Abstract
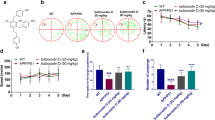
Curcumin (Curc) has been shown to have the potential to ameliorate or prevent the development of Alzheimer's disease (AD). However, most of them are in vitro and in vivo short-term studies. This study was conducted to investigate whether long-term, low-dose dietary Curc intake in mouse of AD might suppress short-term memory retention, amyloid-beta (Aβ) deposition and tau phosphorylation, delaying the onset of AD and prolonging the lifespan of the animals. Short-term memory was examined by the Y-maze method after 6 months old. Immunohistochemical analysis was performed at 10 months old to determine changes in Aβ deposition, tau phosphorylation, and glial cell number in brain tissue. Furthermore, we investigated the survival rate for 12 months old and evaluated the AD prevention effect. The alternation rates of short-time memory in the wild type and AD mice were 56.2% and 25.9%, respectively. These rates in the experimental groups (0.02% and 0.5% Curc) were in the range of 44.4–45.7%. The area of Aβ42 deposition in AD mice was approximately 25,000 µm2, while the experimental groups had a significantly reduced area of 5000–10,000 µm2. Survival rate was 34% in the AD control group, 100% in the 0.02% Curc, and 83% in the 0.5% Curc group, significantly longer in the Curc groups than the AD control group. This study demonstrates that long-term intake of low concentrations of Curc may act on the tau- phosphorylation, suppress brain inflammation, delay the onset of AD, and prolong the lifespan of the mouse.











Similar content being viewed by others
Data availability
The data are available within the article.
Abbreviations
- Curc:
-
Curcumin
- AD:
-
Alzheimer's disease
- Aβ:
-
Amyloid-beta
- WT:
-
Wild type
- NFTs:
-
Neurofibrillary tangles
- APP:
-
Amyloid precursor protein
- PS1:
-
Presenilin 1
- GSK3β:
-
Glycogen synthase kinase 3 beta
- Asp:
-
Aspirin
- PCR:
-
Polymer
- p-Tau:
-
Tau protein phosphorylation
- Iba1:
-
Ionized calcium-binding adapter molecule 1
- GFAP:
-
Glial fibrillary acidic protein
- ABC:
-
Avidin–Biotin Complex
- KLK7:
-
Kallikrein-related peptidase7
References
Alarcon R, Fuenzalida C, Santibanez M, Von Bernhardi R (2005) Expression of scavenger receptors in glial cells. Comparing the adhesion of astrocytes and microglia from neonatal rats to surface-bound β-amyloid. J Biol Chem 280:30406–30415. https://doi.org/10.1074/jbc.M414686200
Begum AN, Jones MR, Lim GP, Morihara T, Kim P, Heath DD et al (2008) Curcumin structure-function, bioavailability, and efficacy in models of neuroinflammation and Alzheimer’s disease. J Pharmacol Exp Ther 326(1):196–208. https://doi.org/10.1124/jpet.108.137455
Cameron B, Landreth GE (2010) Inflammation, microglia, and Alzheimer’s disease. Neurobiol Dis 37(3):503–509. https://doi.org/10.1016/j.nbd.2009.10.006
De Jonghe C, Esselens C, Kumar-Singh S, Craessaerts K, Serneels S, Checler F et al (2001) Pathogenic APP mutations near the gamma-secretase cleavage site differentially affect Abeta secretion and APP C-terminal fragment stability. Hum Mol Genet 10(16):1665–1671. https://doi.org/10.1093/hmg/10.16.1665
Dellu F, Contarino A, Simon H, Koob GF, Gold LH (2000) Genetic differences in response to novelty and spatial memory using a two-traial recognition task in mice. Neurobiol Learn Mem 73:31–48. https://doi.org/10.1006/nlme.1999.3919
