Abstract
Varroa destructor, an ectoparasite of Apis cerana (the Eastern honeybee), has attained global notoriety following the decimation caused by its host shift to Apis mellifera. Previous studies indicated that Varroa mites have geographic and host-specific mitochondrial haplotypes (mitotypes) in A. mellifera colonies, suggesting that only a few genetic strains (i.e., Korean and Japanese mitotypes) can effectively infest A. mellifera. However, growing evidence indicates that native mite populations of A. cerana can hybridize with the mites imported with A. mellifera colonies. This raises the possibility for continuous host shifts of Varroa mites and the consequential hybridizations among mites from different localities, leading to erasure of host specificity among mites of different mitotypes. We compared mitochondrial sequence data with restriction site-associated DNA sequencing (RAD-seq) to test the veracity of the patterns indicated by mitochondrial data and to clarify the genetic differentiation of Varroa mites in Vietnam and Taiwan. Our mitotype analyses replicate previous studies, indicating that only a few Varroa mite mitotype lineages parasitize A. mellifera. However, our RAD-seq data reveal little evidence of the genetic isolation as suggested by the mitotype analyses, instead indicating that hybridization is likely occurring among Varroa mites from multiple different hosts and localities. This indicates that the local mite community of A. cerana might contribute to the genetic pool of mites parasitizing A. mellifera. Our findings suggest that the Varroa mite control strategies and the risk estimation of international apicultural trade should be reassessed.
Similar content being viewed by others
1 Introduction
Varroa destructor, the obligate ectoparasites of honeybees (genus Apis), has been an exemplar of the devastating consequences following host shifts from native hosts to a novel and economically important species (Rosenkranz et al. 2010). This species is a natural ectoparasite of the Eastern honeybee Apis cerana in Asia and has gained global notoriety for its broad-scale decimation of colonies of the western honeybee Apis mellifera. While these mites cause only limited damage to their native host because of a range of presumably co-evolved resistance mechanisms (Peng et al. 1987; Traynor et al. 2020), the combination of the direct effects of parasitism and the indirect effects of vectored viral diseases can be devastating to populations of A. mellifera (Genersch et al. 2010; Rosenkranz et al. 2010; Traynor et al. 2020). At least four instances of host shifts of Varroa mites from local A. cerana colonies (i.e., the A. cerana population native to a geographic area) to the imported A. mellifera colonies have been discovered in Asia (Beaurepaire et al. 2015; Crane 1978; Roberts et al. 2015; Sakai and Okada 1973). Among these four events, the two earlier events (Crane 1978; Sakai and Okada 1973) of infiltration of V. destructor mites to farmed A. mellifera colonies resulted in the rapid dispersal of these viral disease vectors throughout the world, causing serious damage to the global apiculture industry (Traynor et al. 2020; Zhang 2000). While a significant amount of research has been devoted to assessing the history of host shifts in Varroa mites, very little attention has been paid to searching for evidence of spill back of the Varroa mite populations from farmed A. mellifera to the local A. cerana colonies in Asia. Similarly, there has been no assessment of the potential for hybridization of local mites on A. cerana and imported mites on A. mellifera (Dietemann et al. 2019). This may be a crucial knowledge gap, given that hybridization can give rise to rapid parasite evolution, which could result in serious damage to the host species, broaden host range/shifts, increase transmission potential, break down host-specific adaptations, and lead to the development of tolerance/resistance in hybrid populations (King et al. 2015; Pennisi 2018; Schelkle et al. 2012).
Various genetic studies, predominantly relying on mitochondrial DNA (Anderson and Trueman 2000; Beaurepaire et al. 2015; Navajas et al. 2010; Traynor et al. 2020), have attempted to clarify the genetic relationships between populations of V. destructor, to help reconstruct the details of host shifts and subsequent global expansion of the species. Mitotypes of V. destructor were originally identified as belonging to Varroa jacobsoni, another parasite of honeybees with very similar morphology (Anderson and Trueman 2000). This was later recognized to be a species complex, and V. destructor was redefined as new species distinct from V. jacobsoni (Anderson and Trueman 2000). Subsequent studies of mites (Beaurepaire et al. 2015; Navajas et al. 2010; Traynor et al. 2020) using mitochondrial data identified two separate host shifts, specifically from Varroa mites of the Korea 1 (K1) mitotype (Anderson and Trueman 2000) and Japan 1 (J1) mitotype (Anderson and Trueman 2000). In addition, microsatellite data (Beaurepaire et al. 2015) indicated that the Philippine A. cerana and imported A. mellifera colonies shared genetically homogeneous Varroa mites, suggesting an additional host shift. Finally, additional microsatellite data showed evidence of a potential host shift from V. jacobsoni to the recently imported A. mellifera colonies in New Guinea (Roberts et al. 2015) and Thailand (Dietemann et al. 2019). In total, this constitutes four host shift events of Varroa mites from their native A. cerana to A. mellifera. Interestingly, according to these studies, other mitotypes of V. destructor (i.e., the China 1 to China 3, or C1–C3; and Vietnam 1, or V1, mitotypes) are apparently restricted to the original host, A. cerana, suggesting that Varroa mites of different mitotypes are geographically isolated and host-specific, i.e., only Varroa mites of specific mitotypes can shift to A. mellifera (Beaurepaire et al. 2015; Navajas et al. 2010; Traynor et al. 2020). Current apiculture management plans are typically formulated upon this basis, ignoring the potential for hybridization between local Varroa mites and imported Varroa mites (except Dietemann et al. 2019). Assessing possible gene flow and hybridization among populations of vectors is therefore a crucial step toward developing a comprehensive management plan.
