Abstract
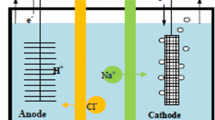
This study reports on the treatment of wastewater containing dissolved organic matter (DOM). DOM introduces complexing agents, promotes bacterial growth, and affects the color plus taste of water negatively. The normal practice for treatment such pollutants is the use of introduction of oxidizing agents in the water. However, this introduces secondary pollutants to the treated water, and to overcome the challenge, this study has developed an electrochemical method for treating wastewater with no secondary pollutants. A two-chamber electrochemical cell separated by a proton exchange membrane (PEM) was constructed with inert electrodes. The PEM was made from a conducting polymer inert in aqueous media. The anode water was bubbled with air for reduction of oxygen and therefore formation of strong oxidizing agent for the degradation of DOM. The degradation was monitored using Ultraviolet–visible (UV–VIS) spectroscopy as the potential difference across the cell was monitored. There was a significant reduction in the color and the decay followed first-order kinetics, for unimolecular degradation with a constant of 0.0148 min−1. A high potential of 1.25 V was registered within the first 40 min confirming that the degradation was spontaneous making the water safe for consumption. The degradation was confirmed by voltametric method where the concentration of iron within the vegetative matter was observed to increase at a potential of − 0.18 V with time due to the release of the labile metal ions. This shows that the constructed cell has a potential application in the remediation of domestic wastewater at a point of use.









Similar content being viewed by others
References
W.B. Jensen, J. Chem. Educ. 86(5), 545 (2009). https://doi.org/10.1021/ed086p545
L. Carrette, K.F. Andreas, U. Stimming, ChemPhysChem (2000). https://doi.org/10.1002/1439-7641(20001215)1:4%3c162:AID-CPHC162%3e3.0.CO;2-Z
B. D. James, F. D. Lomax, C.E. Thomas Jr., Directed technologies, Inc. 5 (1999)
L. Khotseng, Intechopen (2017). https://doi.org/10.5772/intechopen.79098
D. Singh, D. Pratap, Y. Baranwal, B. Kumar, R.K. Chaudhary, Ann. Biol. Res. (2010). http://scholarsresearchlibrary.com/archive.html
A. E. Franks, N. Malvankar, K. P. Nevin, Biofuels. (2014). https://doi.org/10.4155/bfs.10.2
R.M. Allen, H.P. Bennetto, Appl. Biochem. Biotechnol. (1993). https://doi.org/10.1007/bf02918975
L. Shi, W. Li, F. Wang, J. Solut. Chem. (2009). https://doi.org/10.1007/s10953-009-9391-6
M.S. Karve, N.R. Kale, A spectrophotometric method for the determination of iodine binding capacity for starch and its components. Starch 44(1), 19–21 (1992). https://doi.org/10.1002/star.19920440106
C.U. Cano, Rev. Mex. Fis. 59(2), 85–92 (2013)
D. Grieshaber, R. MacKenzie, J. Vörös, E. Reimhult, Sensor (2008). https://doi.org/10.3390/s80314000
B. Akinwolemiwa, C. Peng, Z. George, Chen. J. Electrochem. Soc. (2015). https://doi.org/10.1149/2.0111505jes
S. Mukamel, Annu. Rev. Phys. Chem. 51, 691–729 (2000)
B. Wijnen, G.C. Anzalone, J.M. Pearce, J. Water Sanit. Hyg. Dev. (2014). https://doi.org/10.2166/washdev.2014.137
I. Mwangi, G. Kiriro, S. Swaleh, R. Wanjau, P. Mbugua, J.C. Ngila, Int. J. Recycl. Org. Waste Agric. (2019). https://doi.org/10.1007/s40093-019-0242-1
K. Shigehara, E. Tsuchida, C. Fred, A. Kiy, Solid State Ionics 14, 85–91 (1984). https://doi.org/10.1016/0167-2738(84)90081-x
G. Gebel, Polymer (2000). https://doi.org/10.1016/S0032-3861(99)00770-3
H. Huang, J. Yao, L. Li, F. Zhu, Z. Liu, X. Zeng, X. Yu, Z. Huang, J. Mater. Sci. (2016). https://doi.org/10.1007/s10853-016-0137-8
A. Tasnim, Simon Fraser University Dissertation (2015)
N. Islam, H. Wang, F. Maqbool, V. Ferro, Molecules (2019). https://doi.org/10.3390/molecules24071271
Y.S. Ho, G. McKay, D.J. Wase, C.F. Foster, Adsorpt. Sci. Technol. 18, 639–650 (2000)
S. Lagergreg, Handlingar 24(4), 1–39 (1898)
F.E. Batal, Indian J. Pure Appl. Phys. 47, 631–642 (2009)
R.J. Toh, H.W.K. Peng, M. Pumera, Direct in vivo electrochemical detection of haemoglobin in red blood cells. Sci. Rep. 4(6209), 1–7 (2014). https://doi.org/10.1038/srep06209
S. E. Oh, B.E. Logan, J. Power Sources. (2007). https://doi.org/10.1016/j.jpowsour.2007.02.016
A. Bard, L. R. Faulkner, 2nd ed. (Wiley, New York: 2001) 864pp
Acknowledgements
The authors acknowledge the fundings and support provided by Kenyatta University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mwangi, I., Kinyua, E., Wanjau, R. et al. Remediation of domestic wastewater by electrochemical oxidation of dissolved organic species. J IRAN CHEM SOC 18, 581–588 (2021). https://doi.org/10.1007/s13738-020-02043-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-020-02043-5