Abstract
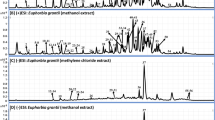
This study investigated the restorative effect of Leea macrophylla ethanol root extract (LMERE) in carbon tetrachloride (CCl4) induced hepatic injury. It also tried to unfold the underlying mechanism through ligand-receptor interactions. Prior to conduct the CCl4 induced animal model study, the in vitro antioxidative capacities of LMERE were investigated. Gas chromatography mass spectroscopy (GC–MS) was accomplished to identify the prevalent bioactive compounds. The molecular docking was performed using Schrödinger Suites 2017-1. Results showed the promising antioxidative potentials of LMERE in in vitro models. Upon treatment of CCl4 intoxicated animals with LMERE, serum ALT and AST were found to be significantly (p < 0.05) reduced compared to the CCl4 control while LMERE50 was noted as the best dose in restoring the hepatic markers. Serum lipids and total protein were significantly restored compared to control. Remarkable changes of cell necrosis, apoptosis and sinusoidal dilution were noticed in histopathological assay of liver tissue. mRNA expression for superoxide dismutase (SOD1) and catalase was multifold increased which are statistically significant compared to reference drug, silymarin. In docking study, octadecanoic acid showed the lowest binding energy and highest binding affinity with the protein (ID: 1VKX) which is a crystallized structure of NF-κB p50/p65 heterodimer involved in cytokine production. The findings demonstrate that LMERE restores the hepatic damage by the mRNA expression of antioxidative enzymes while LMERE50, at a glance, seems the most suitable dose.
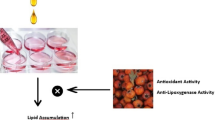
Graphic abstract








Similar content being viewed by others
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- CHL:
-
Cholesterol
- LDL:
-
Low density lipoprotein
- TSP:
-
Total serum protein
- TG:
-
Triglycerides
- SEM:
-
Standard error for mean
- LMERE:
-
Leea macrophylla ethanol root extract
- ROS:
-
reactive oxygen species
- SOD1:
-
Super oxide dismutase 1
- CAT:
-
Catalase
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- LMERE 50:
-
Leea macrophylla ethanol root extract 50 mg per kg body weight
- LMERE 100:
-
Leea macrophylla ethanol root extract 100 mg per kg body weight
- LMERE 200:
-
Leea macrophylla ethanol root extract 200 mg per kg body weight
References
Ali H, Dixit S, Ali D et al (2015) Isolation and evaluation of anticancer efficacy of stigmasterol in a mouse model of DMBA-induced skin carcinoma. Drug Des Dev Ther 9:2793–2800. https://doi.org/10.2147/DDDT.S83514
Benzie IFF, Strain JJ (1996) The ferric reducing ability of plasma (FRAP) as a measure of ‘antioxidant power’: the FRAP assay. Anal Biochem 239:70–76. https://doi.org/10.1006/abio.1996.0292
Byass P (2014) The global burden of liver disease: a challenge for methods and for public health. BMC Med. https://doi.org/10.1186/s12916-014-0159-5
DiMasi JA, Hansen RW, Grabowski HG (2003) The price of innovation: new estimates of drug development costs. J Health Econ 22(2):151–185. https://doi.org/10.1016/S0167-6296(02)00126-1
Domitrovic R, Potocnjak I (2016) A comprehensive overview of hepatoprotective natural compounds: mechanism of action and clinical perspectives. Arch Toxicol 90:39–79. https://doi.org/10.1007/s00204-015-1580-z
Dutta S, Chakraborty AK, Dey P et al (2018) Amelioration of CCl4 induced liver injury in swiss albino mice by antioxidant rich leaf extract of Croton bonplandianus Baill. PLoS ONE 13(4):e0196411. https://doi.org/10.1371/journal.pone.0196411