The risk of hybridization is particularly pertinent in Asia, where natural and farmed A. cerana co-occur with apicultural A. mellifera, raising the possibility for hybridized Varroa populations to spill back to local hosts with potentially catastrophic effects for apiculture and the local honeybee community. Recent work using microsatellite DNA markers on V. destructor in Thailand (Dietemann et al. 2019) has called into question the genetic isolation of mitotypes, instead suggesting that host shifts and introgression between Varroa species have led to lability in genetic structure and host specificity (Dietemann et al. 2019). Thus, while mitochondrial DNA data suggest that Varroa mites with different mitotypes are specialized on different hosts and restricted to particular geographic areas, nuclear microsatellite data suggest that hybridization may be occurring between invasive and local Varroa mites. This discrepancy between mitochondrial and nuclear data raises the question of whether these patterns are unique to Thailand populations or indicative of a general phenomenon in Asian apiculture (Dietemann et al. 2019). To assess this potential threat and develop strategies for parasite control, knowledge of the genetic structure of Varroa mite communities in Asian bee farms and an evaluation of the degree of hybridization and/or host specificity among Varroa strains is essential (Criscione et al. 2006; Meyers et al. 2017). Next-generation sequencing approaches can provide high-resolution genetic information suitable for addressing these questions precisely, yet these techniques have not been applied to the dynamics of population genetic structures of Varroa mites parasitizing A. cerana and A. mellifera.
Here, we use genome-wide single-nucleotide polymorphism data (SNPs) generated from restriction-site associated DNA sequencing (RAD-seq, Andolfatto et al. 2011) to elucidate the population genetic structure and potential for hybridization among V. destructor populations in Vietnam and Taiwan. Colonies of A. cerana are often kept in close proximity to those of A. mellifera in honeybee farms in Vietnam, making this an area of high hybridization potential. Previous studies indicate that Varroa mites in Vietnam were the V1, C1, and K1 mitotypes (Navajas et al. 2010) while those in Taiwan belonged to the J1 type (Navajas et al. 2010), but no studies have explored genetic structure among these populations using genomic data. If host specialization has been established, we predict the genomic data to align with defined mitotypes, thus supporting results from previous studies in Asia (Beaurepaire et al. 2015; Li et al. 2019; Navajas et al. 2010; Traynor et al. 2020). Alternatively, if host shifts are ongoing, we expect Varroa mite genotypes to be largely governed by the local mite communities, with observable hybridization among nearby areas even if they are different mitotypes, indicative of gene flow between Varroa mites of different source populations.
2 Materials and methods
2.1 Taxon sampling
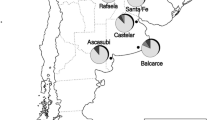
We collected 56 V. destructor from 18 sampling sites in Vietnam and Taiwan (Figure 1 and Supplementary Table S1). Fourteen V. destructor were collected from A. mellifera colonies from six sites in Vietnam, which included Thi tran. Luong Son, Luong Son (LSTT); Cat Que, Hoai Duc (HDCQ); Thuy Phuong, Tu Liem (TLTP); and Ia Ko, Chu Se (CSIK1, CSIK2, and CSIK3). Twenty-nine V. destructor were collected from A. cerana colonies from sites in Vietnam, which included Yen Bai, Ba Vi (BVYB); Van Hoa, Ba Vi (BVVH1 and BVVH2); Mong Hoa, Ky Son (KSMH1, KSMH2, and KSMH3); My Dinh, Tu Liem (TLMD); Phuong Trung, Thanh Oai (TOPT); and Thach Hoa, Thach That (TTTH1 and TTTH2). Thirteen V. destructor were collected from A. mellifera colonies in Tainan (TN) and Kaohsiung (KN), in Taiwan. For each colony, we sampled Varroa mites from the brood cells of drones. Specimens were stored in 95% EtOH at − 20 °C. The collections from Vietnam were housed at Kaohsiung Medical University on a long-term loan from the National Institute Animal of Science, Vietnam.
Collecting sites of Varroa mites in Vietnam and the result of TCS mitotype network analysis. Collecting localities are shown on a broad scale in A & B. The map is collecting sites of Varroa mites in Vietnam. The legend shows the host species of each site. The blue dots show the Varroa mites parasitized A. mellifera. The red dots show the Varroa mites parasitized A. cerana. The TCS network was constructed using COI sequences (C). The gray shading blocks show the mitotypes of Varroa mites from previous studies. Each circle shows the samples collected from different localities. These results indicate that Varroa mites collected from A. cerana colonies in Vietnam (green circles) are the Vietnam 1 (V1) mitotype; the Varroa mites collected from A. mellifera colonies in Vietnam (light orange circles) are the Korea 1 (K1) mitotype; the Varroa mites (yellow circles) collected from A. mellifera colonies in Taiwan are the Japan 1 (J1) mitotype. There are three Varroa mites collected from A. cerana colonies in Vietnam which are isolated from the V1 mitotype and closer to the Laos 1 (L1) mitotype (KSMH2 & KSMH3 mitotype, red box A).
