Abstract
Selecting the best treatment for individual patients with cancer has attracted attention for improving clinical outcomes. Recent progress in organoid culture may lead to the development of personalized medicine. Unlike molecular-targeting drugs, there are no predictive methods for patient response to standard chemotherapies for ovarian cancer. We prepared organoids using the cancer tissue-originated spheroid (CTOS) method from 61 patients with ovarian cancer with 100% success rate. Chemosensitivity assays for paclitaxel and carboplatin were performed with 84% success rate using the primary organoids from 50 patients who received the chemotherapy. A wide range of sensitivities was observed among organoids for both drugs. All four clinically resistant organoids were resistant to both drugs in 18 cases in which clinical response information was available. Five out of 18 cases (28%) were double-resistant, the response rate of which was compatible with the clinical remission rate. Carboplatin was significantly more sensitive in serous than in clear cell subtypes (P = 0.025). We generated two lines of organoids, screened 1135 drugs, and found several drugs with better combinatory effects with carboplatin than with paclitaxel. Some drugs, including afatinib, have shown an additive effect with carboplatin. The organoid sensitivity assay did not predict the clinical outcomes, both progression free and overall survival.






Similar content being viewed by others
Data availability
The organoid lines and data are available from the corresponding author upon reasonable request.
References
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7–34.
du Bois A, Luck HJ, Meier W, et al. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst. 2003;95:1320–9.
Grendys EC Jr, Fiorica JV, Orr JW Jr, et al. Overview of a chemoresponse assay in ovarian cancer. Clin Transl Oncol. 2014;16:761–9.
Dugger SA, Platt A, Goldstein DB. Drug development in the era of precision medicine. Nat Rev Drug Discovery. 2018;17:183–96.
Cree IA, Kurbacher CM, Lamont A, Hindley AC, Love S, Group TCAOCT. A prospective randomized controlled trial of tumour chemosensitivity assay directed chemotherapy versus physician’s choice in patients with recurrent platinum-resistant ovarian cancer. Anticancer Drugs. 2007;18:1093–101.
Burstein HJ, Mangu PB, Somerfield MR, et al. American society of clinical oncology clinical practice guideline update on the use of chemotherapy sensitivity and resistance assays. J Clin Oncol. 2011;29:3328–30.
Fruehauf JP. In vitro assay-assisted treatment selection for women with breast or ovarian cancer. Endocr Relat Cancer. 2002;9:171–82.
Drost J, Clevers H. Organoids in cancer research. Nat Rev Cancer. 2018;18:407–18.
Kondo J, Endo H, Okuyama H, et al. Retaining cell–cell contact enables preparation and culture of spheroids composed of pure primary cancer cells from colorectal cancer. PNAS. 2011;108:6235–40.
Tashiro T, Okuyama H, Endo H, et al. In vivo and ex vivo cetuximab sensitivity assay using three-dimensional primary culture system to stratify KRAS mutant colorectal cancer. PLoS ONE. 2017;12: e0174151.
Kondo J, Ekawa T, Endo H, et al. High-throughput screening in colorectal cancer tissue-originated spheroids. Cancer Sci. 2019;110:345–55.
Endo H, Okami J, Okuyama H, et al. Spheroid culture of primary lung cancer cells with neuregulin 1/HER3 pathway activation. J Thorac Oncol. 2013;8:131–9.
Yoshida T, Okuyama H, Nakayama M, et al. High-dose chemotherapeutics of intravesical chemotherapy rapidly induce mitochondrial dysfunction in bladder cancer-derived spheroids. Cancer Sci. 2015;106:69–77.
Kiyohara Y, Yoshino K, Kubota S, et al. Drug screening and grouping by sensitivity with a panel of primary cultured cancer spheroids derived from endometrial cancer. Cancer Sci. 2016;107:452–60.
Nakajima A, Endo H, Okuyama H, et al. Radiation sensitivity assay with a panel of patient-derived spheroids of small cell carcinoma of the cervix. Int J Cancer. 2015;136:2949–60.
Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228–47.
Nagase S, Ohta T, Takahashi F, Yaegashi N. Board members of the committee on gynecologic oncology of the japan society of O, gynecology. annual report of the committee on gynecologic oncology, the japan society of obstetrics and gynecology: annual patient report for 2017 and annual treatment report for 2012. J Obstet Gynaecol Res. 2021;47:1631–42.
Tewari KS, Burger RA, Enserro D, et al. Final overall survival of a randomized trial of bevacizumab for primary treatment of ovarian cancer. J Clin Oncol. 2019;37:2317–28.
