Abstract
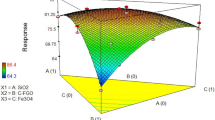
The effective adsorption of lead from wastewater has become a significant problem from both environmental and biological viewpoints. In this study, a composite of reduced graphene oxide (RGO) with zeolitic imidazolate framework-67(ZIF-67) was prepared as adsorbent to remove the Pb(II) ions from aqueous solutions. The composite was characterized by various techniques such as SEM, TEM, FTIR, XRD, BET and TGA. The important factors including sorbent weight, contact time, initial lead concentration, pH and temperature were optimized by central composite design (CCD) of response surface methodology (RSM). The maximum adsorption of 99.2% was achieved at the initial lead concentration of 87.5 ppm, adsorbent weight of 38.68 mg, contact time of 46 min and temperature of 47 °C. Different kinetic and isotherm models were evaluated to describe adsorption data. The results showed that kinetic data have better agreement with the pseudo-second-order kinetic model. From isotherm studies, the experimental results exhibited the best matching with the Langmuir isotherm. The outcomes of thermodynamic investigations confirmed that the adsorption process is endothermic and spontaneous.








Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included in this published article.
References
Q. Xu, Y. Wang, L. Jin, M. Qin, Adsorption of Cu(II), Pb(II) and Cr(VI) from aqueous solutions using black wattle tannin-immobilized nanocellulose. J. Hazard. Mater. 339, 91 (2017)
N. Wang, R.N. Jin, A.M. Omer, X.K. Ouyang, Adsorption of Pb(II) from fish sauce using carboxylated cellulose nanocrystal: Isotherm, kinetics, and thermodynamic studies. Int. Biol. Macromol. 102, 232 (2017)
M.S. Samuel, S.S. Shah, J. Bhattacharya, K. Subramaniam, N.D. Pradeep Singh, Adsorption of Pb(II) from aqueous solution using a magnetic chitosan/graphene oxide composite and its toxicity studies. Int. J. Biol. Macromol. 115, 1142 (2018)
W. Peng, H. Li, Y. Liu, S. Song, Comparison of Pb(II) adsorption onto graphene oxide prepared from natural graphites: Diagramming the Pb(II) adsorption sites. Appl. Surf. Sci. 364, 620 (2016)
H. Ren, Z. Gao, D. Wu, J. Jiang, Y. Sun, C. Luo, Efficient Pb(II) removal using sodium alginate-carboxymethyl cellulose gel beads: Preparation, characterization, and adsorption mechanism. Carbohydr. Polym. 137, 402 (2016)
X. Liang, G. Wei, J. Xiong, F. Tan, H. He, C. Qu, H. Yin, J. Zhu, R. Zhu, Z. Qin, J. Zhang, Adsorption isotherm, mechanism, and geometry of Pb(II) on magnetites substituted with transition metals. Chem. Geol. 470, 132 (2017)
L. Jiang, F. Chai, Q. Chen, Soft magnetic nanocomposite microgels by in-situ crosslinking of poly acrylic acid onto superparamagnetic magnetite nanoparticles and their applications for the removal of Pb(II) ion. Eur. Polym. J. 89, 468 (2017)
W. Tanan, S. Panpinit, S. Saengsuwan, Comparison of microwave-assisted and thermal-heated synthesis of P (HEMA-co-AM)/PVA interpenetrating polymer network (IPN) hydrogels for Pb(II) removal from aqueous solution: Characterization, adsorption and kinetic study. Eur. Poly. J. 143, 110193 (2021)
Y. Chen, J. Tang, S. Wang, L. Zhang, W. Sun, Bimetallic coordination polymer for highly selective removal of Pb(II): Activation energy, isosteric heat of adsorption and adsorption mechanism. Chem. Eng. J. 425, 131474 (2021)