2.2 Laboratory genomic work
2.2.1 DNA extraction of V. destructor
We extracted the DNA from the whole body of V. destructor using a Maxwell RSC Blood DNA Kit (Promega, USA) individually. We incubated the tissue with 300-μl lysis buffer and 30 μl proteinase K at 56 °C for 2 h according to the manufacturer’s protocol. Samples were then put into a Maxwell RSC Instrument (Promega, USA) for DNA extraction. We used the QuantiFluor dsDNA System (Promega, USA) for DNA quantification.
2.2.2 Mitochondrial DNA amplification and sequencing
A 929 base pair fragment of the mitochondrial cytochrome C oxidase subunit I gene (COI) was amplified using 10KbCOIF1 forward primer (5’-CTT GTA ATC ATA AGG ATA TTG GAA C-3’) and 6,5KbCOIR reverse primer (5’-AAT ACC AGT GGG AAC CGC-3’) following Navajas et al. (2010) from each DNA sample. Polymerase chain reactions (PCR) were conducted in a total volume of 25 μl, composed of 12.5 μl 2 × Taq Master Mix (Vazyme, China), 8.5 μl pure water, 1 μl of each primer (10 μM concentration), and 2 μl of DNA solution. The following temperature cycle was used: 94 °C for 4 min for pre-denaturation; 35 cycles of amplification at 94 °C for 30 s, annealing at 50 °C for 30 s, and elongation at 72 °C for 1 min; 72 °C for 7 min for the final extension. Products of PCR were subsequently analyzed on a 3730xl sequencer (Applied Biosystems, USA).
2.2.3 Restriction-site associated DNA sequencing library preparation
To generate genome-wide SNPs, we followed the Multiplex Shotgun Genotyping (MSG, Andolfatto et al. 2011) protocol with in-house modification for conducting high-throughput DNA sequencing libraries. We used 25 ng DNA per sample for the RAD-seq library preparation. The sample DNA was digested by restriction enzyme MseI (3U per reaction, total 20 μl, in CutSmart buffer, NEB, USA) at 37 °C for 3 h, and inactivated at 65 °C for 20 min in a TurboCycler 2 (Blue-Ray Biotech, Taiwan). The solution was further ligated by T4 ligase (400U per reaction, total 50 μl, in T4 ligase buffer, NEB, USA) with five-base pair barcode adaptors at 16 °C for 3 h, and inactivated at 65 °C for 10 min in a TurboCycler 2. After digestion and ligation, we mixed all samples to conduct size selection in Pippin Prep (Sage Science, USA) with a 1.5% agarose gel cassette (size range from 350 to 450 bp, Sage Science, USA). We amplified DNA fragments using a Q5 High-Fidelity PCR Kit (total 50 μl, NEB, USA) with two forward primers, FC1.13 (5’-CAA GCA GAA GAC GGC ATA CGA GAT CAG ATC GTG ACT GGA GTT CAG ACG TGT GCT CTT CCG ATC T-3’) and FC1.14 (5’-CAA GCA GAA GAC GGC ATA CGA GAT ACT TGA GTG ACT GGA GTT CAG ACG TGT GCT CTT CCG ATC T-3’), and one reverse primer FC2 (5’-AAT GAT ACG GCG ACC ACC GAG ATC TAC ACT CTT TCC CTA CAC GAC GCT CTT CCG ATC T-3’), then purified the DNA solution using KAPA Pure Beads (Roche, USA). The DNA libraries were sequenced on a NovaSeq 6000 System using a 2 × 150 bp SP kit (Illumina, USA).
2.2.4 Bioinformatic treatments
COI data were aligned and assembled using Geneious 8.1.9 (Kearse et al. 2012) under default settings. Forward and reverse reads were assembled into a consensus sequence, and then aligned against the mitotype sequences of V. destructor and V. jacobsoni from a previous study (Navajas et al. 2010) to form a 856-bp COI data matrix. The information and accession numbers of mitotypes and COI data are shown in Supplementary Table S2 and S3. The COI data were converted into STRUCTURE (Pritchard et al. 2000) format for population genetic structure analysis, and into VCF format (comprising 11 SNPs) for discriminant analysis of principal components (DAPC), using PGDSpider v 2.1.1.5 (Lischer and Excoffier 2012).
RAD-seq data from 56 Varroa mites were assembled and filtered using the five main steps in iPyRAD v0.9.10 (Eaton and Overcast 2016). We demultiplexed the RAD-seq data using the paired-end Genotyping-By-Sequencing (pairgbs) approach without allowing barcode mismatch. During the filtering step, we allowed up to five low-quality bases in a read. Then, we used BWA (Li and Durbin 2009) and BEDTools (Quinlan and Hall 2010) to map reads to the reference genome of V. destructor (GenBank assembly accession: GCA_002443255.1; Techer et al. 2019). We set the parameters of mapping as the reads with > 80% sequence similarity, the removal of reads with more than 5% Ns, and the minimum depth of majority rule base calling as 6 × reads coverage. We then used the same clustering settings to cluster reads across individuals using MUSCLE (Edgar 2004). Finally, we filtered and formatted output files using the default settings in iPyRAD with 75% completeness per locus. To quantify the genetic differentiation between the Varroa mites in different hosts, we generated a new dataset after dropping the samples collected from Taiwan (which were all collected from A. mellifera colonies), leaving 43 Vietnam samples, which were filtered with the same parameter settings. We subsequently repeated this process after dropping the mites which parasitized A. mellifera, to clarify the genetic structure between Varroa mites from the same host. The data from the remaining 29 individuals from Vietnamese A. cerana were filtered under the same parameter settings.