Ishiguro T, Sato A, Ohata H, et al. Establishment and characterization of an in vitro model of ovarian cancer stem-like cells with an enhanced proliferative capacity. Cancer Res. 2016;76:150–60.
Hill SJ, Decker B, Roberts EA, et al. Prediction of DNA repair inhibitor response in short-term patient-derived ovarian cancer organoids. Cancer Discov. 2018;8:1404–21.
Kopper O, de Witte CJ, Lohmussaar K, et al. An organoid platform for ovarian cancer captures intra- and interpatient heterogeneity. Nat Med. 2019;25:838–49.
Phan N, Hong JJ, Tofig B, et al. A simple high-throughput approach identifies actionable drug sensitivities in patient-derived tumor organoids. Commun Biol. 2019;2:78.
Maru Y, Tanaka N, Itami M, Hippo Y. Efficient use of patient-derived organoids as a preclinical model for gynecologic tumors. Gynecol Oncol. 2019;154:189–98.
de Witte CJ, Espejo Valle-Inclan J, Hami N, et al. Patient-derived ovarian cancer organoids mimic clinical response and exhibit heterogeneous inter- and intrapatient drug responses. Cell Rep. 2020;31: 107762.
Nanki Y, Chiyoda T, Hirasawa A, et al. Patient-derived ovarian cancer organoids capture the genomic profiles of primary tumours applicable for drug sensitivity and resistance testing. Sci Rep. 2020;10:12581.
Maenhoudt N, Defraye C, Boretto M, et al. Developing organoids from ovarian cancer as experimental and preclinical models. Stem Cell Reports. 2020;14:717–29.
Hoffmann K, Berger H, Kulbe H, et al. Stable expansion of high-grade serous ovarian cancer organoids requires a low-Wnt environment. EMBO J. 2020;39: e104013.
Weeber F, Ooft SN, Dijkstra KK, Voest EE. Tumor organoids as a pre-clinical cancer model for drug discovery. Cell Chem Biol. 2017;24:1092–100.
Makinen L, Vaha-Koskela M, Juusola M, et al. Pancreatic cancer organoids in the field of precision medicine: a review of literature and experience on drug sensitivity testing with multiple readouts and synergy scoring. Cancers. 2022. https://doi.org/10.3390/cancers14030525.
Sun H, Wang H, Wang X, et al. Aurora-A/SOX8/FOXK1 signaling axis promotes chemoresistance via suppression of cell senescence and induction of glucose metabolism in ovarian cancer organoids and cells. Theranostics. 2020;10:6928–45.
Ozols RF, Bundy BN, Greer BE, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a gynecologic oncology group study. J Clin Oncol. 2003;21:3194–200.
Thigpen JT, Blessing JA, Ball H, Hummel SJ, Barrett RJ. Phase II trial of paclitaxel in patients with progressive ovarian carcinoma after platinum-based chemotherapy: a gynecologic oncology group study. J Clin Oncol. 1994;12:1748–53.
Sugiyama T, Kamura T, Kigawa J, et al. Clinical characteristics of clear cell carcinoma of the ovary: a distinct histologic type with poor prognosis and resistance to platinum-based chemotherapy. Cancer. 2000;88:2584–9.
Enomoto T, Kumagai S, Yamasaki M. Is clear cell carcinoma and mucinous carcinoma of the ovary sensitive to combination chemotherapy with paclitaxel and carboplatin? Proc Am Soc Clin Oncol. 2003;22:447.
Winter WE 3rd, Maxwell GL, Tian C, et al. Prognostic factors for stage III epithelial ovarian cancer: a gynecologic oncology group study. J Clin Oncol. 2007;25:3621–7.
Berchuck A, Secord AA, Moss HA, Havrilesky LJ. Maintenance poly (ADP-ribose) polymerase inhibitor therapy for ovarian cancer: precision oncology or one size fits all? J Clin Oncol. 2017;35:3999–4002.
Hamanishi J, Mandai M, Ikeda T, et al. Safety and antitumor activity of anti-PD-1 antibody, nivolumab, in patients with platinum-resistant ovarian cancer. J Clin Oncol. 2015;33:4015–22.
Harries M, Gore M. Part II: chemotherapy for epithelial ovarian cancer-treatment of rcurrent disease. Lancet Oncol. 2002;3:537–45.
Wilken JA, Badri T, Cross S, et al. EGFR/HER-targeted therapeutics in ovarian cancer. Future Med Chem. 2012;4:447–69.
Mehner C, Oberg AL, Goergen KM, et al. EGFR as a prognostic biomarker and therapeutic target in ovarian cancer: evaluation of patient cohort and literature review. Genes Cancer. 2017;8:589–99.
