Abstract
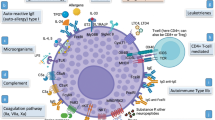
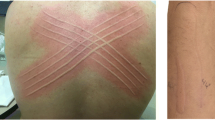
The pathomechanism of chronic spontaneous urticaria (CSU), defined by the recurrence of short-lived wheals for more than 6 weeks without apparent triggers, remained to be mysterious until 1986. In that year, skin reactivity to the injection of autologous serum was reported in about half of patients with CSU; that observation and the subsequent discovery of histamine-releasing autoantibodies against the high-affinity IgE receptor (FcεRI) and against IgE in patients with CSU were major breakthroughs in the pathogenesis of this disorder. A role for complement activation in autoantibody-dependent histamine release was subsequently demonstrated. CSU was hence considered as an autoimmune disorder due to histamine-releasing autoantibodies in a consistent proportion of cases. However, this model does not fit all cases and shows several inconsistencies. Recently, it was shown that sera from most CSU patients are able to degranulate also mast cells lacking FcεRI, suggesting the involvement of other as yet unknown mechanisms. New light on CSU pathogenesis was shed by the demonstration of coagulation activation and eosinophil involvement during active phases of the disease, paralleling urticaria severity. Finally, the efficacy of omalizumab (a recombinant humanized monoclonal IgG anti-IgE antibody) in the treatment of refractory CSU opened a new scenario in the pathophysiology of the disease. Indeed, omalizumab has multiple biological effects relevant in CSU pathogenesis, such as binding of free IgE, downregulation of mast cell function including expression of high affinity IgE receptors, and induction of eosinophil apoptosis.


Similar content being viewed by others
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Grattan CE, Wallington TB, Warin RP, Kennedy CT, Bradfield JW. A serological mediator in chronic idiopathic urticaria—a clinical, immunological and histological evaluation. Br J Dermatol. 1986;114:583–90.
Gruber B, Baeza M, Marchese M, Agnello V, Kaplan AP. Prevalence and functional role of anti-IgE autoantibodies in urticarial syndromes. J Invest Dermatol. 1988;90:213–7.
Grattan CE, Francis DM, Hide M, Greaves MW. Detection of circulating histamine releasing autoantibodies with functional properties of anti-IgE in chronic urticaria. Clin Exp Allergy. 1991;21:695–704.
Hide M, Francis DM, Grattan CEH, Hakimi J, Kochan JP, Greaves MW. Autoantibodies against the high-affinity IgE receptor as a cause of histamine release in chronic urticaria. N Engl J Med. 1993;328:1599–604.
Fiebiger E, Maurer D, Holub H, Reininger B, Hartmann G, Woisetschläger M, et al. Serum IgG autoantibodies directed against the alpha chain of Fc epsilon RI: a selective marker and pathogenetic factor for a distinct subset of chronic urticaria patients. J Clin Invest. 1995;96:2606–12.
Fiebiger E, Hammerschmid F, Stingl G, Maurer D. Anti-FcepsilonRIalpha autoantibodies in autoimmune-mediated disorders. Identification of a structure-function relationship. J Clin Invest. 1998;101:243–51.
Tong LJ, Balakrishnan G, Kochan JP, Kinét JP, Kaplan AP. Assessment of autoimmunity in patients with chronic urticaria. J Allergy Clin Immunol. 1997;99:461–5.
Ferrer M, Kinét JP, Kaplan AP. Comparative studies of functional and binding assays for IgG anti-Fc(epsilon)RIalpha (alpha-subunit) in chronic urticaria. J Allergy Clin Immunol. 1998;101:672–6.
Sabroe RA, Fiebiger E, Francis DM, Maurer D, Seed PT, Grattan CE, et al. Classification of anti-FcepsilonRI and anti-IgE autoantibodies in chronic idiopathic urticaria and correlation with disease severity. J Allergy Clin Immunol. 2002;110:492–9.
Ferrer M, Nakazawa K, Kaplan AP. Complement dependence of histamine release in chronic urticaria. J Allergy Clin Immunol. 1999;104:169–72.
Kikuchi Y, Kaplan A. A role for C5a in augmenting IgG-dependent histamine release from basophils in chronic urticaria. J Allergy Clin Immunol. 2002;109:114–8.
