Abstract
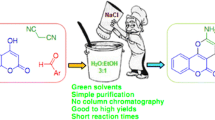
A family of 3,4-dihydropyrano[3,2-c]chromenes were synthesized from 4-hydroxycoumarin and malononitrile via a one-pot reaction under solvent-free conditions at 100 °C catalyzed by Ni@Imine-Li+-MMT. This methodology tolerates most of the substrates and has the merits of lower loading of the catalyst, absence of solvents, excellent yields and reusability of the catalyst. A reasonable mechanism is also proposed. This catalytic system can be reused for at least five times with a negligible loss of activity. The prepared catalyst was characterized by using FTIR, TGA, SEM, TEM, uv-DRS, EDX and XRD.











Similar content being viewed by others
References
G.A. Olah, A. Molnar, Hydrocarbon Chemistry (Wiley, New York, 1995)
P. Ferreira, I.M. Fonseca, A.M. Ramos, J. Vital, J.E. Castanheiro, Catal. Commun. 12, 573 (2011)
S.E. Sen, S.M. Smith, K.A. Sullivan, Tetrahedron 55, 12657 (1999)
H. Salemi, B. Kaboudin, F. Kazemi, T. Yokomatsu, RSC Adv. 6, 52656 (2016)
M. Baghayeri, H. Veisi, H. Veisi, B. Maleki, H. Karimi-Maleh, H. Beitollahi, RSC Adv. 4, 49595 (2014)
M.A. Zolfigol, T. Azadbakht, V. Khakyzadeh, R. Nejatyami, D.M. Perrin, RSC Adv. 4, 40036 (2014)
S. Rostamnia, N. Nouruzi, H. Xin, R. Luque, Catal. Sci. Technol. 5, 199 (2015)
S. Rostamnia, E. Doustkhah, H. Golchin-Hosseini, B. Zeynizadeh, H. Xin, R. Luque, Catal. Sci. Technol. 6, 4124 (2016)
A. Khorshidi, K. Tabatabaeian, H. Azizi, M. Aghaei-Hashjin, E. Abbaspour-Gilandeh, RSC Adv. 7, 17732 (2017)
M. Mamaghani, F. Shirini, M. Sheykhan, M. Mohsenimehr, RSC Adv. 5, 44524 (2015)
K. Tabatabaeian, M.A. Zanjanchi, N.O. Mahmoodi, T. Eftekhari, RSC Adv. 5, 101013 (2015)
M. Masteri-Farahani, S. Abednatanzi, Inorg. Chem. Commun. 37, 39 (2013)
N. Anand, K.H.P. Reddy, V. Swapna, K.S.R. Rao, D.R. Burri, Microporous Mesoporous Mater. 143, 132 (2011)
S.J. Mohr, M.A. Chirigos, F.S. Fuhrman, J.W. Pryor, Cancer Res. 35, 3750 (1975)
D.O. Moon, K.C. Kim, C.Y. Jin, M.H. Han, C. Park, K.J. Lee, Y.M. Park, Y.H. Choi, G.Y. Kim, Int. Immunopharmacol. 7, 222 (2007)
M. Rueping, E. Sugiono, E. Merino, Chem. Eur. J. 14, 6329 (2008)
A.G. Martinez, L.J. Marco, Bioorg. Med. Chem. Lett. 7, 3165 (1997)
T. Raj, R.K. Bhatia, A. Kapur, M. Sharma, A.K. Saxena, M.P.S. Ishar, Eur. J. Med. Chem. 45, 790 (2010)
Y.L. Zhang, B.Z. Chen, K.Q. Zheng, M.L. Xu, X.H. Lei, X.B. Yaoxue, Chem. Abstr. 96, 135383 (1982)
H. Mehrabi, M. Kazemi-Mireki, Chin. Chem. Lett. 22, 1419 (2011)
W. Hong-juan, Z. Jie, Z. Zhan-Hui, Monatsh. Chem. 141, 1107 (2010)
K. Niknam, A. Piran, Green Sustain. Chem. 3, 1 (2013)
M. Safaiee, M.A. Zolfigol, F. Afsharnadery, S. Baghery, RSC Adv. 5, 102340 (2015)
A. Montaghami, N. Montazeri, Orient. J. Chem. 30, 1361 (2014)
S. Abdolmohammadi, S. Balalaie, Tetrahedron Lett. 48, 3299 (2007)
M.J. Khurana, B. Nand, P. Saluja, Tetrahedron 66, 5637 (2010)
A. Khorshidi, S. Shariati, Chin. J. Catal. 36, 778 (2015)
A. Khorshidi, S. Shariati, RSC Adv. 4, 41469 (2014)
A. Khorshidi, S. Heidari, RSC Adv. 5, 32804 (2015)
K. Tabatabaeian, A. Khorshidi, A. Dadashi, M. Khoshnood, Chin. Chem. Lett. 23, 165 (2012)
A. Khorshidi, N. Mardazad, Z. Shaabanzadeh, Tetrahedron Lett. 55, 3873 (2014)
H. Mehrabi, H. Abusaidi, J. Iran. Chem. Soc. 7, 890 (2010)
A. Shaabani, S. Samadi, Z. Badri, A. Rahmati, Catal. Lett. 104, 39 (2005)
H.J. Wang, J. Lu, Z.H. Zhang, Monatsh. Chem. 141, 1107 (2010)
M. Khoobi, L. Ma’mani, F. Rezazadeh, Z. Zareie, A. Foroumadi, A. Ramazani, A. Shafiee, J. Mol. Catal. A Chem. 359, 74 (2012)
W. Hong-juan, Z. Jie, Z. Zhan-Hui, Monatsh. Chem. 141, 1107 (2010)
W. Xiang-Shan, Z. Zhao-Sen, S. Da-Qing, W. Xian- Yong, Z. Zhi-Min, Chin. J. Org. Chem. 25, 1138 (2005)
J. Zheng, Y. Li, Mendeleev Commun. 21, 280 (2011)
H. Nagabhushana, S.S. Saundalkar, L. Muralidhar, B.M. Nagabhushana, C.R. Girija, D. Nagaraja, M.A. Pasha, V.P. Jayashankara, Chin. Chem. Lett. 22, 143 (2011)
K. Niknam, A. Jamali, Chin. J. Catal. 33, 1840 (2012)
H. Mehrabi, M. Kazemi-Mireki, Chin. Chem. Lett. 22, 1419 (2011)
G.M. Ziarani, A. Badiei, M. Azizi, P. Zarabadi, J. Chem. Eng. 30, 59 (2011)
Acknowledgements
Partial support of this study by the Research Council of University of Guilan is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Azizi, H., Khorshidi, A. & Tabatabaeian, K. Efficient synthesis of 3,4-dihydropyrano[3,2-c]chromenes catalyzed by Ni(II)-functionalized Li+-Montmorillonite in a one-pot fashion under solvent-free conditions. J IRAN CHEM SOC 15, 1023–1032 (2018). https://doi.org/10.1007/s13738-018-1299-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1299-x