Abstract
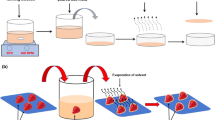
A novel monolithic molecularly imprinted polymer fiber-solid phase microextraction method was developed for the simultaneous determination of cholesterol and β-sitosterol in cow milk, egg yolk and olive oil samples by high-performance liquid chromatography. A flexible MIP monolithic fibers with high chemical and thermal stabilities were simply synthesized using a narrow bore silica capillary as a mold by the copolymerization of methacrylic acid and ethylene glycol dimethacrylate imprinted with β-sitosterol. The prepared MIPs were characterized by Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy (SEM). The main factors that affect the extraction efficiency, such as extraction time, desorption time, desorption solvent, and stirring rate were investigated and optimized. Under the optimized extraction conditions, the limits of detection of the proposed method were 0.03 µg L−1 and 0.05 µg L−1, for cholesterol and β-sitosterol, respectively. Satisfactory linearity was achieved in the range of 0.1–20 µg L−1 for analytes with the correlation coefficients (r) above 0.9975 and RSD less than 3.5%. This procedure was successfully applied for the determination of cholesterol and β-sitosterol in different food samples.








Similar content being viewed by others
References
S. Schrack, C. Hohl, W. Schwack, J. Chromatogr. A 1473, 8–10 (2016)
K.V. Derina, E.I. Korotkova, E.V. Dorozhko, O.A. Voronova, D.A. Vishenkova, Proc. Chem. 10, 508–513 (2014)
F. Stellaard, D. Lutjohann, J. Clin. Lipidol. 9, 14–25 (2015)
J. Chitra, M. Ghosh, H.N. Mishra, Food Cont. 78, 342–349 (2017)
Y. Zhang, G. Wang, Y. Jin, Y. Deng, Y. Zhao, Food Cont. 60, 189–195 (2016)
S. Hirany, D. Li, I. Jialal, Am. J. Med. 102, 48–53 (1997)
L.C. de Figueiredo, E.G. Bonafe, J.G. Martins, A.F. Martins, S.A. Maruyama,, P.B.F. Biondo, M. Matsushita, J.V. Visentainer, O. de Oliveira Santos Junior. Food Chem. 240, 441–447 (2018)
S. Balme, F.O. Gulacar, Food Chem. 132, 613–618 (2012)
S. Alvarez-Sala, V. Blanco-Morales, A. Cilla, G. Garcia-Llatas, L.M. Sánchez-Siles, R. Barberá, M.J. Lagarda, J. Food Compos. Anal. (2017) https://doi.org/10.1016/j.jfca.2017.03.011
M.M. Delgado-Zamarreno, C. Fernandez-Prieto, M. Bustamante-Rangel, L. Perez-Martin, Food Chem. 192, 825–830 (2016)
M.J. Lagarda, G. Garcia-Llatas, R. Farre, J. Pharm. Biomed. Anal. 41, 1486–1496 (2006)
L. Millán, M.C. Sampedro, A. Sanchez, M.A. Goicolea, R.J. Barrio, J. Food Compos. Anal. 42, 171–178 (2015)
Z. Zhang, W. Tan, Y. Hu, G. Li, J. Chromatogr. A 1218, 4275–4283 (2011)
V. Aggarwal, J. Malik, A. Prashant, P.K. Jaiwal, C.S. Pundir, Anal. Biochem. 500, 6–11 (2016)
Y.Z. Chen, S.Y. Kao, H.C. Jian, Y.M. Yu, J.Y. Li, W.H. Wang, J. Food. Drug. Anal. 23, 636–644 (2015)
