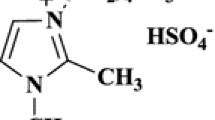
Abstract
Convenient and environmentally benign procedures have been reported for the synthesis of 1,4,5-trisubstituted-1,2,3-triazoles by the reaction of aryl azides with active methylene compounds in ionic liquid [bmim]BF4 in the presence of l-proline as a catalyst and also in task-specific basic ionic liquid [bmim]OH. The methodologies defined herein avoid the severe conditions as posed by earlier existing methods and proved to be efficient in terms of good yields, operational simplicity, easy workup and short reaction time.



Similar content being viewed by others
References
X.M. Chen, Z.J. Li, Z.X. Ren, Z.T. Huang, Carbohydr. Res. 315, 262 (1999)
M.J. Genin, D.A. Allwine, D.J. Anderson, M.R. Barbachyn, D.E. Emmert, S.A. Garmon, D.R. Graber, K.C. Grega, J.B. Hester, D.K. Hutchinson, J. Morris, R.D. Reischer, D. Stper, B.H. Yagi, J. Med. Chem. 43, 953 (2000)
R. Alvarez, S. Velazquez, A. San-Felix, S. Aquaro, E. De Clerq, C.F. Perno, A. Karlsson, J. Balzarini, M.J. Camarasa, J. Med. Chem. 37, 4185 (1994)
S. Velazquez, R. Alvarez, C. Perez, F. Gago, C. De, J. Balzarani, M.J. Camarasa, J Antivir. Chem. Chemother. 9, 481 (1998)
P.K. Kadaba, J. Med. Chem. 31, 196 (1988)
D.R. Buckle, C.J.M. Rockell, H. Smith, B.A. Spicer, J. Med. Chem. 29, 2262 (1986)
H. Wamhoff, A.R. Katritzky, C.W. Rees, E.F.V. Scriven (eds.), Comprehensive Heterocyclic Chemistry, vol. 4 (Elsevier Science, Oxford, 1996)
R. Huisgen, in 1,3 Dipolar Cycloaddition Chemistry, Chap 1, ed. by A. Padwa (Wiley, New York, 1984)
F. Xie, K. Sivakumar, Q. Zeng, M.A. Bruckman, B. Hodges, Q. Wang, Tetrahedron 64, 2906 (2008)
K. Sivakumar, F. Xie, B. Cash, S. Long, H.N. Barnhill, Q. Wang, Org. Lett. 6, 4603 (2004)
P. Wu, A.K. Feldman, A.K. Nugent, C.J. Hawker, A. Scheel, B. Voit, J. Pyun, J.M.J. Fréchet, K.B. Sharpless, V.V. Fokin, Angew. Chem. 116, 4018 (2004)
P. Wu, A.K. Feldman, A.K. Nugent, C.J. Hawker, A. Scheel, B. Voit, J. Pyun, J.M.J. Fréchet, K.B. Sharpless, V.V. Fokin, Angew. Chem. Int. Ed. 43, 3928 (2004)
C.W. Tornøe, C. Christensen, M. Meldal, J. Org. Chem. 67, 3057 (2002)
V.V. Rostovtsev, L.G. Green, V.V. Fokin, K.B. Sharpless, Angew. Chem. 114, 2708 (2002)
V.V. Rostovtsev, L.G. Green, V.V. Fokin, K.B. Sharpless, Angew. Chem. Int. Ed. 41, 2596 (2002)
A. Johnson, J.M. Baskin, C.R. Bertozzi, J.F. Koberstein, N.J. Turro, Chem. Commun. 3064 (2008)
J.A. Codelli, J.M. Baskin, N.J. Agard, C.R. Bertozzi, J. Am. Chem. Soc. 130, 11486 (2008)
M.F. Debets, C.W.J. van der Doelen, F.P.J.T. Rutjes, F.L. van Delft, ChemBioChem 11, 1168 (2010)
B.H.M. Kuijpers, S. Groothuys, A.R. Keereweer, P.J.L.M. Quaedflieg, R.H. Blaauw, F.L. van Delft, F.P.J.T. Rutjes, Org. Lett. 6, 3123 (2004)
J.E. Hein, J.C. Tripp, L.B. Krasnova, K.B. Sharpless, V.V. Fokin, Angew. Chem. 121, 8162(2009)
J.E. Hein, J.C. Tripp, L.B. Krasnova, K.B. Sharpless, V.V. Fokin, Angew Chem. Int. Ed. 48, 8018 (2009)
Q. Wang, T.R. Chan, R. Hilgraf, V.V. Fokin, K.B. Sharpless, M.G. Finn, J. Am. Chem. Soc. 125, 3192 (2003)
J. Gierlich, G.A. Burley, P.M.E. Gramlich, D.M. Hammond, T. Carell, Org. Lett. 8, 3639 (2006)
L.J.T. Danence, Y. Gao, M. Li, Y. Huang, J. Wang, Chem. Eur. J. 17, 3584 (2011)
Y.A. Rozin, J. Leban, W. Dehaen, V.G. Nenajdenko, V.M. Muzalevskiy, O.S. Eltsov, V.A. Bakulev, Tetrahedron 68, 614 (2012)
F. Stazi, D. Cancongi, L. Turco, P. Westerduin, S. Bacchi, Tetrahedron Lett. 51, 5385 (2010)
T. Welton, Chem. Rev. 99, 2071 (1999)
P. Wasserscheid, W. Keim, Angew. Chem. Int. Ed. 39, 3772 (2000)
R. J. Sheldon, Chem. Soc. Chem. Commun. 2399 (2001)
J. Peng, Y. Deng, Tetrahedron Lett. 42, 917 (2001)
N.D. Obushak, N.T. Pokhodylo, N.I. Pidlypnyi, V.S. Matiichuk, Russ. J. Org. Chem. 44, 1522 (2008)
V.R. Kamalraj, S. Senthil, P. Kannan, J. Mol. Struct. 892, 210 (2008)
S. Zeghada, G.B. Ababsa, A. Derdour, S. Adedlmouim, L.R. Domongo, J.A. Saez, T. Rosnel, E. Nasser, F. Margin, Org. Biomol. Chem. 9, 4295 (2011)
Acknowledgments
HS and JS thank UGC, New Delhi, India for the Grant of Junior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, H., Sindhu, J. & Khurana, J.M. Efficient, green and regioselective synthesis of 1,4,5-trisubstituted-1,2,3-triazoles in ionic liquid [bmim]BF4 and in task-specific basic ionic liquid [bmim]OH. J IRAN CHEM SOC 10, 883–888 (2013). https://doi.org/10.1007/s13738-013-0224-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-013-0224-6