Friesner RA, Banks JL, Murphy RB et al (2004) Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem 47(7):1739–1749. https://doi.org/10.1021/jm0306430
Friesner RA, Murphy RB, Repasky MP et al (2006) Extra precision glide: docking and scoring incorporating a model of hydrophobic enclosure for protein − ligand complexes. J Med Chem 49(21):6177–6196. https://doi.org/10.1021/jm051256o
Genie SA, Dar TA, Zargar B et al (2014) Antioxidant and hepatoprotective effects of Crataegus Songarica methanol extract. J Environ Pathol Toxicol Oncol 33:131–143. https://doi.org/10.1615/JEnvironPatholToxicolOncol.2014010606
Gilmore TD (2006) Introduction to NF-κB: players, pathways, perspectives. Oncogene 25(51):6680. https://doi.org/10.1038/sj.onc.1209954
Girish CK, Oner BC, Jayanthi S et al (2009) Hepatoprotective activity of six polyherbal formulations in paracetamol induced liver toxicity in mice. Indian J Me Res 129:569–578
Halgren T (2007) New method for fast and accurate binding-site identification and analysis. Chem Biol Drug Des 69(2):146–148. https://doi.org/10.1111/j.1747-0285.2007.00483.x
Halgren TA (2009) Identifying and characterizing binding sites and assessing druggability. J Chem Inf Model 49(2):377–389. https://doi.org/10.1021/ci800324m
Halgren TA, Murphy RB, Friesner RA et al (2004) Glide: a new approach for rapid, accurate docking and scoring. 2. Enrichment factors in database screening. J Med Chem 47(7):1750–1759. https://doi.org/10.1021/jm030644s
Harder E, Damm W, Maple J (2015) OPLS3: a force field providing broad coverage of drug-like small molecules and proteins. J Chem Theory Comput 12(1):281–296. https://doi.org/10.1021/acs.jctc.5b00864
Jahn CE, Schaefer EJ, Taam LA et al (1985) Lipoprotein abnormalities in primary biliary cirrhosis association with hepatic lipase inhibition as well as altered cholesterol esterification. Gastroenterology 89:1266–1278. https://doi.org/10.1016/0016-5085(85)90642-0
Kumar CH, Ramesh A, Kumar JNS, Ishaq BM (2011) A review on hepatoprotective activity of medicinal plants. Int J Pharm Sci Res 23:501–515
Li S, Tan HY, Wang N et al (2015) The role of oxidative stress and antioxidants in liver diseases. Int J Mol Sci 16:26087–26124. https://doi.org/10.3390/ijms161125942
Lin JY, Tang CY (2007) Determination of total phenolic and flavonoid contents in selected fruits and vegetables, as well as their stimulatory effects on mouse splenocyte proliferation. Food Chem 101:140–147. https://doi.org/10.1016/j.foodchem.2006.01.014
Marcellin P, Kutala BK (2018) Liver diseases: a major, neglected global public health problem requiring urgent actions and large-scale screening. Liver Int 38:2–6. https://doi.org/10.1111/liv.13682
Marí M, Colell A, Morales A et al (2010) Redox control of liver function in health and disease. Antioxid Redox Signal 12:1295–1331. https://doi.org/10.1089/ars.2009.2634
Meena AK, Rao MM (2010) Folk herbal medicines used by the Meena community in Rajasthan. Asian J Trad Med 5:19–31
Nayal M, Honig B (2006) On the nature of cavities on protein surfaces: application to the identification of drug-binding sites. Proteins: Struct, Funct, Bioinf 63(4):892–906. https://doi.org/10.1002/prot.20897
Nizami AN, Rahman MA, Ahmed NU, Islam MS (2012) Whole Leea macrophylla ethanolic extract normalizes kidney deposits and recovers renal impairments in an ethylene glycol-induced urolithiasis model of rats. Asian Pac J Trop Med 5:533–538. https://doi.org/10.1016/S1995-7645(12)60094-7
Olsson MH, Søndergaard CR, Rostkowski M, Jensen JH (2011) PROPKA3: consistent treatment of internal and surface residues in empirical pKa predictions. J Chem Theory Comput 7(2):525–537. https://doi.org/10.1021/ct100578z
