Abstract
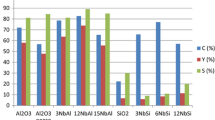
The modification of UiO-66 with urea and melamine were carried out using ZrCl4, benzene dicarboxylic acid and designated as U-B-Zr and M-B-Zr, followed by immobilization of vanadyl sulphate on U-B-Zr and M-B-Zr as U-B-Zr-VO-and M-B-Zr-VO, respectively. The prepared compounds as catalysts were characterized by XRD, BET, XPS, TGA, SEM, FTIR and CHN techniques. It was found that the prepared catalysts show moderate to high activity towards the epoxidation of some allyl alcohols such as trans-2-hexene-1-ol and 1-octen-3-ol and geraniol with 80%–100% conversions and 100% selectivity towards the formation of the corresponding epoxides. The prepared catalysts were also found to be reusable up four catalytic cycles without any appreciable loss in activity.












Similar content being viewed by others
References
G. Ferey, Chem. Soc. Rev. 37, 191–214 (2008)
J.-R. Li, R.J. Kuppler, H.-C. Zhou, Chem. Soc. Rev. 38, 1477–1504 (2009)
Y. Bai, Y. Dou, L.-H. Xie, W. Rutledge, J.-R. Li, H.-C. Zhou, Chem. Soc. Rev. 45, 2327–2367 (2016)
A. Ma, Z. Luo, C. Gu, B. Li, J. Liu, Inorg. Chem. Communi. 77, 68–71 (2017)
P. Kumar, A. Pournara, K.-H. Kim, V. Bansal, S. Rapti, M.J. Manos, Prog. Mater. Sci. 86, 25–74 (2017)
A. Kirchon, L. Feng, H.F. Drake, E.A. Josepha, H.-C. Zhou, Chem. Soc. Rev. 47, 8611–8638 (2018)
J.H. Cavka, S. Jakobsen, U. Olsbye, N. Guillou, C. Lamberti, S. Bordiga, K.P. Lillerud, J. Am. Chem. Soc. 130, 13850–13851 (2008)
A.M. Ebrahim, T.J. Bandosz, Micropor. Mesopor. Mater. 188, 149–162 (2014)
H.G.T. Nguyen, N.M. Schweitzer, C.-Y. Chang, T.L. Drake, M.C. So, P.C. Stair, O.K. Farha, J.T. Hupp, S.T. Nguyen, ACS Catal. 4, 2496–2500 (2014)
H.G.T. Nguyen, L. Mao, A.W. Peters, C.O. Audu, Z.J. Brown, O.K. Farha, J.T. Hupp, S.T. Nguyen, Catal. Sci. Tech. 5, 4444–4451 (2015)
H. Fei, J. Shin, Y.S. Meng, M. Adelhardt, J. Sutter, K. Meyer, S.M. Cohen, J. Am. Chem. Soc. 136, 4965–4973 (2014)
E. Geravand, F. Farzaneh, M. Ghiasi, J. Mol. Struct. 1198, 126940 (2019)
R. Gil-San-Millan, E. López-Maya, M. Hall, N.M. Padial, G.W. Peterson, J.B. DeCoste, L.M. Rodríguez-Albelo, J.E. Oltra, E. Barea, J.A.R. Navarro, A.C.S. Appl, Mater. Inter. 9, 23967–23973 (2017)
P. Van Der Voort, K. Leus, Y.-Y. Liu, M. Vandichel, V. Van Speybroeck, M. Waroquier, S. Biswas, New. J. Chem. 38, 1853–1867 (2014)
E. Geravand, F. Farzaneh, M. Ghiasi. J. Mol. Struct. 1253, 132248 (2022)
E. Geravand, F. Farzaneh, R. Gil-San-Millan, F.J. Carmona, J.A.R. Navarro, Inorg. Chem. 59, 16160–16167 (2020)
S. Sadeghi, M. Jafarzadeh, A. Reza Abbasi, K. Daasbjerg, New. J. Chem. 41, 12014–12027 (2017)
Y. Luan, Y. Qi, Z. Jin, X. Peng, H. Gao, G. Wang, RSC Adv. 5, 19273–19278 (2015)
J.M. Roberts, B.M. Fini, A.A. Sarjeant, O.K. Farha, J.T. Hupp, K.A. Scheidt, J. Am. Chem. Soc. 134, 3334–3337 (2012)
Z. Su, Y.-R. Miao, G. Zhang, J.T. Miller, K.S. Suslick, Chem. Sci. 8, 8004–8011 (2017)
G. Silversmit, D. Depla, H. Poelman, G.B. Marin, R. De Gryse, Sur. Sci. 600, 3512–3517 (2006)
A. Wang, Y. Zhou, Z. Wang, M. Chen, L. Sun, X. Liu, RSC Adv. 6, 3671–3679 (2016)
S. Dorbes, C. Pereira, M. Andrade, D. Barros, A. Pereira, S. Rebelo, J. Araujo, J. Pires, A. Carvalho, C. Freire, Micropor. Mesopor. Mater. 160, 67–74 (2012)
E. Zamanifar, F. Farzaneh, React. Kinet. Mech. Catal. 104, 197–209 (2011)
L. Hamidipour, F. Farzaneh, Compt. Rendus. Chem. 17, 927–933 (2014)
L.M. Slaughter, J.P. Collman, T.A. Eberspacher, J.I. Brauman, Inorg. Chem. 43, 5198–5204 (2004)
Z. Azarkamanzad, F. Farzaneh, M. Maghami, J. Simpson, M. Azarkish, Appl. Organomet. Chem. 32, 4168 (2018)
A. Rezaeifard, I. Sheikhshoaie, N. Monadi, M. Alipour, Polyhedron 29, 2703–2709 (2010)
Acknowledgements
The authors would appreciate Alzahra University for financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Geravand, E., Talatimehr, S., Ghahremani, M. et al. Investigation of catalytic activity of vanadyl sulphate immobilized on prepared UiO-66 modified with urea and melamine as allyl alcohol epoxidation catalysts. J IRAN CHEM SOC 19, 4669–4677 (2022). https://doi.org/10.1007/s13738-022-02649-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02649-x