Soundararajan S, Kikuchi Y, Joseph K, Kaplan AP. Functional assessment of pathogenic IgG subclasses in chronic autoimmune urticaria. J Allergy Clin Immunol. 2005;115:815–21.
Niimi N, Francis DM, Kermani F, O’Donnell BF, Hide M, Kobza-Black A, et al. Dermal mast cell activation by autoantibodies against the high affinity IgE receptor in chronic urticaria. J Invest Dermatol. 1996;106:1001–6.
De Swerdt A, Van Den Keybus C, Kasran A, Cadot P, Neyens K, Coorevits L, et al. Detection of basophil-activating IgG autoantibodies in chronic idiopathic urticaria by induction of CD 63. J Allergy Clin Immunol. 2005;116:662–7.
Szegedi A, Irinyi B, Gál M, Hunyadi J, Dankó K, Kiss E, et al. Significant correlation between the CD63 assay and the histamine release assay in chronic urticaria. Br J Dermatol. 2006;155:67–75.
Frezzolini A, Provini A, Teofoli P, Pomponi D, De Pità O. Serum-induced basophil CD63 expression by means of a tricolour flow cytometric method for the in vitro diagnosis of chronic urticaria. Allergy. 2006;61:1071–7.
Yasnowsky KM, Dreskin SC, Efaw B, Schoen D, Vedanthan PK, Alam R, et al. Chronic urticaria sera increase basophil CD203c expression. J Allergy Clin Immunol. 2006;117:1430–4.
Greaves MW. Pathology and classification of urticaria. Immunol Allergy Clin North Am. 2014;34:1–9.
Leznoff A, Sussman GL. Syndrome of idiopathic chronic urticaria and angioedema with thyroid autoimmunity: a study of 90 patients. J Allergy Clin Immunol. 1989;84:66–71.
Confino-Cohen R, Chodick G, Shalev V, Leshno M, Kimhi O, Goldberg A. Chronic urticaria and autoimmunity: associations found in a large population study. J Allergy Clin Immunol. 2012;129:1307–13.
Greaves MW. Chronic urticaria. N Engl J Med. 1995;332:1767–72.
Asero R, Tedeschi A, Lorini M, Salimbeni R, Zanoletti T, Miadonna A. Chronic urticaria: novel clinical and serological aspects. Clin Exp Allergy. 2001;31:1105–10.
Fagiolo U, Kricek F, Ruf C, Peserico A, Amadori A, Cancian M. Effects of complement inactivation and IgG depletion on skin reactivity to autologous serum in chronic idiopathic urticaria. J Allergy Clin Immunol. 2000;106:567–72.
Zweiman B, Valenzano M, Atkins PC, Tanus T, Getsy JA. Characteristics of histamine-releasing activity in the sera of patients with chronic idiopathic urticaria. J Allergy Clin Immunol. 1996;98:89–98.
Greaves MW. Pathophysiology of chronic urticaria. Int Arch Allergy Immunol. 2002;127:3–9.
Stadler BM, Miescher S, Horn M, Pachlopnik J, Stadler M, Kricek F, et al. Allergic manifestations as the results of a conditional autoimmune response. Int Arch Allergy Immunol. 2001;124:411–3.
Horn MP, Pachlopnik JM, Vogel M, Dahinden M, Wurm F, Stadler BM, et al. Conditional autoimmunity mediated by human natural anti-Fc(epsilon)RIalpha autoantibodies? FASEB J. 2001;15:2268–74.
Asero R, Lorini M, Chong SU, Zuberbier T, Tedeschi A. Assessment of histamine-releasing activity of sera from patients with chronic urticaria showing positive autologous skin test on human basophils and mast cells. Clin Exp Allergy. 2004;34:1111–4.
Eckman JA, Hamilton RG, Saini SS. Independent evaluation of a commercial test for “autoimmune” urticaria in normal and chronic urticaria subjects. J Invest Dermatol. 2009;129:1584–156.
Natbony SF, Phillips ME, Elias JM, Godfrey HP, Kaplan AP. Histologic studies of chronic idiopathic urticaria. J Allergy Clin Immunol. 1983;71:177–83.
Ying S, Kikuchi Y, Meng Q, Kay AB, Kaplan AP. TH1/TH2 cytokines and inflammatory cells in skin biopsy specimens from patients with chronic idiopathic urticaria: comparison with the allergen-induced late-phase cutaneous reaction. J Allergy Clin Immunol. 2002;109:694–700.
