Abstract
Key message
Climate factors affect seed biomass production which in turn influences autumn wild boar spatial behaviour. Adaptive management strategies require an understanding of both masting and its influence on the behaviour of pulsed resource consumers like wild boar.
Context
Pulsed resources ecosystem could be strongly affected by climate. Disantangling the role of climate on mast seeding allow to understand a seed consumer spatial behaviour to design proper wildlife and forest management strategies.
Aims
We investigated the relationship between mast seeding and climatic variables and we evaluated the influence of mast seeding on wild boar home range dynamics.
Methods
We analysed mast seeding as seed biomass production of three broadleaf tree species (Fagus sylvatica L., Quercus cerris L., Castanea sativa Mill.) in the northern Apennines. Next, we explored which climatic variables affected tree masting patterns and finally we tested the effect of both climate and seed biomass production on wild boar home range size.
Results
Seed biomass production is partially regulated by climate; high precipitation in spring of the current year positively affects seed biomass production while summer precipitation of previous year has an opposite effect. Wild boar home range size is negatively correlated to seed biomass production, and the climate only partially contributes to determine wild boar spatial behaviour.
Conclusion
Climate factors influence mast seeding, and the negative correlation between wild boar home range and mast seeding should be taken into account for designing integrated, proactive hunting management.
Similar content being viewed by others
1 Introduction
The understanding of species responses to climate variation is essential for designing proactive management strategies (Heller and Zavaleta 2009). Changes in climate and land use affect distribution, phenology and population dynamics of several organisms (Bellard et al. 2012; Pagel and Shurr 2012). Changes can affect inner ecosystem processes and functioning at various scales and both flora and fauna have to cope with or adapt to these phenomena (Walther et al. 2002; Parmesan 2007). However, making predictions about the particular response of a species to climate change is difficult because the predictions hinge on knowing how climate influences them directly and indirectly. This implies that an integrated approach is required to properly understand the exact nature and direction of emergent effects because they are the net result of multiple species responding to multiple changes in their environment.
Several studies have demonstrated that plant physiology and phenology are often driven and altered by climatic variables (Michelot et al. 2012; Reyer et al., 2013) and this can create phenological mismatches between different trophic levels within ecosystems (DeLucia et al. 2012; Jamieson et al. 2012; Reed et al. 2013). These effects, in turn, are expected to alter trophic interaction within food chains. Therefore, understanding climatic constraints on plant species is crucial as plants represent the base of ecological food chains (Voight et al. 2003; Barton et al. 2009).
Mast seeding (or masting) is a natural process where pulsed resources create lagged responses in successive trophic levels of ecological communities. Numerous studies have demonstrated that mast seeding in many plant species is driven by external factors such as variations in climate (Williamson and Ickes 2002; Lusk et al. 2007; Drobyshev et al. 2010; Cutini et al. 2015). For example, this was the case of beech masting, which typically occurs after a year characterised by high summer temperature and low precipitation (Piovesan and Adams 2001; Övergaard et al. 2007; Drobyshev et al. 2014). Such strong interplay between plant masting and climate is in line with the environmental prediction hypothesis namely, “large reproductive episodes are timed to anticipate favourable conditions for establishment” (Kelly 1994). In this context, plant reproduction become extremely important in shaping forest ecosystem response to global climate change as the vulnerability or ability of plants to adapt will affect all of the food chain. This relationship between producer and consumer is more evident in pulsed resource ecosystems, where the equilibrium between mast seeding plants and vertebrates heavily relies on their synchronisation (Kelly and Sork 2002; Clotfelter et al., 2007; Boutin et al. 2006; Wauters et al. 2008; Cutini et al. 2013).
In the next century, the Mediterranean basin will be one of the regions most affected by climate change (Giorgi and Lionello 2008) and this could have severe consequences on forest productivity (Boisvenue and Running 2006) including mast seeding. Indeed, masting is a typical feature of the Fagaceae (Hiroki and Matsubara 1995; Hilton and Packham 2003; Espelta et al. 2008; Montserrat-Martì et al. 2009), a tree and shrub family that includes the prevailing species of the Mediterranean region, i.e. beech (Fagus sylvatica L.), chestnut (Castanea sativa Mill.) and various evergreen and deciduous oak (Quercus spp.) species (Skjøth et al. 2008). Nonetheless, the interaction between mast seeding, climate and seed-consumers in the Mediterranean region have received limited attention, particularly regarding ungulate species that are of economic interest from a forest management perspective (Bieber and Ruf 2005; Cutini et al. 2013; Canu et al. 2015; Vetter et al. 2015). Therefore, a comprehensive understanding of the mechanisms involved between climate, forest and ungulate dynamics and behaviour represents an important issue for an effective management of forest and wildlife resources (Chianucci et al. 2015; Cutini et al. 2013).
