Abstract
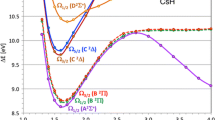
Potential energy curves (PECs) of the lowest singlet and triplet states of the HCl2+ dication have been calculated using density functional theory with the B3LYP and HSE06 functionals and coupled cluster CCSD(T) method, all with 6-311G(3df,3pd) basis set. Analysis of the results of all three methods shows that the triplet state is a pure 3Σ− state, while the singlet state is a mixture of the 1Δ and 1Σ+ states. Spectroscopic constants and Franck–Condon factors corresponding to the transitions from the ground state neutral HCl to these dicationic states have been calculated and compared to the available experimental and computational data. While the calculated spectroscopic constants and equilibrium bond lengths of the PECs obtained by the HSE06 functional are in good agreement with the available data, the predicted vertical offset of the PECs differ significantly with those observed experimentally. Inclusion of a set of floating basis functions corresponding to the Li ghost atom does not improve the results within the available experimental resolution. Addition of centrifugal term to the potential energy decreases the well depth and well width at barrier and increases the barrier height.
Graphical Abstract



Similar content being viewed by others
References
H. Sabzyan, E. Keshavarz, Z. Noorisafa, J Iran Chem Soc 11, 871 (2014)
V. Brites, K. Franzreb, J.N. Harvey, S.G. Sayres, M.W. Ross, D.E. Blumling, A.W. Castleman Jr, M. Hochlaf, Phys Chem Chem Phys 13, 15233 (2011)
D. Schröder, H. Schwarz, J Phys Chem A 103, 7385 (1999)
S. Svensson, L. Karlsson, P. Baltzer, M.P. Keane, B. Wannberg, Phys Rev A 40, 4369 (1989)
R. Thorburn, Proc Phys Soc 73, 122 (1959)
F.H. Dorman, J.D. Morrison, J Chem Phys 35, 575 (1961)
D.M. Curtis, J.H.D. Eland, Int J Mass Spectrom Ion Process 63, 241 (1985)
B.J. Olsson, M. Larsson, J Phys B At Mol Phys 20, L137 (1987)
P.G. Fournier, M. Mousselmal, S.D. Peyerimhoff, A. Banichevich, M.Y. Adam, T.J. Morgan, Phys Rev A 36, 2594 (1987)
A. Banichevich, S.D. Peyerimhoff, M.C. Van Hemert, P.G. Fournier, Chem Phys 121, 351 (1988)
F.R. Bennet, I.R. McNab, Chem Phys Lett 251, 405 (1996)
L. Streit, F.B.C. Machado, R. Custodio, Chem Phys Lett 506, 22 (2011)
P. Candori, S. Falcinelli, F. Pirani, F. Tarantelli, F. Vecchiocattivi, Chem Phys Lett 436, 322 (2007)
K. Ellingsen, T. Matila, T. Saue, H. Aksela, O. Gropen, Phys Rev A 62, 032502 (2000)
D. Ghosh, N.C. Bera, A.K. Das, Chem Phys Lett 461, 348 (2008)
P.M.W. Gill, L. Radom, Chem Phys Lett 147, 213 (1988)
Z.H. Zhu, F.H. Wang, B. Chen, M.L. Tan, H.Y. Wang, Mol Phys 92, 1061 (1997)
Z. Huang, Z.H. Zhu, J Mol Struct 525, 123 (2000)
M.M. Teixidor, F. Pirani, P. Candori, S. Falcinelli, F. Vecchiocattivi, Chem Phys Lett 379, 139 (2003)
P.D. Chong, J Chem Phys 128, 084112 (2008)
F.R. Bennet, A.D. Critchley, G.C. King, R.J. Leroy, I.R. McNab, Mol Phys 97, 35 (1999)
A.G. McConkey, G. Dawber, L. Avaldi, M.A. MacDonald, G.C. King, R.I. Hall, J Phys B At Mol Opt Phys 27, 271 (1994)
A.J. Yencha, G.C. King, M.C.A. Lopes, J.D. Bozek, N. Berrah, Chem Phys Lett 315, 37 (1999)
A.D.J. Critchley, G.C. King, P. Kreynin, M.C.A. Lopes, I.R. McNab, A.J. Yencha, Chem Phys Lett 349, 79 (2001)
T.M. Henderson, A.F. Izmaylov, G. Scalmani, G.E. Scuseria, J Chem Phys 131, 044108 (2009)
M.J. Frisch, G.W. Trucks, H.B. Schlegel, G.E. Scuseria, M.A. Robb, J.R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G.A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H.P. Hratchian, A.F. Izmaylov, J. Bloino, G. Zheng, J.L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajiam, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J.A. Montgomery Jr., J.E. Peralta, F. Ogliaro, M. Bearpark, J.J. Heyd, E. Brothers, K.N. Kudin, V.N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J.C. Burant, S.S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J.M. Millam, M. Klene, J.E. Knox, J.B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R.E. Stratmann, O. Yazyev, A.J. Austin, R. Cammi, C. Pomelli, J.W. Ochterski, R.L. Martin, K. Morokuma, V.G. Zakrzewski, G.A. Voth, P. Salvador, J.J. Dannenberg, S. Dapprich, A.D. Daniels, O. Farkas, J.B. Foresman, J.V. Ortiz, J. Cioslowski, D.J. Fox, Gaussian, Inc. (Wallingford, CT, 2009)
R.J. LeRoy, LEVEL, University of Waterloo Chemical Physics Research Report, CP–555R (1996)
G. Dawber, A.G. McConkey, L. Avaldi, M.A. MacDonald, G.C. King, R.I. Hall, J Phys B At Mol Opt Phys 27, 2191 (1994)
Acknowledgments
We would like to thank University of Isfahan for research facilities and Ministry of Science, Research and Technology of I. R. Iran for scholarships granted to E. K. and Z. N.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sabzyan, H., Keshavarz, E. & Noorisafa, Z. Evaluation of the B3LYP and HSE06 density functionals in the calculation of spectroscopic properties of the HCl2+ dication. J IRAN CHEM SOC 12, 581–586 (2015). https://doi.org/10.1007/s13738-014-0515-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-014-0515-6