Abstract
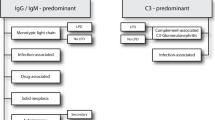
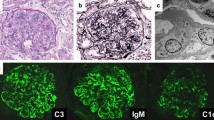
Glomerulopathy associated with shunt infection is commonly membranoproliferative glomerulonephritis, whereas the causative organisms of secondary membranous nephropathy are usually viruses. We report a case of membranous nephropathy associated with shunt infection. The patient was born at 29-week gestation with a birth weight of 1178 g. Ventriculoperitoneal shunt surgery had been performed for congenital hydrocephalus. Thereafter, she had experienced seven shunt infections. At the age 13 years, proteinuria was detected in a school urinary screening. Urinalysis at our hospital demonstrated 3 + protein and 3 + blood. Laboratory testing demonstrated a serum creatinine 0.5 m/dl, albumin 2.5 g/dl, C-reactive protein (CRP) 13.7 mg/dl, and C3 182 mg/dl. Prior to repeat urinalysis, the patient developed vomiting and was admitted with suspected shunt infection. On admission, her body temperature was 36.0 ºC. Physical examination was unremarkable other than small stature and a palpable mass in the left upper quadrant. Urinalysis demonstrated 2 + protein and 1 + blood with no cells or casts. The urinary protein excretion was 3 g/day. Abnormal laboratory tests included erythrocyte sedimentation rate 102 mm/hr, CRP 11.67 mg/dl, IgG 2442 mg/dl, C3 177 mg/dl, and C4 44 mg/dl. Antibiotic therapy was initiated for a presumptive diagnosis of shunt infection and the shunt catheter was removed. Cultures obtained after antibiotic administration were negative. Proteinuria persisted after control of the shunt infection. Histology of a renal biopsy demonstrated membranous nephropathy with diffuse granular IgG staining and subepithelial deposits. Three possible pathomechanisms for her membranous nephropathy were considered.

Similar content being viewed by others
References
Iida H, Mizumura Y, Uraoka T, et al. Membranous glomerulonephritis associated with enterococcal endocarditis. Nephron. 1985;40:88. https://doi.org/10.1159/000183435.
Bulucu F, Can C, Oktenli C, et al. Membranous glomerulonephritis, antiphospholipid syndrome, and persistent low C3 levels associated with meningococcal disease. Nephron. 2002;91:336–8. https://doi.org/10.1159/000058415.
Levy Y, George J, Ziporen L, et al. Massive proteinuria as a main manifestation of primary antiphospholipid syndrome. Pathobiology. 1998;66:49–52. https://doi.org/10.1159/000027995.
Richet G, Fillastre JP, Morel-Maroger L, et al. Change from diffuse proliferative to membranous glomerulonephritis: serial biopsies in four cases. Kidney Int. 1974;5:57–71. https://doi.org/10.1038/ki.1974.57-71.
Kapur S, Salcedo J, Chandra R, et al. Evolution of membranous nephropathy from a proliferative and exudative glomerulonephritis–a report of three cases studied by serial biopsies. Int J Pediatr Nephrol. 1985;6:105–10.
Sotsiou F, Dimitriadis G, Liapis H. Diagnostic dilemmas in atypical postinfectious glomerulonephritis. Semin Diagn Pathol. 2002;19:146–59.
Sotsiou F. Postinfectious glomerulonephritis. Nephrol Dial Transplant. 2001;16:68. https://doi.org/10.1093/ndt/16.suppl_6.68.
Patel C, Chaudhuri NR, Gaur S. Group A streptococcus ventriculoperitoneal shunt infection in a child. Pediatr Infect Dis J. 2012;31:660. https://doi.org/10.1097/INF.0b013e31824c04a5.
Luyckx VA, Brenner BM. The clinical importance of nephron mass [Review]. J Am Soc Nephrol. 2010;21:898–910. https://doi.org/10.1681/ASN.2009121248.
Takizawa K, Miura K, Kaneko N, et al. Renal hypoplasia can be the cause of membranous nephropathy-like lesions. Clin Exp Nephrol. 2020;24:813–20. https://doi.org/10.1007/s10157-020-01902-y.
Watanabe T. Membranous glomerulonephritis in a patient with unilateral renal agenesis. Nephron. 2002;91:159–61. https://doi.org/10.1159/000057619.
Miller P, Lei L, Charu V, et al. Clinicopathologic features of non-lupus membranous nephropathy in a pediatric population. Pediatr Nephrol. 2022;25:1–1.
Menon S, Valentini RP. Membranous nephropathy in children: clinical presentation and therapeutic approach. Pediatr Nephrol. 2010;25:1419–28. https://doi.org/10.1007/s00467-009-1324-5.
Xie Q, Li Y, Xue J, et al. Renal phospholipase A2 receptor in hepatitis B virus-associated membranous nephropathy. Am J Nephrol. 2015;41:345–53. https://doi.org/10.1159/000431331.
Sethi S, Madden BJ, Debiec H, et al. Exostosin 1/Exostosin 2-associated membranous nephropathy. J Am Soc Nephrol. 2019;30:1123–36. https://doi.org/10.1681/ASN.2018080852.
Caza TN, Hassen SI, Dvanajscak Z, et al. NELL1 is a target antigen in malignancy-associated membranous nephropathy. Kidney Int. 2021;99:967–76. https://doi.org/10.1016/j.kint.2020.07.039.
Funding
None.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Consent for publication
Informed consent was obtained from the parents.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Awazu, M., Miyahara, M., Chiga, M. et al. A girl with membranous nephropathy associated with ventriculoperitoneal shunt infection. CEN Case Rep 12, 130–134 (2023). https://doi.org/10.1007/s13730-022-00732-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13730-022-00732-z