Y. He, P. Wu, W. Xiao, G. Li, J. Yi, Y. He, C. Chen, P. Ding, I.Y. Duan, Efficient removal of Pb(II) from aqueous solution by a novel ion imprinted magnetic biosorbent: adsorption kinetics and mechanisms. PLoS ONE 14, e0213377 (2020)
Y. Huang, S. Li, J. Chen, X. Zhang, Y. Chen, Adsorption of Pb(II) on mesoporous activated carbons fabricated from water hyacinth using H3PO4 activation adsorptioncapacity, kinetic and isotherm studies. Appl. Surf. Sci. 293, 160 (2014)
S. Luo, X. Xu, G. Zhou, C. Liu, Y. Tang, Y. Liu, Amino siloxane oligomer-linkedgraphene oxide as an efficient adsorbent for removal of Pb(II) from waste- water. J. Hazard. Mater. 274, 145 (2014)
I.A.H. Hamza, B.S. Martincigh, J.C. Ngila, V.O. Nyamori, Adsorption studies of aqueous Pb(II) onto a sugarcane bagasse/multi-walled carbon nanotube composite. Phys. Chem. Earth. 66, 157 (2013)
Y. Ma, Z. Deng, Z. Li, Q. Lin, Y. Wu, W. Dou, Adsorption characteristics and mechanism for K2Ti4O9 whiskers removal of Pb(II), Cd(II), and Cu(II) cations in wastewater. J. Environ. Chem. Eng. 9, 106236 (2021)
R. Abazari, A.R. Mahjoub, J. Shariati, Synthesis of a nanostructured pillar MOF with high adsorption capacity towards antibiotics pollutants from aqueous solution. J. Hazard. Mater. 366, 439 (2019)
X. Wei, Y. Chai, N. Liu, S. Qiao, Y. Fu, S. Chong, ZIF67@MoO3 NSs@NF composite electrocatalysts reinforced by chemical bonds and oxygen vacancy for efficient oxygen evolution reaction and overall water-splitting. Int. J. Hydrog. Energy. 47, 9606 (2022)
Y. Huang, X. Zeng, L. Guo, J. Lan, L. Zhang, D. Cao, Heavy metal ion removal ofwastewater by zeolite-imidazolate frameworks. Sep. Pur. 194, 462 (2018)
P.Y. Lee, L.Y. Lin, Investigating energy storage ability of ZIF67-derived perovskite fluoride via tuning ammonium fluoride amounts. J. Alloys Compd. 892, 162191 (2021)
M. Jian, B. Liu, G. Zhang, R. Liu, X. Zhang, Adsorptive removal of arsenic from aqueous solution by zeolitic imidazolate framework-8 (ZIF-8) nanoparticles. Colloids Surf. A. 465, 67 (2015)
C. Wang, R. Yang, H. Wang, Synthesis of ZIF-8/Fly ash composite for adsorption of Cu2+, Zn2+ and Ni2+ from aqueous solutions. Materials 13, 214 (2020)
Y. Zhou, Y. Pan, W. Liu, L. Zhang, Removal of heavy metal ions from aqueous solutions by adsorption onto ZIF-8 nanocrystals. Chem. Lett. 44, 758 (2015)
G.K. Ramesha, A.V. Kumara, H.B. Muralidhara, S. Sampath, Graphene and graphene oxide as effective adsorbents toward anionic and cationic dyes. J. Colloid. Interface. Sci. 361, 270 (2011)
A.K. Mishra, S. Ramaprabhu, Functionalized graphene sheets for arsenic removal and desalination of sea water. Desalination 282, 39 (2011)
C. Bulin, Z. Ma, T. Guo, B. Li, Y. Zhang, B. Zhang, R. Xing, X. Ge, Magnetic graphene oxide nanocomposite: One-pot preparation, adsorption performance and mechanism for aqueous Mn(II) and Zn(II). J. Phys. Chem. Solids 156, 110130 (2021)
G. Zhao, X. Ren, X. Gao, X. Tan, J. Li, C. Chen, Y. Huang, X. Wang, Removal of Pb(II) ions from aqueous solutions on few-layered graphene oxide nanosheets. Dalton Trans. 40, 10945 (2011)
M. Zhao, A.T. Reda, D. Zhang, Reduced graphene oxide/ZIF-67 aerogel composite material for uranium adsorption in aqueous solutions. ACS Omega 5, 8012 (2020)
M. Muschi, C. Serre, Progress and challenges of graphene oxide/metal-organic composites. Coord. Chem. Rev. 387, 262 (2019)
S. Cao, T. Tang, C. Xi, Z. Chen, Fabricating magnetic GO/ZIF-8 nanocomposite for amphetamine adsorption from water: Capability and mechanism. Chem. Eng. J. 422, 130096 (2021)