Evans DB, Rank KB, Bhattacharya K, Thomsen DR, Gurney ME, Sharma SK (2000) Tau phosphorylation at Serine 396 and Serine 404 by human recombinant Tau protein kinase II inhibits Tau’s ability to promote microtubule assembly. J Biol Chem 275(32):24977–24983. https://doi.org/10.1074/jbc.M000808200
Fuhrmann M, Bittner T, Jung CKE, Burgold S, Page RM, Mitteregger G et al (2010) Microglial Cx3cr1 knockout prevents neuron loss in a mouse model of Alzheimer’s disease. Nat Neurosci 13(4):411–413. https://doi.org/10.1038/nn.2511
Garcia-Alloza M, Borrelli LA, Rozkalne A, Hyman BT, Bacskai BJ (2007) Curcumin labels amyloid pathology in vivo, disrupts existing plaques, and partially restores distorted neurites in an Alzheimer mouse model. J Neurochem 102(4):1095–1104. https://doi.org/10.1111/j.1471-4159.2007.04613.x
García-Matas S, de Vera N, Aznar AO, Marimon JM, Adell A, Planas AM et al (2010) In vitro and in vivo activation of astrocytes by amyloid-β is potentiated by pro-oxidant agents. J Alzheimer Dis 20(1):226–245. https://doi.org/10.3233/JAD-2010-1365
Goate A, Chartier-Harlin MC, Mullan M, Brown J, Crawford F, Fidani L et al (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349(6311):704–706. https://doi.org/10.1038/349704a0
Godemann R, Biernat J, Mandelkow E, Mandelkow E-M (1999) Phosphorylation of tau protein by recombinant GSK-3β: pronounced phosphorylation at select Ser/The-Pro motifs but no phosphorylation at Ser262 in the repeat domain. FEBS Lett 454(1–2):157–164. https://doi.org/10.1016/S0014-5793(99)00741-3
Gordon MN, Holcomb LA, Jantzen PT, DiCarlo WGD, Boyett KW et al (2002) Time course of the development of Alzheimer-like pathology in the doubly transgenic PS1+APP mouse. Exp Neurol 173(2):183–195. https://doi.org/10.1006/exnr.2001.7754
Hamaguchi T, Ono K, Murase A, Yamada M (2009) Phenolic compounds prevent Alzheimer’s pathology through different effects on the amyloid-β aggregation pathway. Am J Pathol 175(6):2557–2565. https://doi.org/10.2353/ajpath.2009.090417
Kidana K, Tatebe T, Ito K, Hama N, Kakita A, Saito T et al (2018) Loss of kallikrein-related peptidase 7 exacerbates amyloid pathology in Alzheimer’s disease model mice. EMBO Mol Med 10:e8184. https://doi.org/10.15252/emmm.201708184
Kreutzberg GW (1996) Microglia: a sensor for pathological events in the CNS. Trends Neurosci 19:312–318. https://doi.org/10.1016/0166-2236(96)10049-7
Learning and memory tests. Y Maze spontaneous alternation test. Behavioral and functional neuroscience laboratory. Stanford Med. med.stanford.edu/sbfnl/services/bm/lm.html
Li J, Han Y, Li M, Ni C (2019) Curcumin promotes proliferation of adult neural stem cells and the birth of neurons in Alzheimer’s Disease mice via notch signaling pathway. Cell Reprogram 21(3):152–161. https://doi.org/10.1089/cell.2018.0027
Liu F, Iqbal K, Grundke-Iqbal I, Gong C-X (2002) Involvement of aberrant glycosylation in phosphorylation of tau by cdk5 and GSK-3β. FEBS Lett 530(1–3):209–214. https://doi.org/10.1016/S0014-5793(02)03487-7
Lue L-F, Kuo Y-M, Beach T, Walker DG (2010) Microglia activation and anti-inflammatory regulation in Alzheimer’s disease. Mol Neurobiol 41(2–3):115–128. https://doi.org/10.1007/s12035-010-8106-8
Malumbres M, Mangues R, Ferrer N, Suying L, Pellicer A (1997) Isolation of high molecular weight DNA for reliable genotyping of transgenic mice. Biol Technol 22(6):1114–1119. https://doi.org/10.2144/97226st03
Maruyama K, Tomita T, Shinozaki K, Kume H, Asada H, Saido TC et al (1996) Familial Alzheimer’s disease-linked mutations at Val717 of amyloid precursor protein are specific for the increased secretion of Aβ 42(43). Biochem Biophys Res Commun 227(3):730–735. https://doi.org/10.1006/bbrc.1996.1577
