J. Lu et al., Adsorption mechanism of hexavalent chromium on biochar: Kinetic, thermodynamic, and characterization studies. ACS Omega 5, 27323–27331 (2020). https://doi.org/10.1021/acsomega.0c03652
D.T. Hermann, S. Tome, V.O. Shikuku et al., Enhanced Performance of Hydrogen Peroxide Modified Pozzolan-Based Geopolymer for Abatement of Methylene Blue from Aqueous Medium (2021)
T.L. Hill, Statistical mechanics of multimolecular adsorption. I. J. Chem. Phys. 14, 263–267 (1946). https://doi.org/10.1063/1.1724129
Y.S. Ho, Review of second-order models for adsorption systems. J. Hazard Mater. 136, 681–689 (2006). https://doi.org/10.1016/j.jhazmat.2005.12.043
A.U. Itodo, H.U. Itodo, Sorption energies estimation using Dubinin Radushkevich and Temkin adsorption isotherms. Life Sci. 7, 31–39 (2010)
S.A. Katz, H. Salem, The toxicology of chromium with respect to its chemical speciation: a review. J. Appl. Toxicol. 13, 217–224 (1993). https://doi.org/10.1002/jat.2550130314
M. Kaya, Ö. Azahin, C. Saka, Preparation and TG/DTG, FT-IR, SEM, BET surface area, iodine number and methylene blue number analysis of activated carbon from pistachio shells by chemical activation. Int. J. Chem. React. Eng. 16, 1–13 (2018). https://doi.org/10.1515/ijcre-2017-0060
Y. Keren, M. Borisover, N. Bukhanovsky, Sorption interactions of organic compounds with soils affected by agricultural olive mill wastewater. Chemosphere 138, 462–468 (2015). https://doi.org/10.1016/j.chemosphere.2015.06.085
B.D. Kerger, D.J. Paustenbach, G.E. Corbett, B.L. Finley, Absorption and elimination of trivalent and hexavalent chromium in humans following ingestion of a bolus dose in drinking water. Toxicol. Appl. Pharmacol. 141, 145–158 (1996). https://doi.org/10.1016/s0041-008x(96)80020-2
U. Khalil, M.B. Shakoor, S. Ali et al., Selective removal of hexavalent chromium from wastewater by rice husk: kinetic, isotherm and spectroscopic investigation. Water (Switzerland) (2021). https://doi.org/10.3390/w13030263
M.K. Kim, K.D. Zoh, 320 treatment processes. 2. Sour. Transp. Micropollut. Environ. 21, 319–332 (2016)
S. Kurwadkar, Occurrence and distribution of organic and inorganic pollutants in groundwater. Water Environ. Res. 91, 1001–1008 (2019). https://doi.org/10.1002/wer.1166
I. Langmuir, The adsorption of gases on plane surfaces of glass, mica and platinium. J. Am. Chem. Soc. 40, 1361–1403 (1918). https://doi.org/10.1021/ja01269a066
L. Largitte, R. Pasquier, Chemical engineering research and design a review of the kinetics adsorption models and their application to the adsorption of lead by an activated carbon. Chem. Eng. Res. Des. 109, 495–504 (2016). https://doi.org/10.1016/j.cherd.2016.02.006
A. Linos, A. Petralias, C.A. Christophi et al., Oral ingestion of hexavalent chromium through drinking water and cancer mortality in an industrial area of Greece - an ecological study. Environ. Heal. A Glob. Access. Sci. Sour. 10, 1–8 (2011). https://doi.org/10.1186/1476-069X-10-50
L. Liu, X. Liu, D. Wang et al., Removal and reduction of Cr (VI) in simulated wastewater using magnetic biochar prepared by co-pyrolysis of nano-zero-valent iron and sewage sludge. J. Clean. Prod. 257, 120562 (2020). https://doi.org/10.1016/j.jclepro.2020.120562
E.I. Madukasi, K. Oso, C.C. Igwe, Waste- to -Energy Resources: African Oil Bean Seed Husk Combustion. 3:1–5 (2016)
U. Mahapatra, A. Chatterjee, C. Das, A. Kumar, Environmental Technology & Innovation Adsorptive removal of hexavalent chromium and methylene blue from simulated solution by activated carbon synthesized from natural rubber industry biosludge. Environ. Technol. Innov. 22, 101427 (2021). https://doi.org/10.1016/j.eti.2021.101427
