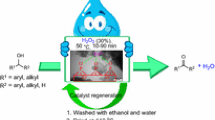
Abstract
Nickel zirconium phosphate nanoparticles have been used as an efficient catalyst for the acetylation of a wide range of alcohols and phenols with acetic anhydride in good to excellent yields under solvent‐free conditions. The steric and electronic properties of the different substrates had a significant influence on the reaction conditions required to achieve the acetylation. The catalyst used in the current study was characterized by inductively coupled plasma optical emission spectroscopy, X‐ray diffraction, N2 adsorption–desorption, scanning electron microscopy, and transmission electron microscopy. This nanocatalyst could also be recovered and reused at least six times without any discernible decrease in its catalytic activity.
Graphical abstract








Similar content being viewed by others
References
F. Li, M. Wei, J. He, Y. Du, P. Sun, D.G. Evans, X. Duan, Chin. J. Catal. 20, 514 (1999)
L. Sun, W.J. Boo, H.J. Sue, A. Clearfield, New J. Chem. 31, 39 (2007)
A.R. Hajipour, H. Karimi, Mater. Lett. 116, 356 (2014)
V.I. Pet’kov, A.V. Markin, I.A. Shchelokov, M.V. Sukhanov, N.N. Smirnova, Russ. J. Phys. Chem. 81, 1728 (2007)
Q. Shi, S. Tan, Y. Ouyang, Q. Yang, A. Chen, W. Li, X. Shu, J. Feng, J. Fang, Y. Chen, Adv. Matter. Sci. 150–151, 852 (2011)
X. Cai, G.J. Dai, S.Z. Tan, Y. Ouyang, Y.S. Ouyang, Q.S. Shi, Mater. Lett. 67, 199 (2012)
A. Clearfield, J.M. Kalnins, J. Inorg. Nuc. Chem. 40, 1933 (1978)
S. Allulli, C. Ferragina, A. La Ginestra, M.A. Massucci, N. Tomassini, A.A.G. Tomlinson, J. Chem. Soc. Dalton Trans. 2115 (1976)
B. Shpeizer, D.M. Poojary, K. Ahn, C.E. Runyan Jr, A. Clearfield, Science 266, 1357 (1994)
B.G. Shpeizer, P. Sylvester, R.A. Cahill, A. Clearfield, Chem. Mater. 11, 1201 (1999)
G. Alberti, M.G. Bernasconi, U. Costantino, J.S. Gill, J. Chromatogr. A 132, 477 (1977)
G. Alberti, M. Casciola, U. Costantino, R. Vivani, Adv. Mater. 8, 291 (1996)
Y. Yang, G. Dai, S. Tan, Y. Liu, Q. Shi, Y. Ouyang, J. Rare Earths 29, 308 (2011)
G. Dai, A. Yu, X. Cai, Q. Shi, Y. Ouyang, S. Tan, J. Rare Earths 30, 820 (2012)
Q.R. Zhang, W. Du, B.C. Pan, B.J. Pan, W.M. Zhang, Q.J. Zhang, Z.W. Xu, Q.X. Zhang, J. Hazard. Mater. 152, 469 (2008)
U. Costantino, L. Szirtes, E. Kuzmann, J. Megyeri, K. Lázár, Solid State Ionics 141–142, 359 (2001)
S. Khare, R. Chokhare, J. Mole. Catal. A: Chem. 344, 83 (2011)
Y. Izumi, Y. Mizutani, Bull. Chem. Soc. Jpn 52, 3065 (1979)
M. Iwamoto, Y. Nomura, S. Kagawa, J. Catal. 69, 234 (1981)
A.I. Pylinina, I.I. Mikhalenko, Russ. J. Phys. Chem. 87, 372 (2013)
A.I. Pylinina, I.I. Mikhalenko, Russ. J. Phys. Chem. 85, 2109 (2011)
A. Hajipour, H. Karimi, M. Karimzadeh, Monatsh. Chem. 145, 1461 (2014)
A.R. Hajipour, H. Karimi, Chin. J. Catal. 35, 1529 (2014)
A.R. Hajipour, H. Karimi, Chin. J. Catal. (2014). doi:10.1016/S1872-2067(14)60185-6
H.J. Yoon, S.M. Lee, J.-H. Kim, H.J. Cho, J.W. Choi, S.H. Lee, Y.S. Lee, Tetrahedron Lett. 49, 3165 (2008)
H. Sharghi, M. Jokar, M.M. Doroodmand, Adv. Synth. Catal. 353, 426 (2011)
S.A. Taghavi, M. Moghadam, I. Mohammadpoor-Baltork, S. Tangestaninejad, V. Mirkhani, A.R. Khosropour, Inorg. Chim. Acta 377, 159 (2011)
I. Montes, D. Sanabria, M. García, J. Castro, J. Fajardo, J. Chem. Educ. 83, 628 (2006)