W.F. Wu, Q.H. Wang, T. Zhang, S.H. Mi, Y. Liu, L.Y. Wang, Clin. Biochem. 46, 993–998 (2013)
M. Hasan, W. Siegmund, S. Oswald, J. Chromatogr. B 1033–1034, 193–199 (2016)
C. Yun, T. Yin, K. Shatzer, D.G. Burrin, L. Cui, Y. Tu, J. Chromatogr. B 1025, 76–82 (2016)
M. Okazaki, S. Usui, M. Nakamura, S. Yamashita, Clin. Chim. Acta. 395(67), 62 (2008)
T.G. Albuquerque, M.B.P.P. Oliveira, A. Sanches-Silva, H.S. Costa, Food Chem. 193, 18–25 (2016)
J. John, A. Reghuwanshi, U.K. Aravind, C.T. Aravindakumar, J. Food. Drug. Anal. 23, 219–224 (2015)
X.H. Xu, R.K. Li, J. Chen, P. Chen, X.Y. Ling, P.F. Rao, J. Chromatogr. B 768, 369–373 (2002)
D. Naviglio, M. Gallo, L.L. Grottaglie, C. Scala, L. Ferrara, A.Santini, Food Chem. 132, 701–708 (2012)
A. Gorassini, G. Verardo, S.C. Fregolent, R. Bortolomeazzi, Food Chem. 230, 604–610 (2017)
M. Narvaez-Rivas, A.J. Pham, M.W. Schilling, M. León-Camacho, Talanta 122, 58–62 (2014)
S. Balme, F.O. Gülaçar, Food Chem. 132, 613–618 (2012)
M.W. Heaven, D. Nash, Food Cont. 27, 214–227 (2012)
S. Ansari, M. Karimi, Talanta 164, 612–625 (2017)
A. Anene, K. Hosni, Y. Chevalier, R. Kalfat, S. Hbaieb, Food Cont. 70, 90–95 (2016)
Y.P. Song, L. Zhang, G. N.Wang, J.X. Liu, J. Liu, J.P. Wang, Food Cont. 82, 233–242 (2017)
I. Pereira, M.F. Rodrigues, A.R. Chaves, B.G.Vaz Talanta 178, 507–518 (2018)
H. Bagheri, A. Shirzadmehr, M. Rezaei, J. Mol. Liq. 212, 96–102 (2015)
R. Mirzajani, Z. Ramezani, F. Kardani, Microchem. J. 130, 93–101 (2017)
D. Djozan, T. Baheri, J. Chromatogr. A 1166, 16–23 (2007)
M. Fayazi, M. Ghanei-Motlagh, M.A. Taher, R. Ghanei-Motlagh, M.R. Salavati, J. Hazard. Mater. 309, 27–36 (2016)
R. Mirzajani, F. Kardani, J. Pharm. Biomed. Anal. 122, 98–109 (2016)
R. Mirzajani, N. Pourreza, J. Burromandpiroze, Ultrason. Sonochem. 40, 101–112 (2018)
C. Husche, O. Weingärtner, H. Pettersson, T. Vanmierlo, M. Böhm, U. Laufs, Chem. Phys. Lipids. 164, 425–431 (2011)
I. Mendiara, K. Bentayeb, C. Nerín, C. Domeño, Talanta 132, 690–697 (2015)
Y. Shi, J.H. Zhang, D. Shi, M. Jiang, Y.X. Zhu, S.R. Mei, J. Pharm. Biomed. Anal. 42, 549–555 (2006)
F. Kardani, A. Daneshfar, R. Sahrai, Anal. Chim. Acta 701, 232–237 (2011)
A. Daneshfar, T. Khezeli, H.J. Lotfi, J. Chromatogr. B 877, 456–460 (2009)
Acknowledgements
The Authors greatly appreciate the financial support of this work by Shahid Chamran University of Ahvaz Research Council.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kardani, F., Mirzajani, R. & Ramezani, Z. Direct cholesterol and β-sitosterol analysis in food samples using monolithic molecularly-imprinted solid-phase microextraction fibers coupled with high performance liquid chromatography. J IRAN CHEM SOC 15, 2877–2888 (2018). https://doi.org/10.1007/s13738-018-1474-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1474-0