Abstract
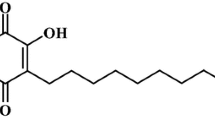
Emergence of antiviral drug resistance in influenza virus remains a major public health concern worldwide. Nowadays, different herbs receive renewed attention because of their enormous antiviral potential. In this study, we investigated the antiviral activity of Camellia sinensis, Persicaria hydropiper, Persicaria orientale, Persicaria lapathifolia, Persicaria stagnina, Mucuna pruriens and Chenopodium album against different influenza strains using both in vitro and in silico approaches. Antiviral effect of plant extracts was evaluated by cytopathic effect (CPE) inhibition assay on influenza infected MDCK (Madin Darby Canine Kidney) cell line. Later, the herb demonstrating antiviral activity was virtually screened for their available bioactive compounds and multiple in silico tools were performed to prioritize and establish these compounds as potential inhibitor. The methanol, but not the n-hexane and ethyl acetate extracts of C. sinensis, P. hydropiper, M. pruriens and C. album exhibited anti-influenza effect with EC50 values within 32–46 µg/ml. Importantly, the extracts remained effective against both amantadine-resistant and -sensitive influenza isolates. The molecular docking analysis showed that flavonoids, steroid and derivatives had strong binding affinity to the target proteins which may remain responsible for the anti-influenza characteristics of plant extracts. Pharmacokinetic properties, bioavailability and drug-likeness score revealed that ferulic acid, sinapic acid, campesterol, cryptomeridiol, eupatin and genistein could be attractive leads as potential influenza inhibitors. Taken together, the botanical ingredients of these herbs could be used as valuable candidates for developing novel therapeutics to control influenza related illnesses.




Similar content being viewed by others
References
Adebowale YA, Adeyemi A, Oshodi AA (2005) Variability in the physicochemical, nutritional and antinutritional attributes of six Mucuna species. Food Chem 89:37–48
Ahmed M, Aleem MA, Roguski K, Abedin J, Islam A, Alam KF, Gurley ES, Rahman M, Azziz-Baumgartner E, Homaira N (2018) Estimates of seasonal influenza-associated mortality in Bangladesh, 2010–2012. Influenza Other Respir Viruses 12:65–71
Amić D, Davidović-Amić D, Bešlo D, Trinajstić N (2003) Structure-radical scavenging activity relationships of flavonoids. Croat Chem Acta 76:55–61
Azziz-Baumgartner E, Alamgir ASM, Rahman M, Homaira N, Sohel BM, Sharker MA, Zaman RU, Dee J, Gurley ES, Al MA (2012) Incidence of influenza-like illness and severe acute respiratory infection during three influenza seasons in Bangladesh, 2008–2010. Bull World Health Organ 90:12–19
Barr IG, Deng YM, Iannello P, Hurt AC, Komadina N (2008) Adamantane resistance in influenza A (H1) viruses increased in 2007 in South East Asia but decreased in Australia and some other countries. Antivir Res 80:200–205
Benet LZ, Hosey CM, Ursu O, Oprea TI (2016) BDDCS, the rule of 5 and drugability. Adv Drug Deliv Rev 101:89–98
Betakova T (2007) M2 protein-a proton channel of influenza A virus. Cur Pharm Des 13:3231–3235
Bickerton GR, Paolini GV, Besnard J, Muresan S, Hopkins AL (2012) Quantifying the chemical beauty of drugs. Nat Chem 4:90–98
Boff L, Schreiber A, da Rocha MA, Del Sarto J, Brunotte L, Munkert J, Melo Ottoni F, Silva Ramos G, Kreis W, Castro Braga F (2020) Semisynthetic cardenolides acting as antiviral inhibitors of influenza A virus replication by preventing Polymerase complex formation. Molecules 25:4853
