Abstract
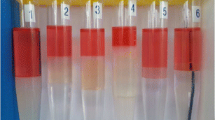
Foams show poor stability in enhanced oil recovery applications and stimulation processes in oil-well operations of oil fields. This paper presents a laboratory study to investigate the effect of Newtonian and non-Newtonian viscosity enhancement materials on improving the stability of foams at bulk conditions. For this goal, glycerol and hydrolyzed polyacrylamide (HPAM) were utilized to enhance the viscosity of foaming agents, which were composed of α-olefin sulfonate surfactant and salinity. To this end, a comparative study of the foam stability in surfactant solution containing different percentages of glycerol, HPAM polymer and a mixture of polymer with glycerol was undertaken. In a foam stability analysis, which was examined in the absence of an oleic phase, several characteristics such as foam volume evolution, foam half-decay time and a liquid fraction of foam were measured over a wide range of concentrations. Evaluating the conductivity and volume of injected gas during foam generation and foam decay provided the foam capacity and the maximum density parameters to characterize the foamability and stability of the generated foam in more detail. The results of bulk foam experiments indicated that polymer and glycerol could either increase or reduce the foamability, but both materials substantially increased foam stability within a certain range of concentrations. This could be explained by increasing the viscosity of the liquid phase of foam in the lamellae that attributed to decreased velocity of liquid drainage from the foam structure. Two regimes of foam drainage and coalescence demonstrated a different behavior for the same viscosity of solutions containing either glycerol or HPAM polymer. The solutions containing glycerol exhibited a small but sharp decay right after stopping the sparging gas, while for high polymer concentrations this did not happen.












Similar content being viewed by others
References
Lake LW, Russell J, Rossen B (2014) Fundamentals of enhanced oil recovery. Society of Petroleum Engineers, Richardson, Texas
Cottin C, Morel DC, Levitt D, Cordelier PR, Pope GA (2012) Alkali surfactant gas injection: attractive laboratory results under the Harsh salinity and temperature conditions of Middle East carbonates. In: Abu Dhabi international petroleum conference and exhibition, UAE, 11–14 November 2012
Li RF, Yan W, Liu S, Hirasaki GJ, Miller CA (2010) Foam mobility control for surfactant enhanced oil recovery. SPE J 15:20–23
Rossen WR (1996) Foams in enhanced oil recovery. In: Prud’homme RK, Khan S (eds) Foams: theory, measurements and applications. Marcel Dekker, New York
Patzek TW (1985) Description of foam flow in porous media by the population balance method. ACS Symp Ser 373:326–341
Tanzil D, Hirasaki GJ, Miller CA (2002) Conditions for foam generation in homogeneous porous media. In: SPE/DOE improved oil recovery symposium, 13–17 April 2002, Tulsa, OK, USA
Rossen WR, Zeilinger SC, Shi J, Lim MT (1999) Simplified mechanistic simulation of foam processes in porous media. Soc Petrol Eng J 4:279–287
Hosseini-Nasab SM, Zitha PLJ (2017) Investigation of chemical-foam design as a novel approach towards immiscible foam flooding for enhanced oil recovery. Energy Fuel 31:10525–10534
Hutzler S, Weaire D (2001) The Physics of foams. Clarendon, Oxford
Sonn JS, Lee JY, Jo SH, Yoon IH, Jung CH (2018) Effect of surface modification of silica nanoparticles by silane coupling agent on decontamination foam stability. Ann Nucl Energy 114:11–18
Hosseini-Nasab SM, Zitha PLJ (2017) Investigation of certain physical–chemical features of oil recovery by an optimized alkali–surfactant–foam (ASF) system. Colloid Polym Sci 295:1873–1886
Nikolov AD, Wasan DT, Huang DW, Edwards DA (2013) The effect of oil on foam stability: mechanisms and implications for oil displacement by foam in porous media. In: SPE annual technical conference and exhibition, New Orleans, Louisiana, 5–8 October 2013
Simjoo M, Rezaei T, Andrianov A, Zitha PLJ (2013) Foam stability in the presence of oil: effect of surfactant concentration and oil type. Colloid Surf A Physicochem Eng Asp 438:148–158
Carey E, Stubenrauch C (2009) Properties of aqueous foams stabilized by dodecyltrimethylammonium bromide. J Colloid Interf Sci 333:619–627
Rio E, Drenckhan W, Salonen A, Langevin D (2014) Unusually stable liquid foams. Adv Colloid Interf Sci 205:74–86
Narsimhan G, Ruckenstein E (1996) Structure, drainage, and coalescence of foams and concentrated emulsions. Foams: theory, measurements, and applications. Taylor & Francis, Milton Park, p. 89
Farajzadeh R, Muruganathan RM, Rossen WR, Krastev R (2011) Effect of gas type on foam film permeability and its implications for foam flow in porous media. Adv Colloid Interf Sci 168:71–78
Craster RV, Matar OK (2009) Dynamics and stability of thin liquid films. Rev Mod Phys 81:1131–1198
Worthen AJ, Bagaria HG, Chen Y, Bryant LS, Huh C (2013) Nanoparticle-stabilized carbon dioxide-in-water foams with fine texture. J Colloid Interf Sci 391:142–151