Pandey MM, Govindarajan R, Rawat AKS, Pushpangadan P (2005) Free radical scavenging potential of Saussarea costus. Acta Pharm 55:297–304
Parejo I, Viladomat F, Bastida J et al (2002) Comparison between the radical scavenging activity and antioxidant activity of six distilled and non-distilled Mediterranean herbs and aromatic plants. J Agric Food Chem 50:6882–6890. https://doi.org/10.1021/jf020540a
Pastor CM, Billiar TR, Losser MR, Payen DM (1995) Liver injury during sepsis. J Crit Care 10:183–197. https://doi.org/10.1016/0883-9441(95)90010-1
Recknagel RO, Glende EA, Dolack JA, Walter RL (1992) Mechanism of carbon tetrachloride toxicity. Pharmacol Ther 43:139–154
Rincón CTS, Montoya JEZ, Gómez GLC (2014) Optimizing the extraction of phenolic compounds from Bixa orellana L. and effect of physicochemical conditions on its antioxidant activity. J Med Plant Res 8:1333–1339
Roy S, Uddin MZ, Hassan MA, Rahman MM (2008) Medico-botanical report on the Chakma community of Bangladesh. Bangl J Plant Taxon 15:67–72. https://doi.org/10.3329/bjpt.v15i1.929
Ruot B, Béchereau F, Bayle G et al (2002) The response of liver albumin synthesis to infection in rats varies with the phase of the inflammatory process. Clin Sci 102:107–114. https://doi.org/10.1042/cs1020107
Sahreen S, Khan MR, Khan RA (2011) Hepatoprotective effects of methanol extract of Carissa opaca leaves on CCl4-induced damage in rat. BMC Complement Altern Med 11:48. https://doi.org/10.1186/1472-6882-11-48
Saleem TSM, Chetty CM, Ramkanth S et al (2010) Hepatoprotective herbs—a review. Int J Res Pharm Sci 1:1–5
Sanja SD, Sheth NR, Patel NK et al (2009) Characterization and evaluation of antioxident activity of Portulaca oleracea. Int J Pharm Pharm Sci 1:74–84
Sastry GM, Adzhigirey M, Day T et al (2013) Protein and ligand preparation: parameters, protocols, and influence on virtual screening enrichments. J Comput Aided Mol Des 27(3):221–234. https://doi.org/10.1007/s10822-013-9644-8
Shelley JC, Cholleti A, Frye LL et al (2007) Epik: a software program for pK a prediction and protonation state generation for drug-like molecules. J Comput Aided Mol Des 21:681–691. https://doi.org/10.1007/s10822-007-9133-z
Singal AK, Jampana SC, Weinman SA (2011) Antioxidants as therapeutic agents for liver disease. Liver Int 31:1432–1448. https://doi.org/10.1111/j.1478-3231.2011.02604.x
Singh J, Chuaqui CE, Boriack-Sjodin PA et al (2003) Successful shape-based virtual screening: the discovery of a potent inhibitor of the type I TGFβ receptor kinase (TβRI). Bioorg Med Chem Lett 13(24):4355–4359. https://doi.org/10.1016/j.bmcl.2003.09.028
Smirnoff N, Cumbes QJ (1989) Hydroxyl radical scavenging activity of compatible solutes. Phytochemistry 28:1057–1060. https://doi.org/10.1016/0031-9422(89)80182-7
Søndergaard CR, Olsson MH, Rostkowski M et al (2011) Improved treatment of ligands and coupling effects in empirical calculation and rationalization of pKa values. J Chem Theory Comput 7(7):2284–2295. https://doi.org/10.1021/ct200133y
Subramoniam A, Pushpangadan P (1999) Development of phytomedicines for liver diseases. Indian J Pharmacol 31:166–175
Swarnkar S, Katewa SS (2008) Ethnobotanical observation on tuberous plants from tribal area of Rajasthan (India). Ethnobot Leaflets 12:647–666
Thabrew MI, Joice PD, Rajatissa W (1987) A comparative study of the efficacy of Pavetta indica and Osbeckia octandra in the treatment of liver dysfunction. Planta Med 53:239–241. https://doi.org/10.1055/s-2006-962691
