Abstract
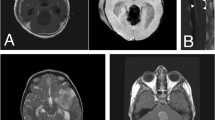
Atypical teratoid/rhabdoid tumor (AT/RT) is a rare intracranial tumor occurring predominantly in young children. The prognosis is poor, and no effective treatment is currently available. To develop novel effective therapies, there is a need for experimental models for AT/RT. In this research, we established a cell line from a patient’s AT/RT tissue (designated ATRT_OCGH) and performed drug screening using 164 FDA-approved anti-cancer agents, to identify candidates for therapeutic options. We found that bortezomib, a proteasome inhibitor, was among the agents for which the cell line showed high sensitivity, along with tyrosine kinase inhibitors, topoisomerase inhibitors, and histone deacetylase inhibitors, which are known to exert anti-AT/RT effects. Concomitant use of panobinostat potentiated the inhibitory effect of bortezomib on AT/RT cell proliferation. Our findings may provide a rationale for considering combination therapy of panobinostat and bortezomib for treatment of AT/RT.




Similar content being viewed by others
References
Fruhwald MC, Biegel JA, Bourdeaut F, Roberts CW, Chi SN. Atypical teratoid/rhabdoid tumors-current concepts, advances in biology, and potential future therapies. Neuro-oncology. 2016;18:764–78.
Richardson EA, Ho B, Huang A. Atypical teratoid rhabdoid tumour: from tumours to therapies. J Korean Neurosurg Soc. 2018;61:302–11.
Yamasaki K, Kiyotani C, Terashima K, et al. Clinical characteristics, treatment, and survival outcome in pediatric patients with atypical teratoid/rhabdoid tumors: a retrospective study by the Japan Children’s Cancer Group. J Neurosurg Pediatr. 2019;25:1–10.
Torchia J, Golbourn B, Feng S, et al. Integrated (epi)-genomic analyses identify subgroup-specific therapeutic targets in CNS rhabdoid tumors. Cancer Cell. 2016;30:891–908.
Johann PD, Erkek S, Zapatka M, et al. Atypical teratoid/rhabdoid tumors are comprised of three epigenetic subgroups with distinct enhancer landscapes. Cancer Cell. 2016;29:379–93.
Narendran A, Coppes L, Jayanthan A, et al. Establishment of atypical-teratoid/rhabdoid tumor (AT/RT) cell cultures from disseminated CSF cells: a model to elucidate biology and potential targeted therapeutics. J Neuro-Oncol. 2008;90:171–80.
Yoshimatsu Y, Noguchi R, Tsuchiya R, et al. Establishment and characterization of NCC-DFSP3-C1: a novel patient-derived dermatofibrosarcoma protuberans cell line. Hum Cell. 2020;33:877.
Bairoch A. The Cellosaurus, a cell-line knowledge resource. J Biomol Tech. 2018;29:25–38.
Moreau P. How I treat myeloma with new agents. Blood. 2017;130:1507–13.
San-Miguel JF, Hungria VT, Yoon SS, et al. Panobinostat plus bortezomib and dexamethasone versus placebo plus bortezomib and dexamethasone in patients with relapsed or relapsed and refractory multiple myeloma: a multicentre, randomised, double-blind phase 3 trial. Lancet Oncol. 2014;15:1195–206.
Hideshima T, Richardson P, Chauhan D, et al. The proteasome inhibitor PS-341 inhibits growth, induces apoptosis, and overcomes drug resistance in human multiple myeloma cells. Cancer Res. 2001;61:3071–6.
Adams J. The proteasome: a suitable antineoplastic target. Nat Rev Cancer. 2004;4:349–60.
Cheah CY, Seymour JF, Wang ML. Mantle cell lymphoma. J Clin Oncol. 2016;34:1256–69.
Carugo A, Minelli R, Sapio L, et al. p53 is a aster regulator of proteostasis in SMARCB1-efficient malignant rhabdoid tumors. Cancer Cell. 2019;35:204–20.
Wu DY, Tkachuck DC, Roberson RS, Schubach WH. The human SNF5/INI1 protein facilitates the function of the growth arrest and DNA damage-inducible protein (GADD34) and modulates GADD34-bound protein phosphatase-1 activity. J Biol Chem. 2002;277:27706–15.
Hertwig F, Meyer K, Braun S, et al. Definition of genetic events directing the development of distinct types of brain tumors from postnatal neural stem/progenitor cells. Cancer Res. 2012;72:3381–92.
Pakos-Zebrucka K, Koryga I, Mnich K, et al. The integrated stress response. EMBO Rep. 2016;17:1374–95.
Schewe DM, Aguirre-Ghiso JA. Inhibition of eIF2alpha dephosphorylation maximizes bortezomib efficiency and eliminates quiescent multiple myeloma cells surviving proteasome inhibitor therapy. Cancer Res. 2009;69:1545–52.
Hollmann TJ, Hornick JL. INI1-deficient tumors: diagnostic features and molecular genetics. Am J Surg Pathol. 2011;35:e47–e63.
Carden MA, Smith S, Meany H, et al. Platinum plus bortezomib for the treatment of pediatric renal medullary carcinoma: two cases. Pediatr Blood Cancer. 2017;64:e26402.
Ronnen EA, Kondagunta GV, Motzer RJ. Medullary renal cell carcinoma and response to therapy with bortezomib. J Clin Oncol. 2006;24:e14.
Raizer JJ, Chandler JP, Ferrarese R, et al. A phase II trial evaluating the effects and intra-tumoral penetration of bortezomib in patients with recurrent malignant gliomas. J Neuro-Oncol. 2016;129:139–46.
Hideshima T, Richardson PG, Anderson KC. Mechanism of action of proteasome inhibitors and deacetylase inhibitors and the biological basis of synergy in multiple myeloma. Mol Cancer Ther. 2011;10:2034–42.
Kawaguchi Y, Kovacs JJ, McLaurin A, et al. The deacetylase HDAC6 regulates aggresome formation and cell viability in response to misfolded protein stress. Cell. 2003;115:727–38.
Moreno N, Kerl K. Preclinical evaluation of combined targeted approaches in malignant rhabdoid tumors. Anticancer Res. 2016;36:3883–7.
Jochems J, Boulden J, Lee BG, et al. Antidepressant-like properties of novel HDAC6-selective inhibitors with improved brain bioavailability. Neuropsychopharmacology. 2014;39:389–400.
Acknowledgements
The authors thank the patient and their family for participating in this research. We thank Y. Matsushita and K. Hatake for their technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval
The study was approved by the Institutional Review Boards at all participating institutions.
Informed consent
Informed, written consent was obtained from the patient’s parent.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nakano, Y., Takadera, M., Miyazaki, M. et al. Drug screening with a novel tumor-derived cell line identified alternative therapeutic options for patients with atypical teratoid/rhabdoid tumor. Human Cell 34, 271–278 (2021). https://doi.org/10.1007/s13577-020-00438-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-020-00438-3