Among ungulates, the wild boar (Sus scrofa L.) exhibits strong responses to food pulses (Groot Bruinderink et al. 1994; Cutini et al. 2013). The increasing wild boar densities observed in Europe during the last five decades (Apollonio et al. 2010) have led to recent increase in human-wildlife conflicts. As a consequence, ecologists have only relatively recently paid attention to the role of pulsed resources and climate-induced variations in wild boar population dynamics (e.g. Cutini et al. 2013) and life-history traits (e.g. Servanty et al. 2009; Canu et al. 2015). On the other hand, the understanding of climatic effects on direct and indirect trophic interactions requires a long-term perspective and a temporal sequence of information including climate, forest and fauna data.
Several factors could influence both wild boar spatial behaviour and plant reproduction but to better understand their interaction and the role of climate as a first contributor, we decided to analyse 23 years masting data in three most common broadleaved tree species in the Apennines (Fagus sylvatica L., Quercus cerris L., Castanea sativa Mill.) and 8 years wild boar spatial behaviour to assess: (1) if climatic variables trigger masting patterns, identifying weather cues that better explain seed biomass production; and (2), by controlling the direct effect of climate experienced by each individual, if a relation exists between wild boar spatial behaviour, i.e. its home range, and mast seeding. This was motivated because wild boar diet in the Apennines heavily relies on acorns, chestnuts and beech nuts (Massei et al. 1996, 1997). Hence, according to the ‘food-exploitation hypothesis’ of Larter and Gates (1994), home range size of a species is inversely related to food availability. Hence, we hypothesised that wild boar home range extension should increase when food availability decrease.
2 Methods
2.1 Study area
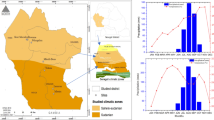
This study was conducted in a mountainous area in the Central Apennines (Alpe di Catenaia, 4579 ha, 43° 48′ N, 11° 49′ E, Fig. 1). The climate is temperate, with warm, dry summers and cold, rainy winters. The mean annual rainfall is 1224 mm, and the mean annual temperature 9.5 °C. Approximately 87% of the area was covered with forests, mainly broadleaved forests (about 90% of the total forested area). The most diffuse tree species in the area are Turkey oak (Quercus cerris L.), beech (Fagus sylvatica L.) and chestnut (Castanea sativa Mill), which cover respectively 14.9, 22.1 and 7.8% of the forest area. These species are mainly represented by even-aged, pure stands, which are currently managed as coppice in transition to high forest (Cutini et al. 2015). These stands are mainly spread between 700 and 1100 m a.s.l and are mainly aged between 50 and 70 years old.
Mountainous study area in the province of Arezzo, Central Italy. Wild boar distribution based on the Minimum Convex Polygon of all locations of individual animals (October–December period from 2002 to 2009). Forested areas with prevalence of Turkey oak, beech or chestnut (grey colour) in the Alpe di Catenaia (Arezzo Province) (main panel) and in the whole of Italy (left top panel; land use data available from CORINE Land Cover—level IV—Commission of the European Communities, 2012)
2.2 Seed biomass production
Three 1-ha permanent plots in beech, chestnut and Turkey oak stands were selected in the study area, in which masting data were collected since the 90s. Annual masting were estimated using the ‘littertrap’ method, following the protocol by Chianucci and Cutini (2013) as described below. Nine 0.25-m2 litter traps were positioned in each stand 1 m above ground level using a systematic grid sampling. Litterfall was collected every 2 weeks during fall and winter and monthly during the rest of the year. The collected litter was separated in laboratory into main components (leaves, twigs, seeds, husks) and then oven-dried at 85° ± 2 °C until constant weight. The obtained seed biomass production was converted as Mg ha−1 year−1.
2.3 Observed climate data
Temperature and precipitation data were collected at three weather stations located inside and around the study area at different elevations (i.e. 420, 500 and 900 m a.s.l.; Fig. 1) by the National Forestry Service (UTB 2015). Daily data were available since 1989 (26 years). We calculated the daily average temperature (°C) and the cumulated daily precipitation for the following periods as predictor variables that potentially influence mast seed production (points 1 and 2; Piovesan and Adams 2001) and eventually wild boar spatial behaviour directly (point 3; Keuling et al. 2008; Thurfjell et al. 2014):
-
1.
Daily average temperature (°C) and cumulated daily precipitation (mm) during spring (March–May—considering the current year, year−1 and year−2 for each mast season);
-
2.
Daily average temperature (°C) and cumulated daily precipitation (mm) during summer (June–August, considering the current year, year−1 and year−2 for each mast season);
-
3.
Daily average temperature (°C) and cumulated daily precipitation (mm) during autumn (October–December, considering the current year for each wild boar home range size).