2.3 Data analyses
2.3.1 Construction of COI gene mitotype networks
We used TCS v1.23 (Clement et al. 2000) to construct a COI mitotype network. This analysis was fixed at a connection limit of 50 steps, and gaps in sequences were treated as a fifth state. TCS results were visualized with TCS beautifier (tcsBU) (Múrias dos Santos et al. 2016). In our analysis, we included the 56 Varroa mite samples in this study and seven mitotypes from previous studies: China 1 (C1), China 2 (C2), China 3 (C3), Korea 1 (K1), Vietnam 1 (V1), and Japan 1 (J1) mitotypes of V. destructor; and the Laos 1 (L1) mitotype of V. jacobsoni (Navajas et al. 2010).
2.3.2 Analyses of population genetic structure of Varroa populations
We used STRUCTURE v2.3.4 (Pritchard et al. 2000) to infer population genetic structure and ancestral clusters for all populations. We conducted separate analyses using the COI sequences, and three datasets from RAD-seq data: one for all 56 mites, one without mites from Taiwan, and one including only mites from A. cerana. Analyses were performed for a hypothesized number of ancestries (K) between one and 10. Each K was repeated 10 times with the MCMC chain length of 5 × 104 after a 5 × 104 burn-in. The most likely value of K was determined using the Delta K approach (Evanno et al. 2005) on the CLUMPAK website (Kopelman et al. 2015, Supplementary Figure S1).
2.3.3 Discriminant analysis of principal components and clustering analysis
We used R v3.6.2 (R Core Team 2013) and packages “pegas” and “adegenet” 2.1.1 (Jombart 2008; Jombart and Collins 2015) to conduct a DAPC with the SNPs of COI and RAD-seq data. Due to the unclear patterns in the Bayesian information criterion (BIC) values for the COI SNPs data (Supplementary Figure S2A), we used TCS and Assemble Species by Automatic Partitioning (ASAP, Puillandre et al. 2021) results to infer the optimal cluster number. The ASAP analysis generates species partition hypotheses based on genetic distance (Tamura et al. 2021) and indicated that partitioning into four species were the most likely hypotheses (Supplementary Table S4, Supplementary Figure S3). We applied four clusters to the DAPC analysis according to the TCS results and the best hypothesis of ASAP. For the RAD-seq data, the optimal number of clusters for the 56 samples was determined through DAPC analysis based on the Bayesian information criterion (BIC, Supplementary Figure S2B) while retaining 25 principal components.
2.3.4 Dispersal map of resistance of Varroa mites in different hosts
To investigate the relationship between the geographical and genetic distance of mites, we constructed a dispersal map of resistance using the R package “ResDisMapper” v1.0 (Tang et al. 2019) using the reassembled RAD-seq dataset of 29 Varroa mites in northern Vietnam, dropping the samples from southern Vietnam and Taiwan for fine-scale geographical and genetic analysis. We appraised the genetic and geographical distance using Edward’s angular distance method (Edwards 1971). The landscape was divided into 50 cells based on the World Geodetic System 1984 (WGS 84) coordinate system to assign the isolation by distance (IBD) residuals in the 3-D plot. ResDisMapper calculates IBD residuals using the difference of RAD-seq SNPs and geographical data for each pair of Varroa mite populations. ResDisMapper then projects these IBD residuals onto grid cells according to geographical information. IBD residuals of each collecting site were further calculated as the significant differences between the resistance values under 95% confidence intervals. The contours of significantly different resistance values were constructed based on a p-value of 0.05.
2.3.5 Fixation index between Varroa populations
We used R v3.6.2 package “StAMPP” (Pembleton et al. 2013) to calculate the pairwise genetic difference (Fst) between Varroa mite populations based on the RAD-seq data of 56 Varroa mites from all 18 sites. We calculated the pairwise Fst using 1000 randomized bootstrapping iterations to infer 95% confidence intervals and p-values to assess significant differences of pairwise Fst among populations.
3 Results
3.1 RAD-seq data assembly
The RAD-seq data was demultiplexed and assembled using a reference-based approach. Following filtering of 216,840 loci, this resulted in a genomic dataset composed of genetic profiles of 56 Varroa mites collected from Vietnam and Taiwan, which consist of 1260 loci and 6322 SNPs. For the dataset of 43 Vietnam samples, 216,874 loci were obtained, yielding 1540 loci and 5829 SNPs after filtering. The RAD-seq data of 29 individuals of A. cerana in Vietnam comprised 216,212 loci, which yielded 4766 SNPs in 1575 loci after filtering.
3.2 TCS COI mitotype networks
The mitotype network results indicate that Varroa mites of Vietnam were mostly comprised of V1 (n = 26) and K1 (n = 14) mitotypes, whereas Varroa mites of Taiwan (n = 13) were all J1 mitotypes (Figure 1C). Three Varroa mites collected from A. cerana in Vietnam were isolated from the mitotype network of V. destructor, and were closely related to the V. jacobsoni (L1 mitotype, shown in the red box A in Figure 1C). The Vietnamese mitotypes were also divided among host species: mites collected from A. cerana were V1 mitotypes, while those collected from A. mellifera were K1 mitotypes. Accordingly, the results of TCS analysis showed that most Varroa mites are V. destructor according to their mitotypes. The individuals in the red box A, which were not assigned to any V. destructor or V. jacobsoni mitotypes, were revealed as a novel mitotype (namely KSMH2 & KSMH3 mitotype).
