Abstract
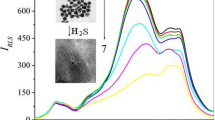
Resonance light-scattering (RLS) technique was developed for studying the interaction of silver nanoparticles (Ag NPs) with bisphenol A. A simple and environmentally friendly method was developed to synthesize Ag NPs using cinnamon extract. Synthesized nanoparticles were characterized using various measurement techniques. The synthesized Ag NPs were nearly spherical, with the sizes ranging from 30 to 60 nm. Spectral analysis indicated that the cinnamon extract acted as the reducing and capping agents on the surface of Ag NPs. RLS technique was used as the detection method. Light-scattering properties of the synthesized nanoparticles in the presence or absence of bisphenol A was selected as the detection signal. Under the optimal conditions, the linear dynamic range and RSD were found to be 0.01–10.0 mg L−1 and 2.78% (n = 3), respectively. A limit of detection of 0.005 mg L−1 was obtained for the determination of bisphenol A. The obtained results showed successful application of the method for the analysis of bisphenol A in real samples.









Similar content being viewed by others
References
T. Madrakian, A. Afkhami, E. Vanaei, M. Ahmadi, Anal. Methods 7, 6299 (2015)
L. Molina-García, M.L. Fernández-de Córdova, A. Ruiz-Medina, Talanta 96, 195 (2012)
J. Peretz, L. Vrooman, W.A. Ricke, P.A. Hunt, S. Ehrlich, R. Hauser, V. Padmanabhan, H.S. Taylor, S.H. Swan, C.A. VandeVoort, Environ. Health Perspect. 122, 775 (2014)
N. Dorival-García, A. Zafra-Gómez, A. Navalón, J. Vílchez, Talanta 101, 1 (2012)
K. Inoue, K. Kato, Y. Yoshimura, T. Makino, H. Nakazawa, J. Chromatogr. B 749, 17 (2000)
M. Kawaguchi, K. Inoue, M. Yoshimura, R. Ito, N. Sakui, N. Okanouchi, H. Nakazawa, J. Chromatogr. B 805, 41 (2004)
D. Yao, A. Liang, W. Yin, Z. Jiang, Luminescence 29, 516 (2014)
Z. Mei, H. Chu, W. Chen, F. Xue, J. Liu, H. Xu, R. Zhang, L. Zheng, Biosens. Bioelectron. 39, 26 (2013)
Z. Mei, W. Qu, Y. Deng, H. Chu, J. Cao, F. Xue, L. Zheng, H.S. El-Nezamic, Y. Wu, W. Chen, Biosens. Bioelectron. 49, 457 (2013)
R.F. Pasternack, C. Bustamante, P.J. Collings, A. Giannetto, E.J. Gibbs, J. Am. Chem. Soc. 115, 5393 (1993)
M. Ahmadi, T. Madrakian, A. Afkhami, Anal. Chim. Acta 852, 250 (2014)
Z. Chen, T. Song, S. Wang, X. Chen, J. Chen, Y. Li, Biosens. Bioelectron. 25, 1947 (2010)
Z. Chen, T. Song, Y. Peng, X. Chen, J. Chen, G. Zhang, S. Qian, Analyst 136, 3927 (2011)
F. Fazl, M. Ahmadi, T. Madrakian, A. Afkhami, Sens. Actuators, B 223, 379 (2016)
L. Yu, Y. Zhang, R. Chen, D. Zhang, X. Wei, F. Chen, J. Wang, M. Xu, Talanta 131, 475 (2015)
H. Cao, M. Wei, Z. Chen, Y. Huang, Analyst 138, 2420 (2013)
J. Dong, A. Liang, Y. Luo, Z. Jiang, Arab. J. Chem. (2015). https://doi.org/10.1016/j.arabjc.2015.02.020
Z. Chen, J. Liu, Y. Han, Talanta 71, 1246 (2007)
J. Zhu, Y. Wang, L. Huang, Y. Lu, Phys. Lett. A 323, 455 (2004)
H. Jiang, Z. Chen, H. Cao, Y. Huang, Analyst 137, 5560 (2012)
Z. Chen, X. Zhang, H. Cao, Y. Huang, Analyst 138, 2343 (2013)
A. Afkhami, S. Sayari, R. Moosavi, T. Madrakian, J. Ind. Eng. Chem. 21, 920 (2015)
T. Madrakian, A. Afkhami, H. Mahmood-Kashani, M. Ahmadi, Talanta 105, 255 (2013)
R. Moosavi, S. Ramanathan, Y.Y. Lee, K.C.S. Ling, A. Afkhami, G. Archunan, P. Padmanabhan, B. Gulyás, M. Kakran, S.T. Selvan, RSC Adv. 5, 76442 (2015)
K.J. Rao, S. Paria, ACS Sustain. Chem. Eng. 3, 483 (2015)
Y. Zhang, D. Yang, Y. Kong, X. Wang, O. Pandoli, G. Gao, Nano Biomed. Eng. 2, 252 (2010)
H. Parham, S. Saeed, Talanta 131, 570 (2015)
T. Wriedt, Mie theory: a review, in ed. by W. Hergert, T. Wriedt. The Mie Theory, vol. 169 of Springer Series in Optical Sciences (Springer, Berlin, Germany, 2012)
H. Parham, N. Pourreza, F. Marahel, Talanta 141, 143 (2015)
T. Madrakian, M. Ahmadi, A. Afkhami, M. Soleimani, Analyst 138, 4542 (2013)
Z.J. Tan, F.F. Li, J. Cent. South Univ. 19, 2136 (2012)
P. Viñas, I. López-García, N. Campillo, R.E. Rivas, M. Hernández-Córdoba, Anal. Bioanal. Chem. 404, 671 (2012)
N. Yildirim, F. Long, M. He, H.-C. Shi, A.Z. Gu, Environ. Sci. Technol. 16, 1379 (2014)
S. Cunha, J. Fernandes, Talanta 83, 117 (2010)
Acknowledgements
The Bu-Ali Sina University Research Council and Center of Excellence in Development of Environmentally Friendly Methods for Chemical Synthesis (CEDEFMCS) financially supported this work, and the authors acknowledge that for providing financial resource to this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pirdadeh-Beiranvand, M., Afkhami, A. & Madrakian, T. Ag nanoparticles for determination of bisphenol A by resonance light-scattering technique. J IRAN CHEM SOC 15, 1527–1534 (2018). https://doi.org/10.1007/s13738-018-1350-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1350-y