Abstract
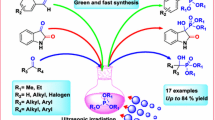
Fused α-pyrones were obtained in a convenient and green protocol from Baylis–Hillman acetates and various cyclic dicarbonyl compounds in the presence of basic ionic liquid 1-butyl-2,3-dimethylimidazolium hydroxide ([bdmim]OH) under ultrasonic irradiation. This method provided the desired α-pyrones in good to high yields (82–98 %) and lower reaction times (5–15 min).



Similar content being viewed by others
References
F. Cassidy, J.M. Evans, M.S. Hadley, A.H. Haladij, P.E. Leach, G. Stemp, J. Med. Chem. 35, 1623 (1992)
K.S. Atwal, G.J. Grover, F.N. Ferrara, S.Z. Ahmed, P.G. Sleph, S. Dzwonczyk, D.E. Normandin, J. Med. Chem. 38, 1966 (1995)
G. Appendino, M. Ottino, N. Marquez, F. Bianchi, A. Giana, M. Ballero, O. Sterner, B.L. Fiebich, E. Munoz, J. Nat. Prod. 70, 608 (2007)
N. Sitachitta, M. Gadepalli, B.S. Davidson, Tetrahedron 52, 8073 (1996)
A.F. Barrero, J.E. Oltra, M.M. Herrador, E. Cabrera, J. F.Sanchez, J.F. Quı´lez, F.J. Rojas, J.F. Reyes, Tetrahedron 49, 141 (1993)
R. Pereda-Miranda, L. Hernandez, M.J. Villavicencio, M. Novelo, P. Ibarra, J. Nat. Prod. 56, 583 (1993)
A. Evidente, A. Cabras, L. Maddau, S. Serra, A. Andolfi, A. Motta, J. Agric. Food Chem. 51, 6957 (2003)
S.R. Parker, H.G. Cutler, J.M. Jacyno, R.A. Hill, J. Agric. Food Chem. 45, 2774 (1997)
C. Altomare, R. Pengue, M. Favilla, A. Evidente, A. Isconti, J. Agric. Food Chem. 52, 2997 (2004)
M.S.C. Pedras, P.B. Chumala, Phytochemistry 66, 81 (2005)
K.M. Jenkins, S.G. Toske, P.R. Jensen, W. Fenical, Phytochemistry 49, 2299 (1998)
H. Kikuchi, K. Sasaki, J. Sekiya, Y. Moeda, A. Amagai, Y. Kubohara, Y. Oshma, Bioorg. Med. Chem. 12, 3203 (2004)
Y. Kamano, T. Nogawa, A. Yamashita, M. Hayashi, M. Inoue, J. Nat. Prod. 65, 1001 (2002)
D.H. Hua, X. Huang, M. Tamura, Y. Chen, M. Woltkamp, L.-W. Jin, E.M. Perchellet, J.-P. Perchellet, P.K. Chiang, I. Namatame, H. Tomoda, Tetrahedron 59, 4795 (2003)
N.M. Sabry, H.M. Mohamed, E.S.A.E.H. Khattab, S.S. Motlaq, A.M. El-Agrody, Eur. J. Med. Chem 46, 765 (2011)
M. Costa, F. Proenca, Tetrahedron 67, 1799 (2011)
M.N. Elinson, A.I. Ilovaisky, V.M. Merkulova, P.A. Belyakov, A.O. Chizhov, G.I. Nikishin, Tetrahedron 66, 4043 (2010)
G. R. Green, J. M. Evans, A. K. Vong, In Comprehensive Heterocyclic Chemistry II vol. 5 A. R. Katritzky, C. W. Rees, E. F. V. Scriven, Eds.; Pergamon: (Oxford, 1995) 469
S.N. Murthy, B. Madhav, V.P. Reddy, Y.V.D. Nageswar, Tetrahedron Lett. 51, 3649 (2010)
D. Ding, C.G. Zhao, Tetrahedron Lett. 51, 1322 (2010)
W. O. Foye, Prinicipi di Chimica Farmaceutica; Piccin: Padova, Italy, 1991; p 416
L. L. Adreani, E. Lapi, Boll. Chem. Farm. 99 (1960) 583. Chem. Abstr. 55 (1961) 2668d.
I. Devi, P.J. Bhuyan, Tetrahedron Lett. 45, 8625 (2004)
G. P. Ellis, The chemistry of heterocyclic compounds in chromene, chromanes and chromone; Weissberger, A.; Taylor, E. C., Eds. John Wiley, New York, 1977, p 13.
