Abstract
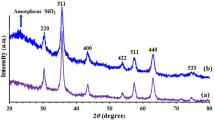
Magnetically separable mesoporous poly-melamine–formaldehyde nanocomposite (Fe3O4@mPMF) has been synthesized and characterized by FTIR spectroscopy, SEM, XRD spectroscopy and EDS. The magnetically separable Fe3O4@mPMF catalyst had excellent efficiency for the synthesis a wide diversity of imidazo[1,2-a]pyridines by a three-component reaction of a 2-aminopyridine, aldehyde and isocyanide in good-to-excellent yields within a short reaction time, through an environmental friendliness and straightforward procedure. The nanocomposite that is easily recoverable and also found to be reusable can be recovered and reused when the reaction is completed for several times without distinct deterioration in catalytic activity.
Graphic abstract







Similar content being viewed by others
References
A. Alizadeh, Q. Oskueyan, S. Rostamnia, Synthesis 17, 2637–2640 (2007)
M.A.P. Martins, C.P. Frizzo, D.N. Moreira, N. Zanatta, H.G. Bonacorso, Chem. Rev. 108, 2015 (2008)
A. Shaabani, A. Maleki, A.H. Rezayan, A. Sarvary, Mol. Divers. 15, 41–68 (2011)
S. Gupta, M. Lakshman, J. Med. Chem. Sci. 2, 51–54 (2019)
S. Sajjadifar, K. Pal, H. Jabbari, O. Pouralimardan, F. Divsar, S. Mohammadi-Aghdam, I. Amini, H. Hamidi, Chem. Methodol. 3, 226–236 (2019)
H. Aghahosseini, A. Ramazani, N. Safarvand-Jalayer, Z. Ranjdoost, A. Souldozi, K. Slepokura, T. Lis, Org. Lett. 21, 22–26 (2019)
A.K. Chakraborti, S. Rudrawar, K.B. Jadhav, G. Kaura, S.V. Chankeshwara, Green Chem. 9, 1335 (2007)
A. Ramazani, Rezaei, Org. Lett. 12, 2852–2855 (2010)
H. Ahankar, A. Ramazani, K. Ślepokura, T. Lis, S.W. Joo, Green Chem. 18, 3582–3593 (2016)
H.C. Hu, Y.H. Liu, B.L. Li, Z.S. Cui, Z.H. Zhang, RSC Adv. 5, 48675 (2015)
P.H. Li, B.L. Li, H.C. Hu, X.N. Zhao, Z.H. Zhang, Catal. Commun. 46, 118 (2014)
I. Yavari, H. Djahaniani, F. Nasiri, Tetrahedron 59, 9409–9412 (2003)
Z. Arzehgar, S. Sajjadifar, M.H. Fekri, Chem. Methodol. 3, 251–260 (2019)
A. Ramazani, M. Khoobi, A. Torkaman, F. Zeinali-Nasrabadi, H. Forootanfar, M. Shakibaie, M. Jafari, A. Amiri, S. Emami, M.A. Faramarzi, A. Foroumadi, A. Shafiee, Eur. J. Med. Chem. 78, 151–156 (2014)
H. Aghahosseini, A. Ramazani, K. Ślepokura, T. Lis, J. Colloid Interface Sci. 511, 222–232 (2018)
I. Yavari, Ramazani, J. Chem. Res. 382–383 (1996)
R. Motamedi, F. Ebrahimi, G. Rezanejade-Bardijee, Asian J. Green Chem. 3, 22–23 (2019)
Z. Arzehgar, S. Sajjadifar, H. Arandiyan, Asian J. Green Chem. 3, 43–52 (2019)
A. Shaabani, E. Soleimani, A. Maleki, Tetrahedron Lett. 47, 3031–3034 (2007)
E. Ruijter, R. Scheffelaar, R.V.A. Orru, Angew. Chem. Int. Ed. 50, 6234 (2011)
B.B. Toure, D.G. Hall, Chem. Rev. 109, 4439 (2009)
A. Shaabani, A. Maleki, Appl. Catal. A Gen. 331, 149–151 (2007)
A. Shaabani, A. Maleki, J. Moghimi-Rad, J. Org. Chem. 72, 6309–6311 (2007)
A. Dömling, I. Ugi, Angew. Chem. Int. Ed. 39, 316831 (2000)
J. Zhu, Eur. J. Org. Chem. 1133 (2007)
H. Bienayme, K. Bouzid, Angew. Chem. Int. Ed. 37, 2234 (1998)
N. Devi, R.K. Rawal, V. Singh, Tetrahedron 71, 183 (2015)