Medeiros R, Kitazawa M, Assos GF, Baglietto-Vargas D, Cheng D, Cribbs DH et al (2013) Aspirin-triggered lipoxin A4 stimulates alternative activation of microglia and reduces alzheimer desease-link pathology in mice. Am J Pathol 182(5):1780–1789. https://doi.org/10.1016/j.ajpath.2013.01.051
Moore BD, Martin J, Mena LD, Sanchez J, Cruz PE, Ceballos-Diaz C et al (2018) Short Aβ peptides attenuate Aβ42 toxicity in vivo. J Exp Med 15(1):283–301. https://doi.org/10.1084/jem.20170600
Morales I, Guzán-Martínez L, Cerda-Troncoso C, Farías GA, Maccioni R (2014) Neuroinflammation in the pathogenesis of Alzheimer’s disease. A rational framework for the search of novel therapeutic approaches. Front Cell Neurosci 8:112–125. https://doi.org/10.3389/fncel.2014.00112
Nagele RG, D’Andrea MR, Lee H, Venkataraman V, Wang HY (2003) Astrocytes accumulate Aβ 42 and give rise to astrocytic amyloid plaques in Alzheimer disease brains. Brain Res 971(2):197–209. https://doi.org/10.1016/S0006-8993(03)02361-8
Necula M, Kayed R, Milton S, Glabe CG (2007) Small molecule inhibitors of aggregation indicate that amyloid beta oligomerization and fibrillization pathways are independent and distinct. J Biol Chem 282(14):10311–10324. https://doi.org/10.1074/jbc.M608207200
Nielsen HM, Veerhuis R, Holmqvist B, Janciauskiene S (2009) Binding and uptake of Aβ1–42 by primary human astrocytes in vitro. Glia 57:978–988. https://doi.org/10.1002/glia.20822
Oakley H, Cole SL, Logan S, Maus E, Shao P, Craft J et al (2006) Intraneuronal ß-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J Neurosci 26(40):10129–10140. https://doi.org/10.1523/JNEUROSCI.1202-06
Park S-Y, Kim H-S, Cho E-K, Kwon B-Y, Phark S, Hwang K-W et al (2008) Curcumin protected PC12 cells against beta-amyloid-induced toxicity through the inhibition of oxidative damage and tau hyperphosphorylation. Food Chem Toxicol 46(8):2881–2887. https://doi.org/10.1016/j.fct.2008.05.030
Quintela-Lòpez T, Ortiz-Sanz C, Serrano-Regal MP, Gaminde-Blasco A, Valero J, Baleriola J et al (2019) Aβ oligomers promote oligodendrocyte differentiation and maturation via integrin β1 and Fyn kinase signaling. Cell Death Dis 10:445–461. https://doi.org/10.1038/s41419-019-1636-8
Rodriguez-Vieitez E, Ni R, Gulyas B, Toth M, Haggkvist J, Halldin C et al (2015) Astrocytosis precedes amyloid plaque deposition in Alzheimer APPswe transgenic mouse brain: a correlative positron emission tomography and in vitro imaging study. Eur J Nucl Med Mol Imaging 42:1119–1132. https://doi.org/10.1007/s00259-015-3047-0
Roth AD, Ramírez F, Alarcón R, Bernhardi RV (2005) Oligodendrocyte damage in alzheimer’s disease: Beta amyloid toxicity and inflammation. Biol Res 38:381–387. https://doi.org/10.4067/S0716-97602005000400011
Rudrabhatla P, Jaffe H, Pant HC (2011) Direct evidence of phosphorylated neuronal intermediate filament proteins in neurofibrillary tangles (NFTs): phosphoproteomics of Alzheimer’s NFTs. FASEB J 25(11):3896–3905. https://doi.org/10.1096/fj.11-181297
Saito T, Suemoto T, Brouwers N, Sleegers K, Funamoto S, Mihira N et al (2011) Potent amyloidogenicity and pathogenicity of Aβ43. Nat Neurosci 14:1023–1032. https://doi.org/10.1038/nn.2858
Sarter M, Bodewitz G, Stephens DN (1988) Attenuation of scopolamine-induced impairment of spontaneous alternation behavior by antagonist but not inverse agonist and agonist β-carbolines. Psychopharmacology 94:491–495. https://doi.org/10.1007/BF00212843