2.4 Wild boar spatial behaviour data
Radio-tracking location data were collected from April 2002 to August 2010 by triangulation using bearings obtained from three different points (White and Garrott 1990) using the ‘loudest signal’ method. Bearings were marked out on a 1:10000 scale map (Kenward 1987) of the study area keeping into account that the error box should be smaller than 1 ha. The telemetry data were uniformly distributed over the 24 h (discontinuous telemetry; Swihart and Slade 1985) with 8–12 locations recorded for each boar each month, and considering at least 12 h between consecutive locations to exclude data autocorrelation (Van Winkle 1975). Wild boars were tracked using Wildlife Materials TRX-1000S receivers and a three-element hand-held Yagi antenna. A total of 230 individuals were equipped with VHF tags (collar or VHF-ear tag, with mortality sensor) (Televilt, Sweden, 150-151 MHz wavebands). Location data (minimum 20 locations) were available for 62 adults and subadults wild boars in October–December period. Seasonal home ranges (October–December) were calculated with the R package HRTools (Preatoni and Bisi 2013) and adehabitat (Calenge 2006) using the 95% fixed kernel density estimator (KDE).
2.5 Statistical analyses
The masting time series for each tree species was tested for autocorrelation using an autoregressive model to assess whether there was a pattern in masting behaviour in the different species. To test for possible trend in seed biomass production, we calculate Sen’s slope (Sen 1968; McLeao 2011) and used Mann-Kendall trend test (Mann 1945; Pohlert 2016).
The climate independent variables were standardised as follows:
where WF = climate variable, d = day, s = meteo station, σ = standard deviation.
As proposed by Bisi et al. (2016), we did not focus on single species models because we are not interested in identifying the climatic triggers for each species but we were interested in the overall seed biomass production. As we observed a great difference in seed biomass production between species (see “Mast seeding” paragraph) and we were interested in evaluating mast seeding, we transformed each tree species’ masting series using the formula (Bisi et al. 2016):
where SP = transformed seed biomass production in a given year, \( \overline{SP_{sp}} \)=mean seed biomass production of species sp over entire study period, SPi = seed of species sp in the given year.
The complete standardised dataset of seed biomass production and climate indicators was then analysed using linear mixed effect models (LMEM), with seed biomass production (Mg ha−1) per species and year as dependent variables. Because seed biomass production was not normally distributed (Shapiro-Wilk’s test W = 0.654), it was log-transformed. The log-transformed values met the assumptions of normality (Shapiro-Wilk’s test W = 0.924). We compared a full model with all 12 weather variables and log-transformed seed biomass production of the previous year as predictors.
To reduce the number of predictors, as well as to identify the most effective ones as determinants of the variations in seed biomass production, we performed evaluation of a set of models with all possible combinations (subsets) of terms in the global model, ranking them with AICc (Burnham and Anderson 2002). We estimated goodness of fit as reported in Magee (1990). Since the AICc information criterion penalty for every term is equal to 2, we averaged all the models under delta AICc = 2, weighted by AICc weights.
Finally, correlations between wild boar home range size, annual seed biomass production (not standardised) and climatic variables (autumn temperature and precipitation) were tested by a mixed effects model including sampled boar individual as a random factor. We applied both methods to the complete dataset and to a subsample including only adults (> 24 months old) and subadults (12–24 months old).
All statistical analysis were performed using R software (R Core team 2015) and in particular the packages ‘nlme’ (Pinheiro et al. 2015) and ‘MuMIn’ (Barton 2015).
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
3 Results
3.1 Mast seeding
The autocorrelation function did not show masting patterns for any of the three surveyed tree species, even though a more pronounced autocorrelation was observed in Turkey oak and chestnut; beech showed a weak masting pattern (Online Resource 1). This indicates that reproductive investment each year was not strictly related to the seed produced in the previous year. For all three species, Sen’s slopes are positive (beech 0.012; chestnut 0.035; Turkey oak 0.026), but not statistically significant (beech: tau = 0.111, p = 0.475; chestnut: tau = 0.238, p = 0.139; Turkey oak: tau = 0.169, p = 0.284).
Seed biomass production differed in amount and timing between the tree species (Fig. 2). Annual seed biomass was similar between Turkey oak (mean ± SE = 0.72 ± 0.15 Mg ha−1) and chestnut (mean ± SE = 0.68 ± 0.08 Mg ha−1; Tukey’s HSD p = 0.964), but was significantly lower in beech (mean ± SE = 0.12 ± 0.04 Mg ha−1; Tukey’s HSD, p < 0.001) compared to the other two species.
3.2 Effects of climatic variables on mast seeding
According to the model testing for climate effect on masting, Online Resource 2 reports the first 20 models selected based on the AICc information. Only the first two models had a delta AICc< 2; goodness of fit of the two models are respectively 0.153 for model 1 0.162 for model 2. The average model includes climate variable of year t−1 and specifically ‘Temperature of Spring t−1’ and ‘Precipitation of Summer t−1’, both exhibiting a negative effect on seed biomass production, and ‘Spring Precipitation of year t’ with a positive effect (Table 1).
3.3 Mast seeding and wild boar home range
Models with and without random effects have been run on the same dataset; we only showed results of the model without random effect as AICc value was lower (924 vs 1557); in this model seed biomass production was negatively significantly correlated with wild boar home range size (seed biomass production = − 85.37, SE = 12.12, t = − 7.045, p < 0.001), precipitation was positively correlated (autumn precipitation = 436.87, SE = 126.61, t = 3.45, p < 0.001) and temperature was negatively correlated (− 125.14, SE = 38.96, t = − 3.12). All terms were statistically significant and seed biomass production accounted for the 38% of the total deviance, while temperature accounted for 4.8% and precipitation only for 4%. R2 was 0.47 and model residuals were normally distributed (Shapiro-Wilk normality test = 0.930).