3.3 Population genetic structure inferred from mitochondrial and genomic data
We analyzed the population genetic structure of Varroa mites partitioned by country of collection and by host species (Figure 2). The STRUCTURE results for both COI and RAD-seq datasets indicate that the 56 individuals from Vietnam and Taiwan were most likely derived from two ancestral lineages (an optimal value of K = 2 was resolved, Supplementary Figure S1). Varroa mites that parasitized A. mellifera shared the same pure lineage regardless of which country they were collected from (Figure 2A-C). The Varroa mites that parasitized A. cerana shared a second pure lineage, except for three individuals collected from KSMH2 and KSMH3 which were identified as the same ancestral lineage as the Varroa mites which parasitized A. mellifera. These three individuals are the same as those comprising the isolated group (Figure 1C, KSMH2 & KSMH3 mitotype) in the TCS mitotype network (Figure 2A-D).
Population genetic structure analyses of Varroa mites. The optimal K number in the STRUCTURE analyses is shown at the top of each analysis. Different colors indicate the genetic structure of different ancestors in each column. Hosts and countries of Varroa mites are shown at the bottom of the figure. The population genetic structure of COI data is shown in the first row (A). Note that three individuals of KSMH2 & KSMH3 mitotype show a different pattern of genetic structure in the population of Varroa mites collected from A. cerana in Vietnam. B, C, & D show the population genetic structures of Varroa mites populations using RAD-seq data, indicating the same pattern as the COI results. The 56 Varroa mites from Taiwan and Vietnam are shown in B, while the 43 Varroa mites which parasitized A. mellifera and A. cerana in Vietnam are shown in C, and the 29 Varroa mites from A. cerana are shown in D.
3.4 DAPC and ASAP of Varroa mites
The DAPC based on the SNP matrix generated from COI sequences (11 SNPs) shows that there are four clusters in the 56 Varroa mites from all sites (Figure 3A). Three of these clusters (Figure 3A, gray-shaded areas i, ii, and iv) match the country of collection and host divisions, agreeing with the TCS mitotype analysis. The other cluster (iii) comprises Varroa mites parasitizing A. cerana from KSMH2 and KSMH3 (KSMH2 & KSMH3 mitotype) and supports the result of the STRUCTURE analysis in that these three individuals are more closely related to the Varroa mites (collected from A. cerana) parasitizing A. mellifera in Vietnam. Similar to the clustering of DAPC results, the ASAP results also show the four-cluster hypothesis was the best hypothesis (Figure S3 and Table S4).
The result of discriminant analysis of principal components (DAPC) of mitochondrial (A) and genomic (B) data. The legend shows the individuals corresponding to each collection site. Each point represents one individual. Individuals collected from Taiwan are shown in yellow; individuals collected from Vietnam are shown in blue (collected from A. mellifera colonies) and red (collected from A. cerana colonies). The DAPC result based on COI data (A) shows that the 56 individuals are assigned to four clusters (the shaded blocks in gray, clusters i, ii, iii, and iv). The DAPC result using RAD-seq data (B) shows that the 56 individuals are assigned to three clusters. Two clusters were highly associated with host species (clusters i and iii). One cluster is composed of multiple localities, hosts, or countries (cluster ii).
In contrast to the DAPC based on COI data, the DAPC analysis using RAD-seq data indicated considerable overlap between locations and hosts, and the splitting of some sites between clusters. While all individuals could still be assigned to three distinct clusters (Figure 3B, the gray-shaded areas i, ii and iii). Corresponding to host species, individuals were assigned to cluster i, mites collected from A. mellifera, and cluster iii, mites collected from A. cerana. However, cluster ii was mixed, and composed of mites parasitizing A. mellifera and mites parasitizing A. cerana. Among these three clusters, only the Varroa mites from BVYB, KSMH3, TTH2, CSIK2, and CSIK3 show high affinity to others from their own population.
3.5 Dispersal map of resistance between Varroa populations
The dispersal map of resistance of Varroa populations in Vietnam suggests that some gene flow occurred between the mites from different hosts (Figure 4). The orange and green contours show significant levels of resistance to gene flow across the landscape of collection sites in northern Vietnam. We found that there was high resistance between Varroa mites parasitizing A. mellifera in Thuy Phuong, Tu Liem (TLTP) and Cat Que, Hoai Duc (HDCQ), and high resistance between Varroa mites parasitizing A. cerana in Thach Hoa, Thach That (TTTH1 and TTTH2), Mong Hoa, Ky Son (KSMH1, KSMH2, and KSMH3), and Yen Bai (BVYB). On the other hand, significantly low resistance (green contours) was found between some populations of Varroa mites parasitizing A. cerana: (1) My Dinh, Tu Liem (TLMD) and Mong Hoa, Ky Son (KSMH1, KSMH2, and KSMH3); (2) Van Hoa, Ba Vi (BVVH1 and BVVH2), Thach Hoa, Thach That (TTTH1 and TTTH2) and Phuong Trung, Thanh Oai (TOPT). However, we also found low resistance between mites parasitizing A. cerana (TLMD) and A. mellifera (HDCQ and LSTT). These results thus do not indicate any clear pattern of distance-related resistance to gene flow.
Dispersal map of resistance of Varroa mites in Vietnam. The dispersal map of resistance was calculated based on the RAD-seq data. The dispersal map is shown with a topographic map superimposed to show the geographic barriers among our collection sites in Vietnam. Orange blocks indicate higher resistance areas of the landscape while green blocks indicate the lower resistance areas. Orange and green contours indicate boundaries of significantly high or low resistance respectively. Longitude/latitude coordinates of collection sites have been converted to the projected coordinates as shown on the horizontal and vertical axis. The blue dots indicate Varroa mites collected from A. mellifera colonies, and red dots show mites collected from A. cerana colonies.