Bright RA, Medina M, Xu X, Perez-Oronoz G, Wallis TR, Davis XM, Povinelli L, Cox NJ, Klimov AI (2005) Incidence of adamantane resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: a cause for concern. Lancet 366:1175–1181
Carrat F, Flahault A (2007) Influenza vaccine: the challenge of antigenic drift. Vaccine 25:6852–6862
Chan MCW, Wang MH, Chen Z, Hui DSC, Kwok AK, Yeung ACM, Liu KM, Yeoh YK, Lee N, Chan PKS (2018) Frequent genetic mismatch between vaccine strains and circulating seasonal influenza viruses, Hong Kong, China, 1996–2012. Emerg Infect Dis 24:1825
Chen C, Qiu H, Gong J, Liu Q, Xiao H, Chen X-W, Sun B-L, Yang R-G (2012) (−)-Epigallocatechin-3-gallate inhibits the replication cycle of hepatitis C virus. Arch Virol 157:1301–1312
Cheng PKC, Leung TWC, Ho ECM, Leung PCK, Ng AYY, Lai MYY, Lim WWL (2009) Oseltamivir-and amantadine-resistant influenza viruses A (H1N1). Emerg Infect Dis 15:966
da Costa Cordeiro BMP, de Lima Santos ND, Ferreira MRA, de Araújo LCC, Junior ARC, da Conceição Santos AD, de Oliveira AP, da Silva AG, da Silva Falcão EP, dos Santos Correia MT (2018) Hexane extract from Spondias tuberosa (Anacardiaceae) leaves has antioxidant activity and is an anti-Candida agent by causing mitochondrial and lysosomal damages. BMC Complem Altern Med 18:1–10
Daina A, Michielin O, Zoete V (2017) SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep 7:1–13
Dallakyan S, Olson AJ (2015) Small-molecule library screening by docking with PyRx. Springer, Berlin, pp 243–250
Dawood FS, Iuliano AD, Reed C, Meltzer MI, Shay DK, Cheng P-Y, Bandaranayake D, Breiman RF, Brooks WA, Buchy P (2012) Estimated global mortality associated with the first 12 months of 2009 pandemic influenza A H1N1 virus circulation: a modelling study. Lancet Infect Dis 12:687–695
Deyde VM, Xu X, Bright RA, Shaw M, Smith CB, Zhang Y, Shu Y, Gubareva LV, Cox NJ, Klimov AI (2007) Surveillance of resistance to adamantanes among influenza A (H3N2) and A (H1N1) viruses isolated worldwide. J Infect Dis 196:249–257
Dutt S, Narwal S, Kapoor HC, Lodha ML (2003) Isolation and characterization of two protein isoforms with antiviral activity from Chenopodium album L leaves. J Plant Biochem Biotechnol 12:117–122
Enkhtaivan G, John KMM, Ayyanar M, Sekar T, Jin K-J, Kim DH (2015) Anti-influenza (H1N1) potential of leaf and stem bark extracts of selected medicinal plants of South India. Saudi J Biol Sci 22:532–538
Fan D, Zhou X, Zhao C, Chen H, Zhao Y, Gong X (2011) Anti-inflammatory, antiviral and quantitative study of quercetin-3-O-β-D-glucuronide in Polygonum perfoliatum L. Fitoterapia 82:805–810
Friedman M (2007) Overview of antibacterial, antitoxin, antiviral, and antifungal activities of tea flavonoids and teas. Mol Nutr Food Res 51:116–134
Grienke U, Schmidtke M, von Grafenstein S, Kirchmair J, Liedl KR, Rollinger JM (2012) Influenza neuraminidase: a druggable target for natural products. Nat Prod Rep 29:11–36
Gross G, Meyer K, Pres H, Thielert C, Tawfik H, Mescheder A (2007) A randomized, double-blind, four-arm parallel-group, placebo-controlled Phase II/III study to investigate the clinical efficacy of two galenic formulations of Polyphenon® E in the treatment of external genital warts. J Eur Acad Dermatol Venereol 21:1404–1412
Guo J-P, Pang J, Wang X-W, Shen Z-Q, Jin M, Li J-W (2006) In vitro screening of traditionally used medicinal plants in China against enteroviruses. World J Gastroenterol 12:4078
Gupta M, Chakrabarti S, Bhattacharya S, Rath N (1997) Anti-epileptic and anti-cancer activity of some indigenous plants. Indian J Physiol Allied Sci 51:53–56