Only one synchronous null masting event was observed (2003); during this year, wild boar home range size (adults and subadults) averaged 779 ha (SE = 113 ha), compared to a total average of 407 ha (SE = 29 ha).
4 Discussion
4.1 Mast seeding
We did not find a clear masting patterns for any of the surveyed tree species; this lack of cyclic pattern, along with the absence of synchronisation reported using the same dataset (Cutini et al. 2013) and in other study areas (Nussbaumer et al. 2016), may partly explain the limited influence of climate on seed biomass production, at least at small scale (i.e. stand level), since the different species most likely exhibit different strategies in allocating resources (Genet et al. 2009) and thus different species-specific responses to climatic inputs.
4.2 Masting patterns and climate drivers
Although many studies have already investigated climate-beech mast seeding relation at broad scale, a limited number of studies have analysed such relationships for chestnut and Turkey oak, i.e. two dominant species with important implication for wild boar management in Mediterranean environments. We found that the mast seeding was only slightly influenced by climatic variables, which explained about 16% of the variance. This is probably due to the fact that tree species masting was not synchronous and that these three species in Central Italy are in the optimum of their distributional range (Cutini et al. 2015), implying that climate conditions are not as limiting as in the extremes of species distribution. However, precipitation during spring (positive influence) and summer (negative influence) in the previous year were the most significantly predictors of mast seeding, in accordance with previous studies conducted on beech and oak species (Piovesan and Adams 2001; Övergaard et al. 2007; Espelta et al. 2008; Scharnweber et al. 2011; Drobyshev et al. 2014). Some authors have interpreted a link between climate and masting under the so-called environmental prediction hypothesis (Kelly 1994), which states that masting strategy has evolved to promote natural regeneration after disturbances, since limiting climate conditions like summer drought may reduce competitor pressure due to canopy trees mortality. This leads to favourable light conditions at the forest floor for seedling establishment (Williamson and Ickes 2002). Although the observed negative correlation between summer temperature and masting does not invalidate this hypothesis, we believe that this mechanism may not be prominent in the Apennines and therefore climate conditions are not as limiting as in the extremes of species distribution. Instead, the regular occurrence of mast years in these species supports the predator satiation hypothesis in accordance with previous findings (Kon et al. 2005) as the change between high and low seed biomass production has a strong impact on animal populations like wild boar, as demonstrated in a previous study (Cutini et al. 2013).
There has been widespread interest in how increase in local mean temperature might affects masting (e.g. Rees et al. 2002; Övergaard et al. 2007). A recent study indicated that mast seeding may be unaffected by gradual increases in mean temperature, since masting is more sensitive to year-to-year variation in summer temperature rather than mean temperature (Kelly et al. 2013). Such hypothesis is interesting as it can be applied to other environmental cues like precipitation, which is often overlooked in modelling mast seeding under environmental change. In our study, we observed that precipitation was more correlated than temperature to seed biomass production, for which we conclude that precipitation can be considered a relevant cue at the local scale. Oak can tolerate drier condition than beech (Breda et al. 2006) particularly when water becomes a limiting factor. In many Mediterranean environments, the process of acorn maturation occurs under water-limited conditions, and oaks usually exhibit high abortion rates during this period (Espelta et al. 2008), which may reflect the influence of both direct (increasing respiratory costs) and indirect effects (increasing water stress) on the internal storage of carbohydrates of the tree. In areas with sufficient water availability, like in Apennines, the seed production is likely mainly driven by light availability, which have direct influence on either the light-use efficiency of the trees, which is related to the photosynthetic performance and thus the ability of trees to accumulate resources to produce seed, and either the length of pollen season, which is a more limiting factor in oak than beech (Bogdziewicz et al. 2017); in this line, the higher shade tolerance of beech can favour this species over less tolerant oak species in non-water-limiting conditions (Scharnweber et al. 2011). The consequences of these trade-offs can influence both growth and seed biomass production and therefore all the components of the consumers community relying on tree masting in Apennines.
4.3 Influence of mast seeding on wild boar spatial behaviour
Schley and Roper (2003) reported that wild boar diet is strongly influenced by food availability and they rely strongly on acorns, beech and chestnuts during mast years (e.g. Fournier-Chambrillon et al. 1995; Massei et al. 1996). Thus, the asynchronous patterns among tree species could contribute to support consumer populations through time (Cannon et al. 2007). Furthermore, previous studies demonstrated that the large forage availability is one of the most important factors shaping wild boar food habits (e.g. Groot Bruinderink et al. 1994).