X. Liu, C. Wang, Q. Wu, Z. Wang, Metal-organic framework-templated synthesis of magnetic nanoporous carbon as an efficient adsorbent for enrichment of phenylurea herbicides. Anal. Chim. Acta. 870, 67 (2015)
A.F. Gross, E. Sherman, J.J. Vajo, Aqueous room temperature synthesis of cobalt and zinc sodalite zeolitic imidazolate frameworks. Dalton Trans. 41, 5458 (2012)
H. Wang, X. Yuan, Y. Wu, H. Huang, G. Zeng, Y. Liu, X. Wang, N. Lin, Y. Qi, Adsorption characteristics and behaviors of graphene oxide for Zn(II) removal from aqueous solution. Appl. Surf. Sci. 279, 432 (2013)
S. Gholamiyan, M. Hamzehloo, A. Farrokhnia, RSM optimized adsorptive removal of erythromycin using magnetic activated carbon: Adsorption isotherm, kinetic modeling and thermodynamic studies. Sustain. Chem. Pharm. 17, 100309 (2020)
M. Kaur, S. Kumari, P. Sharma, Removal of Pb(II) from aqueous solution using nanoadsorbent of Oryza sativa husk: Isotherm, kinetic and thermodynamic studies. Biotechnol. Rep. 25, e00410 (2020)
M.D. Yahya, K.S. Obayomi, M.B. Abdulkadir, Y.A. Iyaka, A.G. Olugbenga, Characterization of cobalt ferrite-supported activated carbon for removal of chromium and lead ions from tannery wastewater via adsorption equilibrium. Water Sci. Eng. 13, 202 (2020)
Y. Priastomo, H.R. Setiawan, Y.S. Kurniawan, K. Ohto, Simultaneous removal of lead(II), chromium(III), and copper(II) heavy metal ions through an adsorption process using C-phenylcalix pyrogallolarene material. J. Environ. Chem. Eng. 8, 103971 (2020)
Y. Chen, J. Tang, S. Wang, L. Zhang, Ninhydrin-functionalized chitosan for selective removal of Pb(II) ions: Characterization and adsorption performance. Int. J. Biol. Macromol. 177, 29 (2021)
P. Maneechakr, S. Karnjanakom, Facile utilization of magnetic MnO2@Fe3O4@sulfonated carbon sphere for selective removal of hazardous Pb(II) ion with an excellent capacity: Adsorption behavior/isotherm/kinetic/thermodynamic studies. J. Environ. Chem. Eng. 9, 106191 (2021)
H.J. Kim, J.M. Lee, J.H. Choi, D.H. Kim, G.S. Han, H.S. Jung, Synthesis and adsorption properties of gelatin-conjugated hematite (α-Fe2O3) nanoparticles for lead removal from wastewater. J. Hazard. Mater. 416, 125696 (2021)
Y.S. Ho, G. McKay, A comparison of chemisorption kinetic models applied to pollutant removal on various sorbents. Process Saf. Environ. Prot. 76, 332 (1998)
G.F. Cerofolini, A model which allows for the Freundlich and the Dubinin- Radushkevich adsorption isotherms. Surf. Sci. 51, 333 (1975)
M. Baghdadi, E. Ghaffari, B. Aminzadeh, Removal of carbamazepine from municipal wastewater effluent using optimally synthesized magnetic activated carbon: adsorption and sedimentation kinetic studies. J. Environ. Chem. Eng. 4, 3309 (2016)
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
MAH, AA performed writing-original draft, and done experiment. AAA performed writing–original draft, and supervision. SG contributed to conceptualization, review and editing.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hassan, M.A., Amooey, A.A., Ghasemi, S. et al. Response surface methodology optimized adsorptive removal of the lead(II) ion from aqueous solution using reduced graphene oxide/zeolitic imidazolate framework-67. J IRAN CHEM SOC 20, 57–68 (2023). https://doi.org/10.1007/s13738-022-02643-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-022-02643-3