Heneghan C, Onakpoya I, Jones MA, Doshi P, Del Mar C, Hama R, Thompson MJ, Spencer EA, Mahtani KR, Nunan D (2016) Neuraminidase inhibitors for influenza: a systematic review and meta-analysis of regulatory and mortality data. Science 5:666
Imai M, Yamashita M, Sakai-Tagawa Y, Iwatsuki-Horimoto K, Kiso M, Murakami J, Yasuhara A, Takada K, Ito M, Nakajima N (2020) Influenza A variants with reduced susceptibility to baloxavir isolated from Japanese patients are fit and transmit through respiratory droplets. Nat Microbiol 5:27–33
Kaplan W, Littlejohn TG (2001) Swiss-PDB viewer (deep view). Brief Bioinformatics 2:195–197
Kim Y, Narayanan S, Chang K-O (2010) Inhibition of influenza virus replication by plant-derived isoquercetin. Antivir Res 88:227–235
Laskowski RA (2001) PDBsum: summaries and analyses of PDB structures. Nucleic Acids Res 29:221–222
Laskowski RA, Swindells MB (2011) LigPlot+: multiple ligand–protein interaction diagrams for drug discovery. Science 2:556
Lipinski CA (2004) Lead-and drug-like compounds: the rule-of-five revolution. Drug Discov Today Technol 1:337–341
Liu Z, Zhao J, Li W, Wang X, Xu J, Xie J, Tao K, Shen L, Zhang R (2015) Molecular docking of potential inhibitors for influenza H7N9. Comput Math Methods Med 2:888
Liu J, Zu M, Chen K, Gao L, Min H, Zhuo W, Chen W, Liu A (2018) Screening of neuraminidase inhibitory activities of some medicinal plants traditionally used in Lingnan Chinese medicines. BMC Complem Altern Med 18:1–11
Manzocco L, Anese M, Nicoli MC (1998) Antioxidant properties of tea extracts as affected by processing. LWT Food Sci Technol 31:694–698
McCullers JA, Wang GC, He S, Webster RG (1999) Reassortment and insertion-deletion are strategies for the evolution of influenza B viruses in nature. J Virol 73:7343–7348
Moghadami M (2017) A narrative review of influenza: a seasonal and pandemic disease. Iran J Med Sci 42:2
Mohanraj K, Karthikeyan BS, Vivek-Ananth RP, Chand RPB, Aparna SR, Mangalapandi P, Samal A (2018) IMPPAT: a curated database of I ndian M edicinal P lants, P hytochemistry A nd T herapeutics. Sci Rep 8:1–17
Mukoyama A, Ushijima H, Nishimura S, Koike H, Toda M, Hara Y, Shimamura T (1991) Inhibition of rotavirus and enterovirus infections by tea extracts. Japan J Med Sci Biol 44:181–186
Naeem A, Elbakkouri K, Alfaiz A, Hamed ME, Alsaran H, AlOtaiby S, Enani M, Alosaimi B (2020) Antigenic drift of hemagglutinin and neuraminidase in seasonal H1N1 influenza viruses from Saudi Arabia in 2014 to 2015. J Med Virol 92:3016–3027
Nakayama M, Toda M, Okubo S, Shimamura T (1990) Inhibition of influenza virus infection by tea. Lett Appl Microbiol 11:38–40
Peteranderl C, Herold S, Schmoldt C (2016) Human influenza virus infections. In: Semin Respir Crit Care Med. Thieme Medical Publishers, pp 487–500
Pleschka S, Stein M, Schoop R, Hudson JB (2009) Anti-viral properties and mode of action of standardized Echinacea purpurea extract against highly pathogenic avian influenza virus (H5N1, H7N7) and swine-origin H1N1 (S-OIV). Virol J 6:1–9
Poonia AU (2015) Chenopodium album Linn: review of nutritive value and biological properties. J Food Sci Technol 52:3977–3985
Pulendran B, Maddur MS (2014) Innate immune sensing and response to influenza. Influenza Pathog Control II:23–71
Rahman SR, Ahmed MF, Islam MA, Rahman MM (2016) Effect of risk factors on the prevalence of influenza infections among children of slums of Dhaka city. Springerplus 5:1–6
Rahman M, Hoque SA, Islam MA, Rahman SR (2017) Molecular analysis of amantadine-resistant influenza A (H1N1 pdm09) virus isolated from slum dwellers of Dhaka, Bangladesh. Virus Genes 53:377–385