A previous study by Cutini et al. (2013) conducted in the same wild boar population showed that mast seeding influences the population density; the current study also revealed that mast seeding influences the spatial behaviour of wild boar, in agreement with previous reports on other mammal species (Anderson et al. 2005; Kozakai et al. 2011; Morellet et al. 2013). These population features are linked to two different biological processes: on one hand, the increase of wild boar density after a mast year can be attributed to an increase in reproductive performance of sows, because wild boar females need to reach a threshold body mass (27–33 kg) before breeding (Servanty et al. 2009). On the other hand, increase of home range extension are linked to seasonal food availability, and are particularly driven by the research of alternative food during limiting condition (fall-winter period), consistently with the ‘food-exploitation hypothesis’. However, this species shows a high behavioural plasticity (e.g. Podgórski et al. 2013) and studies testing this hypothesis over a shorter period (3 years) reached contrasting conclusions (Massei et al. 1997) underling the importance of using long time series to understand mammal spatial behaviour. This aspect stresses the fact that the expansion of wild boar populations and their growth may be partially attributed to the behavioural plasticity of the species and its response to different ecological contexts (hunting management, predator presence, forest composition) at population (Podgórski et al. 2013) and individual (Keuling et al. 2008) level. During the wild boar space use monitoring, there has been only one synchronous, null masting event that involved all the sampled species in 2003 and during this year, home range size strongly increased compared to the total average. This emphasises the importance of broadleaf forests for wild boar movement ecology, because alternative food patches require long movements far from wild boar’s usual home ranges when the tree masting was not sufficient. This aspect also demonstrated the plasticity of this species and the importance to collect multiple and long time series dataset to completely understand the ecological role of the different species in the forest ecosystem. The ecological role of wild boar in forest ecosystems should be further investigated because of its complex impact on the plant community, that is in fact not only negative (Genov and Massei 2004; Gomez and Holdar 2008; Wirthner et al. 2012) and can also affect tree masting as reported by Wirthner et al. (2012).
4.4 Management implications
It has been demonstrated that large tree seed availability, particularly during mast years, may reduce wild boar pressure on agricultural crops (e.g. Groot Bruinderink et al. 1994), while low tree seed availability could cause severe damage to agricultural crops (Jezierski and Myrcha 1975; Andrzejewski and Jezierski 1978). Together with increased food availability, the reduced home range of wild boar during mast years offers a clear interpretation of the causal mechanism behind this finding. Therefore, predicting masting pattern of the dominant species can allow to take an adaptive approach when planning proper damage prevention measures and hunting plans. Use of local weather station data can support the prediction of masting patterns in the most dominant species. In addition, forest managers should consider the value of mast produced by the secondary most dominant species as alternative forage for wild boar.
5 Conclusions
This study indicates that climate factors may shape forest dynamics through their influence on mast seeding, but also that the negative correlation between mast seeding and wild boar home range size is an important mechanism whose consequences should be taken into account. Adaptive management strategies require both an understanding of masting and their influence on the behaviour of pulsed resource consumers like wild boar, supporting an integrated wildlife-forest management approach.
References
Anderson DP, Forester JD, Turner MG, Frair JL, Merrill EH, Fortin D, Mao JS, Boyce MS (2005) Factors influencing female home range sizes in elk (Cervus elaphus) in North American landscapes. Landsc Ecol 20:257–271
Andrzejewski R, Jezierski W (1978) Management of a wild boar population and its effects on commercial land. Acta Theriol 23:309–339
Apollonio M, Andersen R, Putman R (2010) Ungulate management in Europe in the XXI century. Cambridge University Press, Cambridge
Barton BT, Beckerman AP, Schmitz OJ (2009) Climate warming strengthens indirect interactions in an old-field food web. Ecology 90:2346–2351
Barton K (2015) MuMIn: multi-model inference. R package version 1(15):1
Bellard C, Bertelsmeier C, Leadley P, Thuiller W, Courchamp F (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15:365–377