Wang ZJ, Liang CL, Li GM et al (2007) Stearic acid protects primary cultured cortical neurons against oxidative stress. Acta Pharmacol Sin 28:315–326. https://doi.org/10.1111/j.1745-7254.2007.00512.x
WHO (1996) Quality assurance of pharmaceuticals: a compendum of guidelines and related materials, good manufacturing practices and inspection, vol 2. World Health Organization, Geneva
Wu ZY, Raven PH, Hong DY (eds) (2007) Flora of China, vol 12 (Hippocastanaceae through Theaceae). Science Press, Beijing; Missouri Botanical Garden Press, St. Louis
Zaoui A, Cherrah Y, Mahassini N, Alaoui K, Amarouch H, Hassar M (2002) Acute and chronic toxicity of Nigella sativa fixed oil. Phytomedicine 9:69–74. https://doi.org/10.1078/0944-7113-00084
Acknowledgements
The authors are grateful to the Department of Biochemistry and Molecular Biology, University of Chittagong, Bangladesh, for providing the facilities to conduct this research work. The authors are also thankful to Dr. Sheikh Bokhtear Uddin, Professor, Department of Botany, University of Chittagong, for identifying the plant. Moreover, we appreciate the tremendous technical supports from laboratory of Eukaryotic Gene Expression and Function (EuGEF), Department of Biochemistry and Molecular Biology, University of Chittagong.
Funding
This research has been partially funded by Ministry of Science and technology, Bangladesh under the special allocation for research with the Grant Number BS-176/180/MOST.
Author information
Authors and Affiliations
Contributions
This work was carried out in collaboration between all authors. Authors MRHB collected the plant materials (root) and carried out the study design, data collection, data interpretation, manuscript preparation and statistical analysis. Author MAR supervised the study design and data interpretation as well as contributed in manuscript preparation and statistical analysis. MAR also acted for all correspondences. Author MZR contributed in histopathological scoring of liver tissue sections. MK and MA contributed in phytochemical analysis especially GC–MS preparation and interpretation of the relevant data. MAHC has conducted all the in vitro antioxidative works on LMERE. TBE and MAI participated in the manuscript draft and has thoroughly checked and revised the manuscript for necessary changes in format, grammar and English standard. All authors read and approved the final version of the manuscript. SK has arranged the in silico docking study.
Corresponding author
Ethics declarations
Ethical Statement
Animals were handled and maintained according to the guidelines of Institutional Animal Ethics Committee of the Faculty of Biological Science of the University of Chittagong, Bangladesh (AERB/FBS/UC/02, 2015).
Conflict of interest
Md. Rakibul Hassan Bulbul has no conflict of interest. Md. Atiar Rahman has no conflict of interest. Md. Zillur Rahman has no conflict of interest. Talha Bin Emran has no conflict of interest. Mirola Afroze has no conflict of interest. Mala Khan has no conflict of interest. Muhammad Abid Hasan Chowdhury has no conflict of interest. Mohammed Auwal Ibrahim has no conflict of interest. Mohammed Sohel Chowdhury has no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Bulbul, M.R.H., Rahman, M.A., Rahman, M.Z. et al. Leea macrophylla (Roxb.) root extract reverses CCl4 induced liver injury through upregulation of antioxidative gene expression: a molecular interaction for therapeutic inception. ADV TRADIT MED (ADTM) 20, 35–52 (2020). https://doi.org/10.1007/s13596-019-00380-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13596-019-00380-2