S. Rostamnia, A. Hassankhani, RSC Adv. 3, 18626 (2013)
S. Rostamnia, K. Lamei, M. Mohammadquli, M. Sheykhan, A. Heydari, Tetrahedron Lett. 53, 5257–5260 (2012)
S. Vidyacharan, A.H. Shinde, D.S. Sharada, Green Chem. 16, 1168 (2014)
J. Lu, X.T. Li, E.Q. Ma, L.P. Mo, Z.H. Zhang, Chem. Catal. Chem. 6, 2854 (2014)
S. Swami, N. Devi, A. Agarwala, V. Singh, R. Shrivastava, Tetrahedron Lett. 57, 1346 (2016)
M. Adib, M. Mahdavi, M.A. Noghani, P. Mirzaei, Tetrahedron Lett. 48, 7263 (2007)
M. Zhang, J. Lu, J.N. Zhang, Z.H. Zhang, Catal. Commun. 78, 26 (2016)
E. Gross, J.H. Liu, F.D. Toste, G.A. Somorjai, Nat. Chem. 4, 947 (2012)
G. Kyriakou, M.B. Boucher, A.D. Jewell, E.A. Lewis, T.J. Lawton, A.E. Baber, Science 335, 1209 (2012)
M. Jakuttis, A. Schönweiz, S. Werner, R. Franke, K.D. Wiese, M. Haumann, P. Wasserscheid, Angew. Chem. Int. Ed. 50, 4492 (2011)
K.S. Palla, T.J. Hurlburt, A.M. Buyanin, G.A. Somorjai, M.B. Francis, J. Am. Chem. Soc. 139, 1967 (2017)
P. Puthiaraj, Y. Lee, S. Zhang, W.S. Ahn, J. Mater. Chem. A 4, 16288 (2016)
T. Blasco, Chem. Soc. Rev. 39, 4685 (2010)
D. Schwarz, J. Weber, Polymer 155, 83 (2018)
D. Schwarz, J. Weber, Macromol. Mater. Eng. 300, 531 (2015)
D. Yang, P. Liu, N. Zhang, J. You, H. Wang, ChemCatChem 6, 3434 (2014)
R.A. Molla, M.A. Iqubal, K. Ghosh, S.M. Kamaluddin, RSC Adv. 4, 48177 (2014)
M.X. Tan, Y.N. Sum, J.Y. Ying, Y. Zhang, Energy Environ. Sci. 6, 3254 (2013)
M.X. Tan, Y. Zhang, J.Y. Ying, Chemsuschem 6, 1186 (2013)
M.X. Tan, L. Gu, N. Li, J.Y. Ying, Y. Zhang, Green Chem. 15, 1127 (2013)
Y. Song, R. Ma, C. Jiao, L. Hao, C. Wang, Q. Wu, Z. Wang, Microchim. Acta 185, 19 (2018)
N. Azizi, F. Ebrahimi, E. Aakbari, F. Aryanasab, M.R. Saidi, Synlett 2797 (2007)
N. Azizi, A. Khajeh-Amiri, H. Ghafuri, M. Bolourtchian, M.R. Saidi, Synlett 2245 (2009)
B. Mirmashhori, N. Azizi, M.R. Saidi, J. Mol. Catal. A Chem. 247, 159 (2006)
N. Azizi, Z. Manocheri, Res. Chem. Intermed. 38, 1495 (2012)
M. Bolourtchian, M.M. Hashemi, Monatshefte fur Chemie 140, 1471 (2009)
T. Sanaeishoar, H. Tavakkoli, F. Mohave, Appl. Catal. A 470, 56–62 (2014)
M.L. Bode, D. Gravestock, S.S. Moleele, C.W. Westhuyzen, S.C. Pelly, Bioorg. Med. Chem. 19, 4227–4237 (2011)
D.B. Salunke, E. Yoo, N.M. Shukla, R. Balakrishna, S.S. Malladi, V.W. Day, X. Wang, J. Med. Chem. 55, 8137–8151 (2012)
A. Shaabani, E. Soleimani, A. Maleki, J. Moghimi-Rad, Synth. Commun. 38, 1090–1095 (2008)
Acknowledgements
The financial support of this work provided by Chemistry and Chemical Engineering Research Center of Iran and Science and Research Branch of Islamic Azad University is gratefully appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Heydari, M., Azizi, N., Mirjafari, Z. et al. Magnetic mesoporous poly-melamine–formaldehyde: an efficient and recyclable catalyst for straightforward one-pot synthesis of imidazo[1,2-a]pyridines. J IRAN CHEM SOC 16, 2357–2363 (2019). https://doi.org/10.1007/s13738-019-01705-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-019-01705-3