Bieber C, Ruf T (2005) Population dynamics in wild boar Sus scrofa: ecology, elasticity of growth rate and implications for the management of pulsed resource consumers. J Appl Ecol 42:1203–1213
Bisi F, von Hardenberg J, Bertolino S, Wauters LA, Imperio S, Preatoni DG, Mazzamuto MV, Provenzale A, Martinoli A (2016) Current and future conifer seed production in the alps: testing weather factors as cues behind masting. Eur J For Res 135:743–754
Bogdziewicz M, Szymkowiak J, Kasprzyk I, Grewling Ł, Borowski Z, Borycka K, Kantorowicz W, Myszkowska D, Piotrowicz K, Ziemianin M, Pesendorfer MB (2017) Masting in wind-pollinated trees: system-specific roles of weather and pollination dynamics in driving seed production. Ecology 98:2615–2625
Boisvenue C, Running SW (2006) Impacts of climate change on natural forest productivity–evidence since the middle of the 20th century. Glob Change Biol 12:862–882
Boutin S, Wauters LA, McAdam AG, Humphries MM, Tosi G, Dhondt AA (2006) Anticipatory reproduction and population growth in seed predators. Science 314:1928–1930
Bréda N, Huc R, Granier A, Dreyer E (2006) Temperate forest trees and stands under severe drought: a review of ecophysiological responses, adaptation processes and long-term consequences. Ann For Sci 63:625–644
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach (2nd ed.), Springer, Verlag
Calenge C (2006) The package adehabitat for the R software: a tool for the analysis of space and habitat use by animals. Ecol Model 197:516–519
Cannon CH, Curran LM, Marshall AJ, Leighton M (2007) Beyond mast-fruiting events: community asynchrony and individual dormancy dominate woody plant reproductive behavior across seven Bornean forest types. Curr Sci India 93:1558–1566
Canu A, Scandura M, Merli E, Chirichella R, Bottero E, Chianucci F, Cutini A, Apollonio M (2015) Reproductive phenology and conception synchrony in a natural wild boar population. Hystrix Ital J Mammal 26:77–84
Chianucci F, Cutini A (2013) Estimation of canopy properties in deciduous forests with digital hemispherical and cover photography. Agric For Meteorol 168:130–139
Chianucci F, Mattioli L, Amorini E, Giannini T, Marcon A, Chirichella R, Apollonio M, Cutini A (2015) Early and long-term impacts of browsing by roe deer in oak coppiced woods along a gradient of population density. Ann Silvicultural Res 39:32–36
Clotfelter ED, Pedersen AB, Cranford JA, Ram N, Snajdr EA, Nolan JV, Ketterson ED (2007) Acorn mast drives long-term dynamics of rodent and songbird populations. Oecologia 154:493–503
Cutini A, Chianucci F, Chirichella R, Donaggio E, Mattioli L, Apollonio M (2013) Mast seeding in deciduous forests of the northern Apennines (Italy) and its influence on wild boar population dynamics. Ann For Sci 70:493–502
Cutini A, Chianucci F, Giannini T, Manetti MC, Salvati L (2015) Is anticipated seed cutting an effective option to accelerate transition to high forest in European beech (Fagus sylvatica L.) coppice stands? Ann For Sci 72:631–640
DeLucia EH, Nabity PD, Zavala JA, Berenbaum MR (2012) Climate change: resetting plant-insect interactions. Plant Physiol 160:1677–1685
Drobyshev I, Övergaard R, Saygin I, Niklasson M, Hickler T, Karlsson M, Sykes MT (2010) Masting behaviour and dendrochronology of European beech (Fagus sylvatica L.) in southern Sweden. For Ecol Manag 259:2160–2171
Drobyshev I, Granström A, Linderholm HW, Hellberg E, Bergeron Y, Niklasson M (2014) Multi-century reconstruction of fire activity in Northern European boreal forest suggests differences in regional fire regimes and their sensitivity to climate. J Ecol 102:738–748
Espelta JM, Cortés P, Molowny-Horas R, Sánchez-Humanes B, Retana J (2008) Masting mediated by summer drought reduces acorn predation in Mediterranean oak forests. Ecology 89:805–817
Fournier-Chambrillon C, Maillard D, Fournier P (1995) Diet of wild boar (Sus scrofa L.) inhabiting the Montpellier garrigue. J Mt Ecol 3:174–179
Genet H, Bréda N, Dufrene E (2009) Age-related variation in carbon allocation at tree and stand scales in beech (Fagus sylvatica L.) and sessile oak (Quercus petraea (Matt.) Liebl.) using a chronosequence approach. Tree Physiol 30:177–192
Genov P, Massei G (2004) The environmental impact of wild boar. Galemys: Boletín informativo de la Sociedad Española para la conservación y estudio de los mamíferos 16:135–145
Giorgi F, Lionello P (2008) Climate change projections for the Mediterranean region. Glob Planet Chang 63:90–104
Gómez JM, Hódar JA (2008) Wild boars (Sus scrofa) affect the recruitment rate and spatial distribution of holm oak (Quercus ilex). For Ecol Manag 256:1384–1389
Groot Bruinderink GWTA, Hazebroek E, van der Voot H (1994) Diet and condition of wild boar, Sus scrofa scrofa, without supplementary feeding. J Zool 233:631–648
Heller NE, Zavaleta ES (2009) Biodiversity management in the face of climate change: a review of 22 years of recommendations. Biol Conserv 142:14–32
Hilton GM, Packam JR (2003) Variation in the masting of common beech (Fagus sylvatica L.) in northern Europe over two centuries (1800–2001). Forestry 76:319–328