Berg T, Nottrup TJ, Roed H. Gemcitabine for recurrent ovarian cancer—a systematic review and meta-analysis. Gynecol Oncol. 2019;155:530–7.
LeBlanc VG, Trinh DL, Aslanpour S, et al. Single-cell landscapes of primary glioblastomas and matched explants and cell lines show variable retention of inter- and intratumor heterogeneity. Cancer Cell. 2022;40(379–92): e9.
Rehman SK, Haynes J, Collignon E, et al. Colorectal cancer cells enter a diapause-like DTP state to survive chemotherapy. Cell. 2021;184(226–42): e21.
Acknowledgements
We thank Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) for providing the drug library.
Funding
This work was supported, in part, by a Grant-in-Aid from P-CREATE, a Japan Agency for Medical Research and Development, Japan, 19cm0106203h0004 (M.I., J.K., K.O.); a Grant-in-Aid from Takeda Science Foundation (M.I.)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M.I. belong and J.K. belonged to the Department of Clinical Bio-resource Research and Development at Kyoto University, which is sponsored by KBBM, Inc. M.I. is an inventor of the patents related to CTOS method.
Ethics approval
This study was approved by the institutional ethics committees of the Osaka International Cancer Institute (1111112098, 1605137104), Osaka University (10182), and Kyoto University (R1575). Animal studies were approved by the Institutional Animal Study Committee of Osaka International Cancer Institute and Kyoto University and performed in compliance with the guidelines.
Informed consent
Surgical specimens and ascites samples from patients with ovarian cancer were obtained from Osaka University and Osaka International Cancer Institute with the approved patients’ informed consent and permission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
13577_2022_827_MOESM1_ESM.xlsx
Supplementary file 1: (XLSX 14 KB) Table S1 Clinical information and results of organoid preparation and xenograft formation in 61 patients in this study.
13577_2022_827_MOESM2_ESM.xlsx
Supplementary file 2: (XLSX 16 KB) Table S2 Information on the clinical response and prognosis of patients and summary of drug testing results.
13577_2022_827_MOESM4_ESM.docx
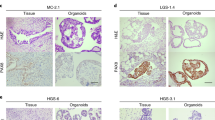
Supplementary file 4: (DOCX 413 KB) Figure S1 Morphological characteristics of the ovarian cancer organoids. Phase-contrast images of primary organoids from the indicated patient tumors. Histological subtypes are shown. Scale bar, 100 \(\upmu\)m. Figure S2 Chemosensitivity assay for paclitaxel and carboplatin using primary organoids. (A) Dose response analysis for paclitaxel and carboplatin. Actual values of the relative ATP values for most sensitive (blue lines) and resistant (red lines) cases to each drug are shown as indicated. SD bars are shown. (B) Scatter plot of the sensitivity assay-ranking with the clinical stage for 42 organoids with which the drug testing was performed. Stage I, blue; stage II, green; stage III orange; stage IV, red. Figure S3 Chemosensitivity assay of the organoid lines, OV114 and OV129. (A, B) The dose response sigmoid curves for paclitaxel (A) and carboplatin (B). The results of the two organoids were added to Figure 2B; OV114 organoids derived from xenografts of passage 2 (px2) and passage 3 (px3), and OV129 organoids derived from primary and xenografts of passage 5 (px5), shown with the indicated colors. Others from Figure 2B are shown with gray. (C) Scatter plot of the sensitivity ranking of Figure 2C adding the data in (A) and (B). These dots are indicated with the indicated colors, and others from Figure 2C are shown with gray. Figure S4 Validation of the hit drugs using the organoid lines. The dose-response curves for candidate drugs other than those shown in Figure 5. The red lines indicate the results for the candidate drugs alone. Blue lines indicate the results of the combination of the candidate drugs with 10 μM carboplatin. SD bars are shown. Figure S5 Effect of the hit drugs in OV101. The organoids of OV101 were prepared from xenograft tumors. (A) The dose response sigmoid curves for carboplatin are from three independent experiments. (B) The results of (A) are added to Figure 2B; each result of the OV101 organoids is shown in red. Others from Figure 2B are shown in gray. (C) In the dose-response curves of OV101 for candidate drugs, the red lines indicate the results for the candidate drugs alone. Blue lines indicate the results of the combination of the candidate drugs with 10 μM carboplatin. SD bars are shown. The experiments were performed 3 times as indicated.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ito, Y., Kondo, J., Masuda, M. et al. Ex vivo chemosensitivity assay using primary ovarian cancer organoids for predicting clinical response and screening effective drugs. Human Cell 36, 752–761 (2023). https://doi.org/10.1007/s13577-022-00827-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-022-00827-w