Abstract
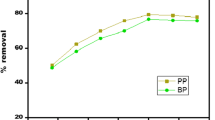
In this study, magnetite nanoparticles-modified low-cost activated carbon (MMAC) was used as an adsorbent for the removal of the anionic and cationic dyes Congo red, reactive blue 19, thionine, janus green B, methylthymol blue and mordant Blue 29, from aqueous solutions. Batch studies were performed to address various experimental parameters like pH, contact time, temperature and adsorbent dosage for the removal of these dyes. Kinetic studies showed that the adsorption of dyes on MMAC was a rapid process. Quasi equilibrium was reached in 30 min. Pseudo-first order and pseudo-second order models were used to fit the experimental data. Pseudo-second order rate equation was able to provide realistic description of adsorption kinetics. Equilibrium isotherms were analyzed by Freundlich, Langmuir, Redliche–Peterson (R–P) and Sips equations using correlation coefficients and RMS error function. Sips equation is found to best represent the equilibrium data for dye–MMAC system. It was clear that affinity of synthesized adsorbent to cationic dyes is much higher than anionic dyes.







Similar content being viewed by others
References
V.K. Gupta, L. Suhas, J. Environ. Manag. 90, 2313 (2009)
Q. Husain, Crit. Rev. Biotechnol. 26, 201 (2006)
F.I. Hai, K. Yamamoto, K. Fukushi, Crit. Rev. Environ. Sci. Technol. 37, 315 (2007)
J.S. Bae, H.S. Freeman, Dyes Pigments 73, 81 (2007)
R.M. Christie, Environmental Aspects of Textile Dyeing (Woodhead, Boca Raton, 2007)
K.L. Hatch, H.I. Maibach, Text. Chem. Color. 1, 53 (1999)
H.S. Rai, M.S. Bhattacharyya, J. Singh, T.K. Bansal, P. Vats, U.C. Banerjee, Crit. Rev. Environ. Sci. Technol. 35, 219 (2005)
T. Robinson, B. Chandran, P. Nigam, Water Res. 36, 2824 (2002)
R.C. Bansal, M. Goyal, Activated Carbon Adsorption (Taylor & Francis Group, Boca Raton, 2005)
A. Afkhami, M. Tehrani, H. Bagheri, Desalination 263, 240 (2010)
A. Afkhami, R. Moosavi, J. Hazard. Mater. 174, 398 (2010)
K. Imamura, E. Ikeda, T. Nagayasu, T. Sakiyama, K. Nakanishi, J. Colloid Interface Sci. 245, 50 (2002)
B.G. Linsen, Physical and Chemical Aspects of Adsorbents and Catalysts (Academic Press, London, 1970)
C. Tien, Adsorption Calculations and Modeling (Butterworth-Heinemann, Boston, 1994)
Y.H. Huang, C.L. Hsueh, C.P. Huang, L.C. Su, C.Y. Chen, Sep. Purif. Technol. 55, 23 (2007)
F. Alexander, G. McKay, Kinetics of removal of basic dye from effluent using silica gel. Chem. Eng. London, 243 (1977)
X. Yuan, S.P. Zhuo, W. Xing, H.Y. Cui, X.D. Dai, X.M. Liu, Z.F. Yan, J. Colloid Interface Sci. 310, 83 (2007)
M.A. Lillo-Rodenas, J.P. Marco-Lozar, D. Cazorla-Amoros, A. Linares-Solano, J. Anal. Appl. Pyrolysis 80, 166 (2007)
T. Madrakian, A. Afkhami, M.A. Zolfigol, M. Ahmadi, N. Koukabi, Nano-Micro Lett. 4, 57 (2012)
P.J.M. Carrott, M.M.L. Ribeiro Carrott, R.A. Roberts, Colloids Surf. 58, 385 (1991)
Y. Al-Degs, M.A.M. Khraisheh, S.J. Allen, M.N.A. Ahmad, Sep. Sci. Technol. 36, 91 (2001)
F.A. Di Giano, A.S. Natter, J. Water Poll. Control Fed. 49, 235 (1977)
C. Pelekani, V.L. Snoeyink, Carbon 38, 1423 (2000)
J.J. Porter, Water Wastes Eng. 9, A8 (1972)
G. McKay, J. Chem. Technol. Biotechnol. 32, 759 (1982)
G.M. Walker, L.R. Weatherley, Water Res. 33, 1895 (1999)
Y. Al-Degs, M.A.M. Khraisheh, S.J. Allen, M.N. Ahmad, Water Res. 34, 927 (2000)
T. Madrakian, A. Afkhami, M. Ahmadi, H. Bagheri, J. Hazard. Mater. 196, 109 (2011)
P. Panneerselvam, N. Morad, K.A. Tan, J. Hazard. Mater. 186, 160 (2011)
S. Lagergren, Kungliga Svenska Vetenkapsaka demiens. Handlingar 24, 1 (1898)
Y.S. Ho, G. Mckay, Process Biochem. 34, 451 (1999)
H.M.F. Freundlich, Z. Phys. Chem. 57, 385 (1906)
I. Langmuir, J. Am. Chem. Soc. 38, 2221 (1916)
O. Redlich, D.L. Peterson, J. Phys. Chem. 63, 1024 (1959)
R. Sips, On the structure of a catalyst surface. J. Chem. Phys. 16, 490 (1948)
L.C.A. Oliveira, R.V.R.A. Rios, J.D. Fabris, K. Sapag, V.K. Gargc, R.M. Lago, Appl. Clay Sci. 22, 169 (2003)
G. Jegadeesan, K. Mondal, S.B. Lalvani, Environ. Prog. 24, 289 (2005)
S. Dawood, T.K. Sen, Water Res. 46, 1933 (2012)
Y. Yu, Y.Y. Zhuang, Z.H. Wang, J. Colloid Interface Sci. 242, 288 (2001)
H.Y. Zhu, Y.Q. Fu, R. Jiang, J.H. Jiang, L. Xiao, G.M. Zeng, S.L. Zhao, Y. Wang, Chem. Eng. J. 173, 494 (2011)
R. Ahmad, R. Kumar, Appl. Surface Sci. 257, 1628 (2010)
M. Ghaedi, M. Nejati Biyareh, S. Nasiri Kokhdan, S. Shamsaldini, R. Sahraei, A. Daneshfar, S. Shahriyar, Materials Sci. Eng. C 32, 725 (2012)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Madrakian, T., Afkhami, A., Mahmood-Kashani, H. et al. Adsorption of some cationic and anionic dyes on magnetite nanoparticles-modified activated carbon from aqueous solutions: equilibrium and kinetics study. J IRAN CHEM SOC 10, 481–489 (2013). https://doi.org/10.1007/s13738-012-0182-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-012-0182-4