Hiroki S, Matsubara T (1995) Fluctuation of nut production and seedling appearance of a Japanese beech (Fagus crenata Blume). Ecol Res 10:161–169
Jamieson MA, Trowbridge AM, Raffa KF, Lindroth RL (2012) Consequences of climate warming and altered precipitation patterns for plant-insect and multitrophic interactions. Plant Physiol 160:1719–1727
Jezierski W, Myrcha A (1975) Food requirements of a wild boar population. Polish Ecol Stud 1:61–83
Kelly D (1994) The evolutionary ecology of mast seeding. Trends Ecol Evol 9:465–470
Kelly D, Sork VL (2002) Mast seeding in perennial plants: why, how, where? Ann Rev Ecol Syst 33:427–447
Kelly D, Geldenhuis A, James A, Penelope Holland E, Plank MJ, Brockie RE, Cowan PE, Harper GA, Lee WG, Maitland MJ, Mark AF, Mills JA, Wilson PR, Byrom AE (2013) Of mast and mean: differential temperature cue makes mast seeding insensitive to climate change. Ecol Lett 16:90–98
Kenward R (1987) Wildlife radio tagging. Academic Press, London
Keuling O, Stier N, Roth M (2008) Annual and seasonal space use of different age classes of female wild boar Sus scrofa L. Eur J Wildl Res 54:403–412
Kon H, Noda T, Terazawa K, Koyama H, Yasaka M (2005) Proximate factors causing mast seeding in Fagus crenata: the effects of resource level and weather cues. Botany 83:1402–1409
Kozakai C, Yamazaki K, Nemoto Y, Nakajima A, Koike S, Abe S, Masaki T, Kaji K (2011) Effect of mast production on home range use of Japanese black bears. J Wildlife Manage 75:867–875
Larter NC, Gates CC (1994) Home range size of wood bison: effects of age, sex and forage availability. J Mammal 75:142–149
Lusk JJ, Swihart RK, Goheen JR (2007) Correlates of interspecific synchrony and interannual variation in seed production by deciduous trees. For Ecol Manag 242:656–670
Magee L (1990) R2 measures based on Wald and likelihood ratio joint significance tests. Am Stat 44:250–253
Mann HB (1945) Nonparametric tests against trend. Econometrica 13:245–259
Massei G, Genov PV, Staines BW (1996) Diet, food availability and reproduction of wild boar in a Mediterranean coastal area. Acta Theriol 41:307–320
Massei G, Genov P, Staines BW, Groman ML (1997) Factors influencing home range and activity of wild boar (Sus scrofa) in a Mediterranean coastal area. J Zool 242:411–423
McLeod AI (2011) Kendall: Kendall rank correlation and Mann-Kendall trend test. R package version 2.2. https://CRAN.R-project.org/package=Kendall
Michelot A, Bréda N, Damesin C, Dufrêne E (2012) Differing growth responses to climatic variations and soil water deficits of Fagus sylvatica, Quercus petraea and Pinus sylvestris in a temperate forest. For Ecol Manag 265:161–171
Montserrat-Martí G, Camarero JJ, Palacio S, Pérez-Rontomé C, Milla R, Albuixech J, Maestro M (2009) Summer-drought constrains the phenology and growth of two coexisting Mediterranean oaks with contrasting leaf habit: implications for their persistence and reproduction. Trees 23:787–799
Morellet N, Bonenfant C, Börger L, Ossi F, Cagnacci F, Heurich M, Kjellander P, Linnell JDC, Nicoloso S, Sustr P, Urbano F, Mysterud A (2013) Seasonality, weather and climate affect home range size in roe deer across a wide latitudinal gradient within Europe. J Anim Ecol 82:1326–1339
Nussbaumer A, Waldner P, Etzold S, Gessler A, Benham S, Thomsen IM, Jørgensen BB, Timmermann V, Verstraeten A, Sioen G, Rautio P (2016) Patterns of mast fruiting of common beech, sessile and common oak, Norway spruce and Scots pine in Central and Northern Europe. For Ecol Manag 363:237–251
Övergaard R, Gemmel P, Karlsson M (2007) Effects of weather conditions on mast year frequency in beech (Fagus sylvatica L.) in Sweden. Forestry 80:555–565
Pagel J, Schurr FM (2012) Forecasting species ranges by statistical estimation of ecological niches and spatial population dynamics. Glob Ecol Biogeogr 21:293–304
Parmesan C (2007) Influences of species, latitudes and methodologies on estimates of phenological response to global warming. Glob Change Biol 13:1860–1872
Pinheiro J, Bates D, DebRoy S, Sarkar D, R Core Team, 2015 _nlme: linear and nonlinear mixed effects models_. R package version 3.1–121, http://CRAN.R-project.org/package=nlme
Piovesan G, Adams JM (2001) Masting behaviour in beech: linking reproduction and climatic variation. Can J Bot 79:1039–1047
Podgórski T, Baś G, Jędrzejewska B, Sönnichsen L, Śnieżko S, Jędrzejewski W, Okarma H (2013) Spatiotemporal behavioral plasticity of wild boar (Sus scrofa) under contrasting conditions of human pressure: primeval forest and metropolitan area. J Mammal 94:109–119
Pohlert T (2016) Trend: non-parametric trend tests and change-point detection. R package version 0.2.0. https://CRAN.R-project.org/package=trend
Preatoni DG, Bisi F (2013) HRTools: commodity functions for home range calculation. R package version 1.0. http://home.prea.net/