Petkova R, Tcholakova S, Denkov ND (2012) Foaming and foam stability for mixed polymer-surfactant solutions: effects of surfactant type and polymer charge. Langmuir 28:4996–5009
Campbell RA, Yanez Arteta M, Angus-Smyth A, Nylander T, Varga I (2011) Effects of bulk colloidal stability on adsorption layers of poly(diallyldimethylammonium chloride)/sodium dodecyl sulfate at the air-water interface studied by neutron reflectometry. J Phys Chem B 115:15202–15213
Karakashev SI, Georgiev P, Balashev K (2012) Foam production -ratio between foaminess and rate of foam decay. J Colloid Interf Sci 379:144–147
Stubenrauch C, Klitzing R von (2003) Disjoining pressure in thin liquid foam and emulsion films—new concepts and perspectives. J Phys Condens Matter 15:R1197–R1232
Gauchet S, Durand M, Langevin D (2014) Foam drainage. Possible influence of a non-Newtonian surface shear viscosity. J Colloid Interf Sci 449:373–376
Kapetas L, Vincent Bonnieu S, Danelis S, Rossen WR, Farajzadeh R (2016) Effect of temperature on foam flow in porous media. J Ind Eng Chem 36:229–237
Pradhan MS, Sarma DSHSR, Khilar KC (1990) Stability of aqueous foams with polymer additives. J Colloid Interf Sci 139:519–526
Cervantes Martinez A, Rio E, Delon G, Saint-Jalmes A, Langevin D, Binks BP (2008) On the origin of the remarkable stability of aqueous foams stabilised by nanoparticles: link with microscopic surface properties. Soft Matter 4:1531–1535
Exerowa D, Kruglyakoc P (1998) Foam and foam films: theory, experiment, application. 1st ed., Elsevier, Amsterdam
Hanamertani AS, Pilus RM, Manan NA, Hmed S, Awang M (2018) Ionic liquid application in surfactant foam stabilization for gas mobility control. Energy Fuels 32:6545–6556
Stocco A, Drenckhan W, Rio E, Langevina D, Binks B (2009) Particle-stabilised foams: an interfacial study. Soft Matter 5:2215–2222
Fameau AL, Saint-Jalmes A (2017) Non-aqueous foams: current understanding on the formation and stability mechanisms. Adv Colloid Interf Sci 247:454–464
AlYousef ZA, Almobarky MA, Schechter DS (2018) The effect of nanoparticle aggregation on surfactant foam stability. J Colloid Interf Sci 511:365–373
Vikingstad AK, Skauge A, Høiland H, Aarra M (2005) Foam–oil interactions analyzed by static foam tests. Colloid Surf A Physicochem Eng Asp 260:189–198
Romero C, Valero EM, Alvarez JM, Romero OM (2001) Designing a mobility control foam for Western Venezuela reservoirs: experimental studies. In: SPE Latin American and Caribbean petroleum engineering conference, Buenos Aires, Argentina, 25–28 March 2001
Shen C, Nguyen Q, Huh C, Rossen WR (2006) Does Polymer Stabilize Foam in Porous Media? In: SPE/DOE symposium on improved oil recovery, Tulsa, Oklahoma, USA, 22–26 April 2006
Telmadarreie A, Trivedi JJ (2016) New insight on carbonate-heavy-oil recovery: pore-scale mechanisms of post-solvent carbon dioxide foam/polymer-enhanced-foam flooding. SPE J 21:1655–1668
Hernando L, Bertin HJ, Omari A, Dupuis G, Zaitoun A (2016) Polymer-enhanced foams for water profile control. In: SPE improved oil recovery conference, Tulsa, Oklahoma, USA, 11–13 April 2016
Wang D, Han D, Xu G, Yang L (2008) Influence of partially hydrolyzed polyacrylamide on the foam capability of α-olefin sulfonate surfactant. Pet Explor Dev 35:335–338
Srivastava A, Qiao W, Wu Y, Li X, Bao L, Liu C (2017) Effects of silica nanoparticles and polymers on foam stability with sodium dodecylbenzene sulfonate in water–liquid paraffin oil emulsions at high temperatures. J Mol Liq 241:1069–1078
Takamura K, Fischer H, Morrow NR (2012) Physical properties of aqueous glycerol solutions. J Pet Sci Eng 98–99:50–60
Tcholakova S, Mitrinova Z, Golemanov K, Denkov ND, Vethamuthu M (2011) Control of Ostwald ripening by using surfactants with high surface modulus. Langmuir 27:14807–14819
Hosseini-Nasab SM, Zitha PLJ (2015) Systematic phase behaviour study and foam stability analysis for optimal alkaline/surfactant/foam enhanced oil recovery. In: IOR 2015–18th European symposium on improved oil recovery, Dresden, Germany, 14–16 April 2015
Verma A, Chauhan G, Ojha K (2018) Characterization of α-olefin sulfonate foam in presence of cosurfactants: stability, foamability and drainage kinetic study. J Mol Liq 264:458–469
Jones SA, Laskaris G, Vincent-Bonnieu S, Farajzadeh R, Rossen WR (2016) Effect of surfactant concentration on foam: from coreflood experiments to implicit-texture foam-model parameters. J Ind Eng Chem 37:268–276
Acknowledgements
We acknowledge Shell Global Solution BV for providing chemicals and also support in performing some of experiments in the Rock & Fluid Physics Laboratory of Shell Global Solution Company at Rijswijk, The Netherlands. We would like to thank E Meivogel and J van Haagen for technical support at Dietz Laboratory of the Geoscience and Engineering Department of Delft University of Technology.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Hosseini-Nasab, S.M., Taal, M., Zitha, P.L.J. et al. Effect of Newtonian and non-Newtonian viscosifying agents on stability of foams in enhanced oil recovery. Part I: under bulk condition. Iran Polym J 28, 291–299 (2019). https://doi.org/10.1007/s13726-019-00698-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13726-019-00698-3