3.6 Genetic differentiation between Varroa populations
The summarized results of the pairwise fixation indices (Fst) for each collection site are shown in Figure 5. At the country level, the values of Fst between mites in Taiwan (− 0.0085) show low genetic differentiation. The average Fst in Vietnam (mean ± SD: 0.0635 ± 0.0733, range = − 0.1038–0.2781) indicates moderate genetic differentiation between populations. There was moderate differentiation between mite populations in Taiwan and Vietnam (mean Fst = 0.072 ± 0.0343, range = 0.0104–0.1351). However, in both cases, Fst values were highly variable (i.e., had high standard deviations). We therefore focused only on the results of pairwise Fst values among different sites. At the site level, the highest Fst was 0.2781, between Varroa populations of the sample from Yen Bai, Ba Vi (BVYB) and the third sample from Ia Ko, Chu Se (CSIK3) in Vietnam. The only Fst among Varroa populations in Taiwan was − 0.0085. The highest Fst among Varroa populations between Taiwan and Vietnam was 0.1351, which was lower than the highest Fst between BVYB and CSIK3 in Vietnam, indicating that differentiation between local populations can be higher than that between countries. With regard to variation among sites within each of the hosts, the Fst values of the new host, A. mellifera, showed low differentiation (mean = 0.035 ± 0.0554, range = − 0.1038–0.1419. The Fst among sites of the original host, A. cerana, also showed low differentiation (mean = 0.0228 ± 0.0461, range = − 0.0351–0.1683). A comparison of Fst between mites parasitizing A. cerana and A. mellifera, indicated moderate differentiation (mean = 0.0989 ± 0.0626, range = − 0.0073–0.2781). Overall, our analysis of Fst indicates that population differentiation of mites was associated with country and host species, but there were still Fst between hosts, localities, or countries show conflicting genetic relatedness compared to the average value of Fst at each level.
Pairwise fixation index (Fst) for each collection site. The SNPs of RAD-seq data were used to calculate pairwise fixation indexes following Wright (1978). Classification of Fst is shown in a gradient of color from white to red: low genetic differentiation (values lower than 0.05 = white); moderate genetic differentiation (values between 0.05 and 0.15 = coral pink); high genetic differentiation (values between 0.15 and 0.25 = light red) and very high differentiation (values higher than 0.25 = dark red). The p-values are indicated as follows: *0.05 to 0.01; **0.01 to 0.001, and *** ≤ 0.001.
4 Discussion
Our population genomic and genetic analyses of Varroa mites from two Apis hosts in Taiwan and Vietnam produced some contrasting results. While our analyses of mitochondrial data supported the conclusions of several previous studies (Beaurepaire et al. 2015; Navajas et al. 2010; Traynor et al. 2020) that different K1 and J1 mitotypes are specialized to A. mellifera, our STRUCTURE and DAPC analyses (Figures 2B-D and 3B) based on genome-wide SNPs only partially support these patterns. The estimations of gene flow and genetic differentiation (dispersal map of resistance, Figure 4, and Fst, Figure 5) showed that although the gene flow of mites could have been restricted between populations in different countries and different hosts, there are some exceptions indicating evidence of gene flow between mites of different hosts. Overall, our data indicate that there is sufficient evidence of gene flow and hybridization to conclude that Varroa mites have the potential to hybridize even if they parasitize different hosts, or belong to different mitotypes, regardless of patterns of isolation indicated by mitochondrial analyses. As the hybridization of V. destructor is likely ongoing in Vietnam, we suggest that the genetic pool of the local communities of Varroa mites parasitizing A. cerana should be considered a source for potential hybridization with imported mites parasitizing A. mellifera in bee farms that keep both Apis species. Apicultural movements and multi-species farming practices in particular can be expected to intensify hybridization between populations of V. destructor. Compared to analyses of mitochondrial DNA, our genomic data show greater evidence for potential hybridization and indicate that genetic differentiation among populations cannot be explained solely by the host or geographic area.
Hybridization and introgression between different Varroa species have been reported in Thailand (Dietemann et al. 2019) based on analyses of eight microsatellite markers. In contrast, Beaurepaire et al. (2015) reported that data from six microsatellite markers indicated complete genetic isolation of Varroa mites from A. mellifera and A. cerana kept in the same apiaries in North Vietnam and the Philippines. Our mitochondrial and genomic results of the KSMH2 & KSMH3 mitotypes are similar to the case in Thailand, in which Varroa mites showed introgression or hybridization between V. destructor and V. jacobsoni. However, more V. jacobsoni from Vietnam or other geographic areas are needed to assess the degree of interaction between these two parasitic species. By contrasting the STRUCTURE results of COI and RAD-seq data (Figure 2), it becomes clear that the genetic subdivisions of most Varroa populations are strongly associated with hosts rather than that of mitotypes. The V. destructor parasitizing A. mellifera in Vietnam shared the same ancestry as those in Taiwan, even though they were different mitotypes in the TCS result (Figure 1C), which indicates that the K1 mitotype in Vietnam and J1 mitotype in Taiwan share the same genetic pool. Our DAPC results of RAD-seq data showed that the clustering is likely influenced by multiple factors and cannot be explained by country, host species, or site, as suggested by the DPAC of COI data. While two clusters in the DAPC of RAD-Seq data were strongly associated with hosts, the third cluster was comprised of V. destructor of a mixture of different hosts and countries. Indeed, of all individuals collected from A. cerana, only those from the BVYB and TTTH2 colonies could all be allocated to a pure A. cerana cluster. Most individuals from A. cerana colonies show mixture according to the major components of genome.