R Core Team 2015 R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Reed TE, Grøtan V, Jenouvrier S, Sæther BE, Visser ME (2013) Population growth in a wild bird is buffered against phenological mismatch. Science 340:488–491
Rees M, Kelly D, Bjornstad O (2002) Snow tussocks, chaos, and the evolution of mast seeding. Am Nat 160:44–59
Reyer CP, Leuzinger S, Rammig A, Wolf A, Bartholomeus RP, Bonfante A, de Lorenzi F, Dury M, Gloning P, Jaoudé RA, Klein T, Kuster TM, Martins M, Niedrist G, Riccardi M, Wohlfahrt G, De Angelisi P, De Dato G, Francois L, Menzel A, Pereira M (2013) A plant's perspective of extremes: terrestrial plant responses to changing climatic variability. Glob Change Biol 19:75–89
Sen PK (1968) Estimates of the regression coefficient based on Kendall’s tau. J Am Stat Assoc 63:1379–1389
Servanty S, Jean-Michel G, Carole T, Serge B, Eric B (2009) Pulsed resources and climate-induced variation in the reproductive traits of wild boar under high hunting pressure. J Anim Ecol 78:1278–1290
Scharnweber T, Manthey M, Criegee C, Bauwe A, Schröder C, Wilmking M (2011) Drought matters–declining precipitation influences growth of Fagus sylvatica L. and Quercus robur L. in north-eastern Germany. For Ecol Manag 262:947–961
Schley L, Roper T (2003) Diet of wild boar Sus scrofa in Western Europe, with particular reference to consumption of agricultural crops. Mammal Rev 33:43–56
Skjøth CA, Geels C, Hvidberg M, Hertel O, Brandt J, Frohn LM, Hansen KM, Hedegaard GB, Christensen JH, Moseholm L (2008) An inventory of tree species in Europe—an essential data input for air pollution modelling. Ecol Model 217:292–304
Swihart RK, Slade NA (1985) Testing for independence of observation in animal movements. Ecology 66:1176–1184
Thurfjell H, Spong G, Ericsson G (2014) Effects of weather, season and daylight on female wild boar movement. Acta Theriol 59:467–472
UTB (2015) Ufficio Territoriale per la Biodiversità, Pieve S. Stefano, Province of Arezzo, official data: http://www.indicepa.gov.it/ricerca/n-dettagliouffici.php?prg_ou=37163
Van Winkle W (1975) Comparison of several probabilistic home range models. J Wildl Manag 33:118–123
Vetter SG, Ruf T, Bieber C, Arnold W (2015) What is a mild winter? Regional differences in within-species responses to climate change. PLoS One 10:e0132178
Voight W, Perner J, Davis AJ, Eggers T, Schumacher J, Bährmann R, Fabian B, Heinrich W, Köhler G, Lichter D, Marstaller R, Sander FW (2003) Trophic levels are differentially sensitive to climate. Ecology 84:2444–2453
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TJC, Formentin JM, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
Wauters LA, Githiru M, Bertolino S, Molinari A, Tosi G, Lens L (2008) Demography of alpine red squirrel populations in relation to fluctuations in seed crop size. Ecography 31:104–114
White GC, Garrott RA (1990) Analysis of wildlife radio-tracking data. London Academic Press, San Diego
Williamson GB, Ickes K (2002) Mast fruiting and ENSO cycles–does the cue betray a cause? Oikos 97:459–461
Wirthner S, Schütz M, Page-Dumroese DS, Busse MD, Kirchner JW, Risch AC (2012) Do changes in soil properties after rooting by wild boars (Sus scrofa) affect understory vegetation in Swiss hardwood forests? Can J For Res 42:585–592
Acknowledgements
We thank all other participants of PRIN project who contributed in discussion and shared unpublished results giving important indication to improve first draft of this manuscript. We further acknowledge the Ufficio Territoriale per la Biodiversità for climate data supply and all students and field assistants for wild boar data collection. We thank John Gurnell for useful comments. Finally, we acknowledge the Editor and two anonymous Reviewers for their precious comments.
Funding
This work was partially supported by the Italian Minister of Education, University and Research (PRIN 2010-2011, 20108 TZKHC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Following the Italian law 157/92 on wildlife management, the authors acquired the permission of catching wild boars in 2002 and 2005 from the Regional Government of Tuscany.
Conflict of interest
There are no conflicts of interest and research integrity and ethical standards are maintained.
Additional information
Handling Editor: Andreas Bolte
Contribution of the co-authors
FB: performed statistical analysis and wrote the manuscript; RC: collected the data and wrote the manuscript; FC: collected data and wrote the paper; JvH: helped in statistical designs; AC: wrote the paper; AM: wrote the paper and supervised the work; MA: wrote the paper, supervised the work and coordinated the research project.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Bisi, F., Chirichella, R., Chianucci, F. et al. Climate, tree masting and spatial behaviour in wild boar (Sus scrofa L.): insight from a long-term study. Annals of Forest Science 75, 46 (2018). https://doi.org/10.1007/s13595-018-0726-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13595-018-0726-6