Rajão DS, Pérez DR (2018) Universal vaccines and vaccine platforms to protect against influenza viruses in humans and agriculture. Front Microbiol 9:123
Rajasekaran D, Palombo EA, Chia Yeo T, Lim Siok Ley D, Lee TuC, Malherbe F, Grollo L (2013) Identification of traditional medicinal plant extracts with novel anti-influenza activity. PLoS ONE 8:e79293
Rajeshwar Y, Gupta M, Mazumder UK (2005) In vitro lipid peroxidation and antimicrobial activity of Mucuna pruriens seeds. Science 5:7996
Ren C-Z, Hu W-Y, Li J-C, Xie Y-H, Jia N-N, Shi J, Wei Y-Y, Hu T-J (2020) Ethyl acetate fraction of flavonoids from Polygonum hydropiper L. modulates pseudorabies virus-induced inflammation in RAW264. 7 cells via the NF-κB and MAPK pathways. J Vet Med Sci 2:20–263
Sadati SM, Gheibi N, Ranjbar S, Hashemzadeh MS (2019) Docking study of flavonoid derivatives as potent inhibitors of influenza H1N1 virus neuraminidase. Biomed Rep 10:33–38
Sathiyanarayanan L, Arulmozhi S (2007) Mucuna pruriens Linn.-A comprehensive review. Pharmacogn Rev 1:6888
Shivanika C, Kumar D, Ragunathan V, Tiwari P, Sumitha A (2020) Molecular docking, validation, dynamics simulations, and pharmacokinetic prediction of natural compounds against the SARS-CoV-2 main-protease. J Biomol Struct 1:998
Shoji M, Woo S-Y, Masuda A, Win NN, Ngwe H, Takahashi E, Kido H, Morita H, Ito T, Kuzuhara T (2017) Anti-influenza virus activity of extracts from the stems of Jatropha multifida Linn. collected in Myanmar. BMC Complem Altern Med 17:1–7
Sivaraman D, Ratheeshkumar KS, Muralidaran P (2010) Effect of ethanolic seed extract of Mucuna pruriens (L.) DC var. utilis on haloperidol induced tardive dyskinesia in rats. Int J Pharm Sci Rev Res 3:6000
Song J-M, Lee K-H, Seong B-L (2005) Antiviral effect of catechins in green tea on influenza virus. Antivir Res 68:66–74
Tran TT, Kim M, Jang Y, Lee HW, Nguyen HT, Nguyen TN, Park HW, Le Dang Q, Kim J-C (2017) Characterization and mechanisms of anti-influenza virus metabolites isolated from the Vietnamese medicinal plant Polygonum chinense. BMC Complem Altern Med 17:1–11
Yamayoshi S, Kawaoka Y (2019) Current and future influenza vaccines. Nat Med 25:212–220
Yang Y, Yu T, Jang H-J, Byeon SE, Song S-Y, Lee B-H, Rhee MH, Kim TW, Lee J, Hong S (2012) In vitro and in vivo anti-inflammatory activities of Polygonum hydropiper methanol extract. J Ethnopharmacol 139:616–625
Zu M, Yang F, Zhou W, Liu A, Du G, Zheng L (2012) In vitro anti-influenza virus and anti-inflammatory activities of theaflavin derivatives. Antivir Res 94:217–224
Acknowledgements
The study was funded by Ministry of Science and Technology, Government of the People’s Republic of Bangladesh.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical statement
This article does not contain any studies involving animals performed by any of the authors. This article does not contain any studies involving human participants performed by any of the authors.
Conflict of interest
Md Abu Sayem Khan has no conflict of interest. Rifat Parveen has no conflict of interest. Sheikh Ariful Hoque has no conflict of interest. Md Firoz Ahmed has no conflict of interest. Abu Shara Shamsur Rouf has no conflict of interest. Sabita Rezwana Rahman has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Khan, M.A.S., Parveen, R., Hoque, S.A. et al. Implementing in vitro and in silico approaches to evaluate anti-influenza virus activity of different Bangladeshi plant extracts. ADV TRADIT MED (ADTM) 23, 915–928 (2023). https://doi.org/10.1007/s13596-022-00669-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-022-00669-9