Abstract
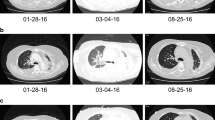
Dabrafenib plus trametinib is active against metastatic lung cancer with the BRAF V600E mutation. However, the feasibility of dabrafenib plus trametinib for patients with a poor performance status (PS) has not been reported. We report the case of an 80-year-old woman was diagnosed with metastatic large-cell lung carcinoma. Her general statuses worsened due to cancer, resulting in a PS of 4. Genotype testing revealed a BRAF V600E mutation. The patient received dabrafenib plus trametinib without significant adverse effects. This report is the first to describe dabrafenib plus trametinib administration for large-cell lung carcinoma in a patient with a poor PS.


Similar content being viewed by others
Data availability
There are no available data.
References
Bray F et al (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Planchard D et al (2016) Dabrafenib plus trametinib in patients with previously treated BRAF(V600E)-mutant metastatic non-small cell lung cancer: an open-label, multicentre phase 2 trial. Lancet Oncol 17(7):984–993
Planchard D et al (2017) Dabrafenib plus trametinib in patients with previously untreated BRAF(V600E)-mutant metastatic non-small-cell lung cancer: an open-label, phase 2 trial. Lancet Oncol 18(10):1307–1316
Inoue A et al (2009) First-line gefitinib for patients with advanced non-small-cell lung cancer harboring epidermal growth factor receptor mutations without indication for chemotherapy. J Clin Oncol 27(9):1394–1400
Iwama E et al (2017) Alectinib for patients with alk rearrangement-positive non-small cell lung cancer and a poor performance status (lung oncology group in Kyushu 1401). J Thorac Oncol 12(7):1161–1166
Nakashima K et al (2020) Osimertinib for patients with poor performance status and EGFR T790M mutation-positive advanced non-small cell lung cancer: a phase II clinical trial. Invest New Drugs 38(6):1854–1861
Paik PK et al (2011) Clinical characteristics of patients with lung adenocarcinomas harboring BRAF mutations. J Clin Oncol 29(15):2046–2051
Board, P.D.Q.A.T.E. (2002) Non-small cell lung cancer treatment (PDQ®): health professional version. In: PDQ cancer information summaries. National Cancer Institute (US), Bethesda
Flaherty KT et al (2012) Improved survival with MEK inhibition in BRAF-mutated melanoma. N Engl J Med 367(2):107–114
Society, T.J.L.C. (2021) Guidelines for diagnosis and treatment of the lung cancer/malignant pleural mesothelioma/thymic tumors 2020. Kanehara
Ettinger DS, W D, Aisner DL, et al (2021) NCCN clinical practice guidelines in oncology: non-small cell lung cancer. Version 6.2021. Accessed Sept 30
Hesketh PJ et al (2008) Southwest oncology group phase ii trial (S0341) of erlotinib (OSI-774) in patients with advanced non-small cell lung cancer and a performance status of 2. J Thorac Oncol 3(9):1026–1031
Nakamura Y et al (2020) Clinical utility of circulating tumor DNA sequencing in advanced gastrointestinal cancer: SCRUM-Japan GI-SCREEN and GOZILA studies. Nat Med 26(12):1859–1864
Acknowledgements
None declared
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the publication of this article.
Consent for publication
Consent for publication of the case was provided by the patient.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Nakajima, Y., Nishijima, Y., Niida, A. et al. Dabrafenib plus trametinib treatment for a patient with BRAF V600E-mutated large-cell lung carcinoma and a poor performance status. Int Canc Conf J 11, 266–269 (2022). https://doi.org/10.1007/s13691-022-00556-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13691-022-00556-9