Y. Morinaka, K. Takahashi, Jpn. Patent, JP52017498, 1977
E.A. Hafez, M.H. Elnagdi, A.A. Elagamey, F.A. El-Taweel, Heterocycles 26, 903 (1987)
Y. Kuninobu, H. Takata, A. Kawata, K. Takai, Org. Lett. 10, 3133 (2008)
Y. Kuninobu, A. Kawata, M. Nishi, H. Takata, K. Takai, Chem. Commun. (2008), 6360
K. Trisuwan, V. Rukachaisirikul, Y. Sukpondma, S. Preedanon, S. Phongpaichit, N. Rungjindamai, J. Sakayaroj, J. Nat. Prod. 71 1323
A.H. Aly, R. Edrada-Ebel, V. Wray, W.E.G. Muller, S. Kozytska, U. Hentschel, P. Proksch, R. Ebel, Phytochemistry 69, 1716 (2008)
E.-S. Choi, C.-G. Cho, Angew. Chem., Int. Ed. 46, 230 (2007)
S.-I. Chung, J. Seo, C.-G. Cho, J. Org. Chem. 71, 6701 (2006)
F.Bellina, A.Carpita, L.Mannocci, R.Rossi, Eur. J. Org. Chem.2610 (2004)
T. Luo, M. Dai, S.-L. Zheng, S.L. Schreiber, Org. Lett. 13, 2834 (2011)
R.C. Larock, X. Han, M.J. Doty, Tetrahedron Lett. 39, 5713 (1998)
R.C. Larock, M.J. Doty, X. Han, J. Org. Chem. 64, 8770 (1999)
S.J. Kim, H.S. Lee, J.N. Kim, Tetrahedron Lett. 48, 1069 (2007)
S. Riusset, M. Abarbri, J. Thibonnet, A.Ducheˆne, J.-L.Parrain, Chem. Commun. 1987 (2000)
K. Cherry, J.-L. Parrain, J. Thibonnet, A. Ducheˆne, M. Abarbri, J. Org. Chem. 70, 6669 (2005)
J.N. Kim, J.M. Kim, K.Y. Lee, S. Gowrisankar, Bull. Korean Chem. Soc. 25, 1733 (2004)
S.C. Kim, H.S. Lee, J.N. Kim, Bull. Korean Chem. Soc. 28, 147 (2007)
W. Zhong, Y. Zhao, W. Su, Tetrahedron 64, 5491 (2008)
S.I. Kotretsou, M.P. Georgiadis, Org. Prep. Proced. Int. 32, 161 (2000)
B.J. Stanovnik, Heterocycl. Chem. 36, 1581 (1999)
V. Kepe, S. Polanc, M. Kocevar, Heterocycles 48, 671 (1998)
R.C. Larock, M.J. Doty, X. Han, J. Org. Chem. 64, 8770 (1999)
R.C. Larock, X. Han, M.J. Doty, Tetrahedron Lett. 39, 5713 (1998)
S. Cerezo, M. Moreno-Manas, R. Pleixats, Tetrahedron 54, 7813 (1998)
T. Yao, R.C. Larock, J. Org. Chem. 68, 5936 (2003)
S. Ma, S. Yu, S. Yin, J. Org. Chem. 68, 8996 (2003)
X.-F. Zhu, A.-P. Schaffner, R.C. Li, O. Kwon, Org. Lett. 7, 2977 (2005)
F. Bellina, M. Biagetti, A. Carpita, R. Rossi, Tetrahedron. 57 2857 (2001)
F. Bellina, M. Biagetti, A. Carpita, R. Rossi, Tetrahedron Lett. 42, 2859 (2001)
S. Ma, S. Yin, L. Li, F. Tao, Org. Lett. 4, 505 (2002)
M. Biagetti, F. Bellina, A. Carpita, P. Stabile, R. Rossi, Tetrahedron 58, 5023 (2002)
M Biagetti, F Bellina, A Carpita, S Viel, L Mannina, R Rossi (2002) Eur. J. Org. Chem