T.S. Reddy, M. Narasimhulu, N. Suryakiran, K.C. Mahesh, K. Ashalatha, Y. Venkateswarlu, Tetrahedron Lett. 47, 6825 (2006)
S.K. Prajapti, A. Nagarsenkar, B.N. Babu, Tetrahedron Lett. 55, 1784 (2014)
M.M. Heravi, F.K. Behbahani, V. Zadsirjan, H.A. Oskooie, J. Braz. Chem. Soc. 17, 1045 (2006)
P. Yadav, R. Lagarkha, Z.A. Balla, Asian J. Chem. 22, 5155 (2010)
L. Osiglio, G. Romanelli, M. Blanco, J. Mole. Catal. A Chem. 316, 52 (2010)
F. Tamaddon, M.A. Amrollahi, L. Sharafat, Tetrahedron Lett. 46, 7841 (2005)
R. Dalpozzo, A. De Nino, L. Maiuolo, A. Procopio, M. Nardi, G. Bartoli, R. Romeo, Tetrahedron Lett. 44, 5621 (2003)
R. Gupta, V. Kumar, M. Gupta, S. Paul, R. Gupta, Indian J. Chem. Sec. B 47, 1739 (2008)
R. Tayebee, F. Cheravi, Bull. Korean Chem. Soc. 30, 2899 (2009)
Z. Liu, Q. Ma, Y. Liu, Q. Wang, Org. Lett. 16, 236 (2014)
A.K. Chakraborti, R. Gulhane, Shivani, Synthesis 111 (2004)
K. Niknam, D. Saberi, Appl. Catal. A 366, 220 (2009)
M. Lakshmi Kantam, K. Aziz, P.R. Likhar, Catal. Commun. 7, 484 (2006)
W. Wang, W. Cheng, L. Shao, J. Yang, Catal. Lett. 121, 77 (2008)
Y. Liu, L. Liu, Y. Lu, Y.Q. Cai, Monatsh. Chem. 139, 633 (2008)
C. Yue, Q. Liu, T. Yi, Y. Chen, Monatsh. Chem. 141, 975 (2010)
F. Shirini, M.A. Zolfigol, M. Abedini, Monatsh. Chem. 140, 1495 (2009)
K. Niknam, D. Saberi, Tetrahedron Lett. 50, 5210 (2009)
F. Shirini, N.G. Khaligh, Chin. J. Catal. 34, 695 (2013)
M. Hajjami, A. Ghorbani-Choghamarani, M. Norouzi, Chin. J. Catal. 33, 1661 (2012)
N. Nowrouzi, S.Z. Alizadeh, Chin. J. Catal. 34, 1787 (2013)
A. Ghorbani-Choghamarani, N. Pourbahar, Chin. J. Catal. 33, 1470 (2012)
S. Farhadi, K. Jahanara, Chin. J. Catal. 35, 368 (2014)
A. Zarei, A.R. Hajipour, L. Khazdooz, Synth. Commun. 41, 1772 (2011)
A.R. Hajipour, L. Khazdooz, A.E. Ruoho, J. Chin. Chem. Soc. 56, 398 (2009)
P. Kumar, R.K. Pandey, M.S. Bodas, S.P. Dagade, M.K. Dongare, A.V. Ramaswamy, J. Mole. Catal. A Chem. 181, 207 (2002)
J. Iqbal, R.R. Srivastava, J. Org. Chem. 57, 2001 (1992)
F. Dehghani, A.R. Sardarian, M.M. Doroodmand, J. Iran. Chem. Soc. 11, 673 (2014)
V. Constantinou-Kokotou, A. Peristeraki, Synth. Commun. 34, 4227 (2004)
M. Alam, A. Rahman, N.M. Alandis, M.R. Shaik, Arabian J. Chem. 7, 53 (2014)
M. Esmaeilpour, A.R. Sardarian, Iran. J. Sci. Technol. Trans. A Sci. 38, 175 (2014)
F. Shirini, S. Akbari-Dadamahaleh, A. Mohammad-Khah, A.R. Aliakbar, C. R. Chim. 17, 164 (2014)
G. Meshram, V.D. Patil, Synth. Commun. 39, 4384 (2009)
S. Farhadi, K. Jahanara, A. Sepahdar, J. Iran. Chem. Soc. 11, 1103 (2014)
F. Rajabi, R. Luque, Catal. Commun. 45, 129 (2014)
A.R. Hajipour, H. Karimi, Appl. Catal. A 482, 99 (2014)
A.R. Hajipour, H. Karimi, Chin. J. Catal. 35, 1136 (2014)
T.A. Egerton, F.S. Stone, J. Chem. Soc. Faraday Trans. 1. 69, 22 (1973)
J. Sneddon, Biochem. Pharmacol. 36, 3723 (1987)
R. Pierotti, J. Rouquerol, Pure Appl. Chem. 57, 603 (1985)
Acknowledgments
We gratefully acknowledge the funding support received for this project from the Isfahan University of Technology (IUT), IR Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hajipour, A.R., Karimi, H. & Kohi, A. Highly efficient and recyclable acetylation of phenols and alcohols by nickel zirconium phosphate under solvent-free conditions. J IRAN CHEM SOC 13, 55–64 (2016). https://doi.org/10.1007/s13738-015-0711-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0711-z