Kasperska-Zajac A, Sztylc J, Machura E, Jop G. Plasma IL-6 concentration correlates with clinical disease activity and serum C-reactive protein concentration in chronic urticaria patients. Clin Exp Allergy. 2011;41:1386–91.
Tedeschi A, Asero R, Lorini M, Marzano AV, Cugno M. Plasma levels of matrix metalloproteinase-9 in chronic urticaria patients correlate with disease severity and C-reactive protein but not with circulating histamine-releasing factors. Clin Exp Allergy. 2010;40:875–81.
Sabroe RA, Poon E, Orchard GE, Lane D, Francis DM, Barr RM, et al. Cutaneous inflammatory cell infiltrate in chronic idiopathic urticaria: comparison of patients with and without anti-FcepsilonRI or anti-IgE autoantibodies. J Allergy Clin Immunol. 1999;103:484–93.
Asero R, Tedeschi A, Riboldi P, Cugno M. Plasma of patients with chronic urticaria shows signs of thrombin generation, and its intradermal injection causes wheal-and-flare reactions much more frequently than autologous serum. J Allergy Clin Immunol. 2006;117:1113–7.
Asero R, Tedeschi A, Coppola R, Griffini S, Paparella P, Riboldi P, et al. Activation of the tissue factor pathway of blood coagulation in patients with chronic urticaria. J Allergy Clin Immunol. 2007;119:705–10.
Asero R, Tedeschi A, Riboldi P, Griffini S, Bonanni E, Cugno M. Severe chronic urticaria is associated with elevated plasma levels of d-dimer. Allergy. 2008;63:176–80.
Cirino G, Cicala C, Bucci MR, Sorrentino L, Maranganore JM, Stone SR. Thrombin functions as an inflammatory mediator through activation of its receptors. J Exp Med. 1996;183:821–7.
Huber-Lang M, Sarma JV, Zetoune FS, Rittirsch D, Neff TA, et al. Generation of C5a in the absence of C3: a new complement activation pathway. Nat Med. 2006;12:682–7.
Cugno M, Marzano AV, Tedeschi A, Fanoni D, Venegoni L, Asero R. Expression of tissue factor by eosinophils in patients with chronic urticaria. Int Arch Allergy Clin Immunol. 2009;148:170–4.
Moosbauer C, Morgenstern E, Cuvelier SL, Manukyan D, Bidzhekov K, Albrecht S, et al. Eosinophils are a major intravascular location for tissue factor storage and exposure. Blood. 2007;109:995–1002.
Wang F, Tang H, Xu JH, Kang KF. Activation of the blood coagulation cascade is involved in patients with chronic urticaria. J Allergy Clin Immunol. 2009;123:972–3.
Takahagi S, Mihara S, Iwamoto K, Morioke S, Okabe T, Kameyoshi Y, et al. Coagulation/fibrinolysis and inflammation markers are associated with disease activity in patients with chronic urticaria. Allergy. 2010;65:649–56.
Kasperska-Zajac A, Brzoza Z. Increased d-dimer concentration in plasma of patients with severe acute urticaria. Br J Dermatol. 2009;161:1409–10.
Fujii K, Usuki A, Kan-No Y, Ohgou N. Elevation of circulating thrombin-antithrombin III complex and fibrin degradation products in urticaria. A laboratory finding unrelated to intravascular coagulopathy. J Dermatol. 2008;35:308–10.
Takeda T, Sakurai Y, Takahagi S, Kato J, Yoshida K, Yoshioka A, et al. Increase of coagulation potential in chronic spontaneous urticaria. Allergy. 2011;66:428–33. A study showing that also the intrinsic pathway of coagulation cascade is activated via factor XI and suggesting that mast cells can be stimulated through both protease activated receptors: PAR-1 by thrombin and PAR-2 by the FVIIa/FXa/TF complex.
Tedeschi A, Asero R, Marzano AV, Lorini M, Fanoni D, Berti E, et al. Plasma levels and skin-eosinophil expression of vascular endothelial growth factor (VEGF) in patients with chronic urticaria. Allergy. 2009;64:1616–22.