Our dispersal map of resistance analyses and pairwise Fst results also showed a low differentiation between some populations from different hosts or countries. We infer that this can be attributed to the apicultural trade, as beekeepers in search of resources for beekeeping must move colonies seasonally in our study areas. These apicultural movements also result in a relocation of diseases and/or parasites of honeybees (Gordon et al. 2014). Our dispersal map of resistance indicates that geographic barriers could be associated with genetic distances between populations, especially for Varroa mites from the hills (Figure 4). However, there were significantly low genetic resistances between populations parasitizing in different hosts or populations separated by long distances. The Fst of Varroa mites indicated moderate differentiation between the populations between countries or from different hosts, but the Fst at the site level indicated that there was variation in the degree of gene flow, i.e., between mite populations in Vietnam (Figure 5). Not all colonies in different sites exhibited patterns which might be expected from isolation of host species and location. There were some colonies showed low differentiation even though they parasitized different hosts, which may be an indication of host shift and hybridization of different Varroa mites. These patterns of genetic structure boundaries among Varroa mites indicate continued genome mixing with gene flow. This has the potential to facilitate transmission among different hosts.
5 Conclusions
Quantifying the risk of host shifts and hybridization between Varroa mites is key to formulating effective control strategies. Our results show the potential for V. destructor of V1, J1, and K1 mitotypes to infest multiple hosts. Potential new sources of Varroa mites, such as hybrid Varroa mites, could thus be a major threat to the apicultural industry. If beekeepers continue management practices that consider these populations as a single gene pool, as suggested by previous studies of mitotypes (Beaurepaire et al. 2015; Navajas et al. 2010; Traynor et al. 2020), interbreeding among local Varroa mite communities composed of foreign and native Varroa mites may represent an unmanaged biological time-bomb. Furthermore, the potential impacts of this hybridization risk extend beyond increased decimation of A. mellifera, as the high diversity of these populations may also lead to back-transmission to its original host, A. cerana, resulting in the decimation of the native bee community. We conclude that these local communities of V. destructor in A. cerana, which are connected by those apicultural activities and infesting between colonies in different areas, are key to effective parasite control in apicultural management, highlighting the importance of the periodic survey and genotyping of Varroa mite populations using genome-wide approaches.
Data availability
Data are available in supplementary information.
Code availability
Not applicable.
References
Anderson D, Trueman J (2000) Varroa jacobsoni (Acari: Varroidae) is more than one species. Exp Appl Acarol 24:165–189
Andolfatto P, Davison D, Erezyilmaz D, Hu TT, Mast J, Sunayama-Morita T, Stern DL (2011) Multiplexed shotgun genotyping for rapid and efficient genetic mapping. Genome Res 21:610–617
Beaurepaire AL, Truong TA, Fajardo AC, Dinh TQ, Cervancia C, Moritz RF (2015) Host specificity in the honeybee parasitic mite, Varroa spp. in Apis mellifera and Apis cerana. PloS One 10.8:e0135103
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659
Crane E (1978) Beekeeping round the world. Bee World 59:164–167
Criscione CD, Cooper B, Blouin MS (2006) Parasite genotypes identify source populations of migratory fish more accurately than fish genotypes. Ecology 87:823–828
Dietemann V, Beaurepaire A, Page P, Yañez O, Buawangpong N, Chantawannakul P, Neumann P (2019) Population genetics of ectoparasitic mites Varroa spp. in Eastern and Western honey bees. Parasitology 146:1429–1439
Eaton DA, Overcast I (2016) ipyrad: interactive assembly and analysis of RADseq datasets. Bioinformatics 36:2592–2594
Edgar RC (2004) MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 32:1792–1797
Edwards AWF (1971) Distances between populations on the basis of gene frequencies. Biometrics 873–881
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14:2611–2620
Genersch E, Von Der Ohe W, Kaatz H, Schroeder A, Otten C, Büchler R, Berg S, Ritter W, Mühlen W, Gisder S (2010) The German bee monitoring project: a long term study to understand periodically high winter losses of honey bee colonies. Apidologie 41:332–352
Gordon R, Bresolin-Schott N, East I (2014) Nomadic beekeeper movements create the potential for widespread disease in the honeybee industry. Aust Vet J 92:283–290
Jombart T (2008) Adegenet: a R package for the multivariate analysis of genetic markers. Bioinformatics 24:1403–1405
Jombart T, Collins C (2015) A tutorial for discriminant analysis of principal components (DAPC) using adegenet 2.0. 0. UK: Imperial College London. MRC Centre for Outbreak Analysis and Modelling 43:1–43
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C (2012) Geneious Basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
King KC, Stelkens RB, Webster JP, Smith DF, Brockhurst MA (2015) Hybridization in parasites: consequences for adaptive evolution, pathogenesis, and public health in a changing world. PLOS Pathog 11(9):e1005098