Sherrington R, Rogaev EI, Liang Y, Rogaeva EA, Levesque G, Ikeda M et al (1995) Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375(6534):754–760. https://doi.org/10.1038/375754a0
Soscia SJ, Kirby JE, Washicosky KJ, Tucker SM, Ingelsson M, Hyman B et al (2010) The Alzheimer’s disease-associated amyloid β-protein is an antimicrobial peptide. PLoS ONE 5:e9505. https://doi.org/10.1371/journal.pone.0009505.g001
Suzuki N, Cheung TT, Cai XD, Odaka A, Otvos L Jr, Eckman C (1994) An increased percentage of long amyloid beta protein secreted by familial amyloid β protein precursor (β APP717) mutants. Science 264(5163):1336–1340. https://doi.org/10.1126/science.8191290
Thapa A, Jett SD, Chi EY (2016) Curcumin attenuates amyloid-b aggregate toxicity and modulates amyloid-β aggregation pathway. ACS Chem Neurosci 7:56–68. https://doi.org/10.1021/acschemneuro.5b00214
Wood JG, Mirra SS, Pollock NJ, Binder L (1986) Neurofibrillary tangles of Alzheimer disease share antigenic determinants with the axonal microtubule-associated protein tau (tau). Proc Natl Acad Sci 83:4040–4043. https://doi.org/10.1073/pnas.83.11.4040
Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS et al (2005) Curcumin inhibits formation of amyloid β oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem 280(7):5892–5901. https://doi.org/10.1074/jbc.M404751200
Zhang HR, Peng JH, Cheng XB, Shi BZ, Zhang MY, Xu RX (2015) Paeoniflorin atttenuates amyloidogenesis and the inflammatory responses in a transgenic mouse of alzheimer’s desease. Neurochem Res 40:1583–1592. https://doi.org/10.1007/s11064-015-1632-z
Acknowledgements
We would like to thank many graduate students for their cooperation in supporting our research. This research was financially supported by JSPS KAKENHI Grant Number 22580145, the United Faculty of Health Sciences, Kitasato University (no. 25), and the Urakami Food Culture Promotion Foundation (2012, vol.21).
Funding
We would like to thank many graduate students for their cooperation in supporting our research. This research was financially supported by JSPS KAKENHI Grant Number 22580145, the United Faculty of Health Sciences, Kitasato University (Grant Number 25), and the Urakami Food Culture Promotion Foundation (Grant Number vol.21, 2012).
Author information
Authors and Affiliations
Contributions
Conceptualization of the research studies in the article: (HM, FK, NA and TT); Data curation for entire study: (TO, FK, TK, RS and HM); Writing—review and editing: (HM, TO and TL); Data curation for the entire study: (TO and HM).
Corresponding author
Ethics declarations
Conflict of interest
Hiroko Maruyama has no conflict of interest. Takahito Ooizumi has no conflict of interest. Fumitaka Kawakami has no conflict of interest. Thet‑Thet Lwin has no conflict of interest. Hisanao Akita has no conflict of interest. Takuya Kunii has no conflict of interest. Ryota Shirai has no conflict of interest. Tohoru Takeda has no conflict of interest.
Animal studies
The experimental design was approved by the Animal Care and Use Committee of Kitasato University School of Allied Health Sciences before the start of the experiment (reference numbers 11–40).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Maruyama, H., Ooizumi, T., Kawakami, F. et al. Long-term oral administration of curcumin is effective in preventing short-term memory deterioration and prolonging lifespan in a mouse model of Alzheimer’s disease. ADV TRADIT MED (ADTM) 24, 373–385 (2024). https://doi.org/10.1007/s13596-023-00706-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-023-00706-1