Puccetti A, Bason C, Simeoni ME, Tinazzi E, Beri R, Peterlana D, et al. In chronic idiopathic urticaria autoantibodies against Fc epsilonRII/CD23 induce histamine release via eosinophil activation. Clin Exp Allergy. 2005;35:1599–607.
Shakoory B, Fitzgerald SM, Lee SA, Chi DS, Krishnaswamy G. The role of human mast cell-derived cytokines in eosinophil biology. J Interferon Cytokine Res. 2004;24:271–81.
Lampinen M, Carlson M, Hakansson LD, Venge P. Cytokine-regulated accumulation of eosinophils in inflammatory disease. Allergy. 2004;59:793–805.
Tedeschi A, Asero R, Lorini M, Marzano AV, Cugno M. Serum eotaxin levels in patients with chronic spontaneous urticaria. Eur Ann Allergy Clin Immunol. 2012;44:188–92. A study confirming the involvement of eosinophils, an under-rated cell line, in the pathogenesis of chronic spontaneous urticaria.
Asero R, Cugno M, Tedeschi A. Eosinophils in chronic urticaria: supporting or leading actors? World Allergy Organ J. 2009;2:213–7.
Bossi F, Frossi B, Radillo O, Cugno M, Tedeschi A, Riboldi P, et al. Mast cells are critically involved in serum-mediated vascular leakage in chronic urticaria beyond high-affinity IgE receptor stimulation. Allergy. 2011;66:1538–45. This is the first study demonstrating that sera from spontaneous chronic urticaria patients are able to induce mast cell degranulation by a mechanism that is independent on the high-affinity IgE receptor.
Altrichter S, Peter HJ, Pisarevskaja D, Metz M, Martus P, Maurer M. IgE mediated autoallergy against thyroid peroxidase—a novel pathomechanism of chronic spontaneous urticaria? PLoS One. 2011;6(4):e14794.
Easthope S, Jarvis B. Omalizumab. Drugs. 2001;61:253–60.
Maurer M, Rosén K, Hsieh HJ, Saini S, Grattan C, Gimenéz-Arnau A, et al. Omalizumab for the treatment of chronic idiopathic or spontaneous urticaria. N Engl J Med. 2013;368:924–35. This is one of two large and well-conducted studies establishing the efficacy of omalizumab in the treatment of chronic spontaneous urticaria.
Kaplan A, Ledford D, Ashby M, Canvin J, Zazzali JL, Conner E, et al. Omalizumab in chronic idiopathic/spontaneous urticaria patients symptomatic despite standard combination therapy. J Allergy Clin Immunol. 2013;132:101–9. This is one of two large and well-conducted studies establishing the efficacy of omalizumab in the treatment of chronic spontaneous urticaria.
Beck LA, Marcotte GV, MacGlashan D, Togias A, Saini S. Omalizumab-induced reductions in mast cell Fc epsilon RI expression and function. J Allergy Clin Immunol. 2004;114:527–30.
Noga O, Hanf G, Brachmann I, Klucken AC, Kleine-Tebbe J, Rosseau S, et al. Effect of omalizumab treatment on peripheral eosinophil and T-lymphocyte function in patients with allergic asthma. J Allergy Clin Immunol. 2006;117:1493–9.
Maurer M, Altrichter S, Bieber T, Biedermann T, Bräutigam M, Seyfried S, et al. Efficacy and safety of omalizumab in patients with chronic urticaria who exhibit IgE against thyroperoxidase. J Allergy Clin Immunol. 2011;128:202–9.
Chang TW, Chen C, Lin CJ, Metz M, Church MK, Maurer M. The potential pharmacologic mechanisms of omalizumab in patients with chronic spontaneous urticaria. J Allergy Clin Immunol. 2014. doi:10.1016/j.jaci.2014.04.036.
Conflict of Interest
R Asero, A Tedeschi, AV Marzano, and M Cugno all declare no conflicts of interest.
Human and Animal Rights and Informed Consent
All studies by the authors involving animal and/or human subjects were performed after approval by the appropriate institutional review boards. When required, written informed consent was obtained from all participants.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Asero, R., Tedeschi, A., Marzano, A.V. et al. Pathomechanisms of Chronic Spontaneous Urticaria: What Is Known and Up to Date. Curr Derm Rep 3, 191–196 (2014). https://doi.org/10.1007/s13671-014-0088-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13671-014-0088-8