Kopelman NM, Mayzel J, Jakobsson M, Rosenberg NA, Mayrose I (2015) Clumpak: a program for identifying clustering modes and packaging population structure inferences across K. Mol Ecol Resour 15:1179–1191
Li H, Durbin R (2009) Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 25(14):1754–1760
Li W, Wang C, Huang ZY, Chen Y, Han R (2019) Reproduction of distinct Varroa destructor genotypes on honey bee worker brood. Insects 10:372
Lischer HE, Excoffier L (2012) PGDSpider: an automated data conversion tool for connecting population genetics and genomics programs. Bioinformatics 28:298–299
Meyers AC, Meinders M, Hamer SA (2017) Widespread Trypanosoma cruzi infection in government working dogs along the Texas-Mexico border: discordant serology, parasite genotyping and associated vectors. PLoS Negl Trop Dis 11:e0005819
Múrias dos Santos A, Cabezas MP, Tavares AI, Xavier R, Branco M (2016) tcsBU: a tool to extend TCS network layout and visualization. Bioinformatics 32:627–628
Navajas M, Anderson DL, De Guzman LI, Huang ZY, Clement J, Zhou T, Le Conte Y (2010) New Asian types of Varroa destructor: a potential new threat for world apiculture. Apidologie 41:181–193
Pembleton LW, Cogan NO, Forster JW (2013) StAMPP: an R package for calculation of genetic differentiation and structure of mixed-ploidy level populations. Mol Ecol Resour 13:946–952
Peng YS, Fang Y, Xu S, Ge L (1987) The resistance mechanism of the Asian honey bee, Apis cerana Fabr., to an ectoparasitic mite, Varroa jacobsoni Oudemans. J Invertebr Pathol 49:54–60
Pennisi E (2018) Hybridization may give some parasites a leg up. Science 361:832–833
Pritchard JK, Stephens M, Donnelly P (2000) Inference of population structure using multilocus genotype data. Genetics 155:945–959
Puillandre N, Brouillet S, Achaz G (2021) ASAP: assemble species by automatic partitioning. Mol Ecol Resour 21.2:609–620. https://bioinfo.mnhn.fr/abi/public/asap/. Accessed 9 Jun 2022
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26(6):841–842
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for statistical computing, Vienna, Austria
Roberts J, Anderson D, Tay W (2015) Multiple host shifts by the emerging honeybee parasite, Varroa jacobsoni. Mol Ecol 24:2379–2391
Rosenkranz P, Aumeier P, Ziegelmann B (2010) Biology and control of Varroa destructor. J Invertebr Pathol 103:S96–S119
Sakai T, Okada I (1973) Present beekeeping in Japan. Gleanings in Bee Culture 101:356–357
Schelkle B, Faria PJ, Johnson MB, van Oosterhout C, Cable J (2012) Mixed infections and hybridisation in monogenean parasites. PLoS ONE 7(7):e39506
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38(7):3022–3027
Tang Q, Fung T, Rheindt FE (2019) ResDisMapper: an r package for fine-scale mapping of resistance to dispersal. Mol Ecol Resour 20:819–831
Techer MA, Rane RV, Grau ML, Roberts JM, Sullivan ST, Liachko I, Childers AK, Evans JD, Mikheyev AS (2019) Divergent evolutionary trajectories following speciation in two ectoparasitic honey bee mites. Commun Biol 2(1):1–16
Traynor KS, Mondet F, de Miranda JR, Techer M, Kowallik V, Oddie MA, Chantawannakul P, McAfee A (2020) Varroa destructor: a complex parasite, crippling honey bees worldwide. Trends Parasitol 36:592–606
Wright S (1978) Evolution and the genetics of populations: a treatise in four volumes: Vol. 4: variability within and among natural populations. University of Chicago Press
Zhang ZQ (2000) Notes on Varroa destructor (Acari: Varroidae) parasitic on honeybees in New Zealand. Syst Appl Acarol Spec Publ 5:9–14
Acknowledgements
We thank Mr. Bing-Heng Lee, Dr. Wei-Yu Chen, and members of Ecology and Evolutionary Genomics laboratory at the Kaohsiung Medical University for assistance in this research. We thank National Institute Animal of Science, Vietnam, for issuing sample loaning and Mr. Thanh Nguyen Thong for his contribution to fieldwork in Vietnam.
Funding
This work was supported by the Ministry of Science and Technology (MOST), Taiwan funding to Y. C. S. (funding numbers MOST 107–2321-B-168–001 (Co-PI); MOST 110–2621-B-037–001-MY3).
Author information
Authors and Affiliations
Contributions
Y. F. C. handled fieldwork, lab work, analyses, and writing. Y. C. S. formed the project, lead writing, fieldwork, and analyses. T. T. H. N. and P. T. Y. did fieldwork. P. P., A. L. C., and Y. C. S. assisted in analyses and revisions.
Corresponding author
Ethics declarations
Ethics approval
National Institute Animal of Science, Vietnam issued loan samples from Vietnam. Samples from Taiwan were collected under the local regulation of farmed insects.
Consent to participate
All authors have read and approved the submitted manuscript in the current form.
Consent for publication
This manuscript is original unpublished work, which has not been accepted and is not under consideration for publication elsewhere.
Conflict of interest
The authors declare no competing interests.
Additional information
Manuscript editor: Zachary Huang
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chiu, YF., Nguyen, T.T.H., Yeh, PT. et al. Genome-wide SNPs show hybridization of Varroa mites from different Apis hosts in Vietnam and Taiwan. Apidologie 54, 25 (2023). https://doi.org/10.1007/s13592-023-01001-3
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13592-023-01001-3