M. Biagetti, F. Bellina, A. Carpita, R. Rossi, Tetrahedron Lett. 44, 607 (2003)
A.R. Hajipour, F. Rafiee, J. Iran. Chem. Soc. 6, 647 (2009)
M.J. Earle, S.P. Katdare, K.R. Seddon, Org. Lett. 6, 707 (2004)
M.J. Earle, P.B. McCormac, K.R. Seddon, Green Chem. 1, 23 (1999)
R. Vijayaraghavan, D.R. MacFarlane, J. Aust. Chem. 57, 129 (2004)
J.N. Rosa, C.A.M. Afonso, A.G. Santos, Tetrahedron 57, 4189 (2001)
Y. Chauvin, L. Mussmann, H. Olivier, Angew. Chem., Int. Ed. Engl. 34, 2698 (1995)
M.A. Klingshirn, R.D. Rogers, K.H. Shaughnessy, J. Organomet. Chem. 690, 3620 (2005)
E. Mizushima, T. Hayashi, Tanaka. Green Chem. 3, 76 (2001)
J.S. Yadav, B.V.S. Reddy, G. Baishya, K.V. Reddy, A.V. Narsaiah, Tetrahedron 61, 9541 (2005)
M. Johansson, A.A. Linden, J.-E. Baeckvall, Organomet. Chem. 690, 3614 (2005)
A. Serbanovic, L.C. Branco, M. Nunes da Ponte, C.A.M. Afonso, J. Organomet. Chem. 690, 3600 (2005)
S. Chowdhury, R. Mohan, J.L. Scott, Tetrahedron 63, 2363 (2007)
K. Gong, H.L. Wang, D. Fang, Z.L. Liu, Catal. Commun. 9, 650 (2008)
L. Chen, X.J. Huang, Y.Q. Li, M.Y. Zhou, W. Zheng, J. Monatsh. Chem. 140, 45 (2009)
B.C. Ranu, S. Banerjee, S. Roy, Indian J. Chem. 47B, 1108 (2008)
J.-T. Li, Y. Yin, L. Li, M.-XA. Sun. Ultrason. Sonochem. 17, 11 (2010)
L. Pizzuti, P.L.G. Martins, B.A. Ribeiro, F.H. Quina, E. Pinto, A.F.C. Flores, D. Venzke, C.M.P. Pereira, Ultrason. Sonochem. 17, 34 (2010)
C.-L. Ni, X.-H. Song, X.-Q. Song, R.-G. Zhong, Ultrason. Sonochem. 17, 367 (2010)
A. Bazgir, S. Ahadi, R. Ghahremanzadeh, H.R. Khavasi, P. Mirzaei, Ultrason. Sonochem. 17, 447 (2010)
Z.-H. Zhang, J.-J. Li, T.-S. Li, Ultrason. Sonochem. 15, 673 (2008)
M. Mamaghani, S. Dastmard, Ultrason. Sonochem. 16, 445 (2009)
M. Nikpassand, M. Mamaghani, F. Shirini, Kh Tabatabaeian, Ultrason. Sonochem. 17, 301 (2010)
M. Mamaghani, A. Loghmanifar, M.R. Taati, Ultrason. Sonochem. 18, 45 (2011)
W. Zhong, Y. Zhao, W. Su, Tetrahedron 64, 5491 (2008)
Acknowledgments
Financial support for this work by the research council of Islamic Azad University, Rasht Branch is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mamaghani, M., Alavi, F. An expedient and green ultrasound-promoted synthesis of fused α-pyrones from Baylis–Hillman acetates using basic ionic liquid [bdmim]OH. J IRAN CHEM SOC 12, 2161–2167 (2015). https://doi.org/10.1007/s13738-015-0693-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0693-x