T. Mahmood, M.T. Saddique, A. Naeem et al., Comparison of different methods for the point of zero charge determination of NiO. Ind. Eng. Chem. Res. 50, 10017–10023 (2011). https://doi.org/10.1021/ie200271d
T. Masinga, M. Moyo, V.E. Pakade, Removal of hexavalent chromium by polyethyleneimine impregnated activated carbon: intra-particle diffusion, kinetics and isotherms. J. Mater. Res. Technol. 18, 1333–1344 (2022). https://doi.org/10.1016/j.jmrt.2022.03.062
J.B. Mbouombouo, L.C. Nintedem Magapgie, G.P. Tchieta et al., Adsorption of chromium (VI ) onto activated carbons prepared from Raphia farinifera fruit kernels by chemical activation with phosphoric acid (H3PO4). Int. J. Sci. Res. Methodol. 18, 81–109 (2021)
M. Molina-Sabio, F. Rodríguez-Reinoso, Role of chemical activation in the development of carbon porosity. Colloids Surfaces A: Physicochem. Eng. Aspects. 241, 15–25 (2004)
M. Mouamfon, N. Guedje, I. Pepainyiene et al., Pentaclethra macrophylla Benth. dans la forêt communautaire de Payo (Est- Cameroun): inventaire, productivité et commercialisation. Int. J. Biol. Chem. Sci. 9, 200 (2015). https://doi.org/10.4314/ijbcs.v9i1.18
J. Ndjama, G. Mafany, B.E.B. Yvette Clarisse Mfopou Mewouo et al., Evaluation of surface water contamination using heavy metal pollution indices in the Mgoua Watershed Southwestern Cameroon. Int. J. Curr. Microbiol. Appl. Sci. 10, 142–156 (2021). https://doi.org/10.20546/ijcmas.2021.1011.018
P. Ninduangdee, V.I. Kuprianov, E.Y. Cha et al., Thermogravimetric studies of oil palm empty fruit bunch and palm kernel shell: TG/DTG analysis and modeling. Energy Procedia 79, 453 (2015)
C.L. Nintedem Magapgie, T. Geordamie Chimi, R.N. Tchoumi, P.G. Tchieta, J.B. Mbouombouo et al., Adsorption of chromium (VI) and orange methyl onto activated carbon obtained from the cores of Canarium ovatum: influence of functionalization. Int. J. Sci. Res. Methodol. 18, 1–23 (2021)
J.B. Njewa, E. Vunain, T. Biswick, Synthesis and characterization of activated carbons prepared from agro-wastes by chemical activation. J. Chem. (2022). https://doi.org/10.1155/2022/9975444
S.D. Noa Tang, E.B.A. Zacharie, T.K. Brice et al., Heavy metal contamination and ecological risk assessment of overlying water and sediments of Nkozoa Lake (Southern Cameroon). Annu. Res. Rev. Biol. (2021). https://doi.org/10.9734/arrb/2021/v36i430366
J.N. Nsami, J.K. Mbadcam, The Adsorption Efficiency of Chemically Prepared Activated Carbon from Cola Nut Shells by ZnCl 2 on Methylene Blue (2013)
C.A. Nunes, M.C. Guerreiro, Estimation of surface area and pore volume of activated carbons by methylene blue and iodine numbers. Quim Nova 34, 472–476 (2011). https://doi.org/10.1590/S0100-40422011000300020
O.D. Onukwuli, C.C. Okoye, I.C. Nwokedi, Removal of heavy metal Pb(II) ions from aqueous solution using Pentaclethra macrophylla and Tetracarpidium conophorum seed shells based activated carbons: equilibrium. Kinet Thermodyn Stud. 16, 1–20 (2016). https://doi.org/10.9734/BJAST/2016/27255
T.D.P. Protásio, M.G. Junior, S. Mirmehdi et al., Combustão da biomassa E do carvão vegetal da casca do coco babaçu. Cerne 23, 1–10 (2017). https://doi.org/10.1590/01047760201723012202
N.A.A. Qasem, R.H. Mohammed, D.U. Lawal, Removal of heavy metal ions from wastewater: a comprehensive and critical review. NPJ Clean Water (2021). https://doi.org/10.1038/s41545-021-00127-0
P.A. Raut, A. Chahande, Y. Moharkar, Various techniques for the removal of chromium and lead from waste water: review. Int. J. Emerg. Trends Eng. Basic Sci. 2, 64–67 (2015)
R. Saadi, Z. Saadi, R. Fazaeli, N.E. Fard, Monolayer and multilayer adsorption isotherm models for sorption from aqueous media. 32, 1–13 (2015). https://doi.org/10.1007/s11814-015-0053-7
M.M. Sabzehmeidani, S. Mahnaee, M. Ghaedi et al., Carbon based materials: a review of adsorbents for inorganic and organic compounds. Mater. Adv. 2, 598–627 (2021). https://doi.org/10.1039/d0ma00087f
R. Singh, N. Gautam, A. Mishra, R. Gupta, Heavy metals and living systems: an overview. Indian J. Pharmacol. 43, 246–253 (2011). https://doi.org/10.4103/0253-7613.81505
R. Sips, On the structure of a catalyst surface. J. Chem. Phys. 16, 490–495 (1948). https://doi.org/10.1063/1.1746922
S. Roginsky, Y.B. Zeldovich, Acta Phys. Chem. USSR (1934)
M. Temkin, V. Pyzhev, Kinetics of Ammonia Synthesis on Promoted Iron Catalysts. Acta Physicochimica U.R.S.S., 12, 327–356 (1940)
T.K. Trinh, T. Tsubota, S. Takahashi et al., Carbonization and activation of fern Dicranopteris linearis and electrochemical properties for electric double layer capacitor electrode. Sci. Rep. (2020). https://doi.org/10.1038/s41598-020-77099-7
V. Velma, S.S. Vutukuru, P.B. Tchounwou, Ecotoxicology of hexavalent chromium in freshwater fish: a critical review. Rev. Environ. Health 24, 129–145 (2009)
E. Vunain, J.B. Njewa, T.T. Biswick, A.K. Ipadeola, Adsorption of chromium ions from tannery effluents onto activated carbon prepared from rice husk and potato peel by H3PO4 activation. Appl. Water Sci. 11, 1–14 (2021). https://doi.org/10.1007/s13201-021-01477-3
W.J. Weber, J.C. Morris, Kinetics of adsorption on carbon from solutions. J. Sanit. Eng. Div. 89, 31–39 (1963)
WHO, Guidelines for drinking-water quality: fourth edition incorporating the first addendum. Geneva: World Health Organization; Licence: CC BY-NC-SA 3.0 IGO (2017)
H. Yang, Z. Li, P. Fu, G. Zhang, Cr(VI) removal from a synthetic solution using a novel carbonaceous material prepared from oily sludge of tank bottom. Environ. Pollut. (2019). https://doi.org/10.1016/j.envpol.2019.03.065
A.S. Yusuff, Adsorption of hexavalent chromium from aqueous solution by Leucaena leucocephala seed pod activated carbon: equilibrium, kinetic and thermodynamic studies. Arab J Basic Appl Sci 26, 89–102 (2019). https://doi.org/10.1080/25765299.2019.1567656
L. Zhao, W. Zheng, O. Mašek et al., Roles of phosphoric acid in biochar formation: synchronously improving carbon retention and sorption capacity. J Environ Qual 46, 393–401 (2017). https://doi.org/10.2134/jeq2016.09.0344
M. Zięzio, B. Charmas, K. Jedynak et al., Preparation and characterization of activated carbons obtained from the waste materials impregnated with phosphoric acid (V). Appl. Nanosci. 10, 4703–4716 (2020). https://doi.org/10.1007/s13204-020-01419-6
Author information
Authors and Affiliations
Contributions
Tchatchouang and Boresse collected the plant material and carried out the experimental work, Tchatchouang drafted the manuscript, Dzoujo assisted in data analysis, Tome and Victor realized and contributed in interpreting the structural and adsorption data, Achille Nouga provided access to his laboratory for batch adsorption experiments, Pierre Gerard and François Eya’ane designed and supervised the project. All authors read an approved the final manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chimi, T., Hannah, B.U., Lincold, N.M. et al. Preparation, characterization and application of H3PO4-activated carbon from Pentaclethra macrophylla pods for the removal of Cr(VI) in aqueous medium. J IRAN CHEM SOC 20, 399–413 (2023). https://doi.org/10.1007/s13738-022-02675-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02675-9