Abstract
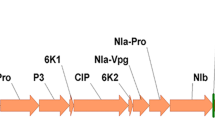
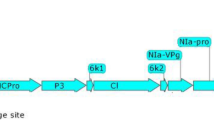
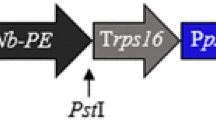
Neutralizing nanobodies against vascular endothelial growth factor (VEGF) and its receptors are the most important types of medicine which are used in cancer therapy strategies. To increase the level of anti-VEGF (Nb42) nanobody accumulation and also to simplify its subsequent purification, we fused codon-optimized Nb42 nanobody to hydrophobin I (HFBI) stabilizing partner, and transiently expressed the fusion gene cassette in cucurbit plants using a zucchini yellow mosaic virus-based viral vector. The successful transcription of the target gene was confirmed with the use of RT-PCR and Real-time PCR analyses. According to the immunoblot assay results the expression level of Nb42-HFBI fusion protein, 22 μg/g fresh leaf tissue, was twofolds higher than the unfused nanobody. Furthermore, the endoplasmic reticulum (ER)-targeted Nb42-HFBI fusion protein was successfully purified from leaf extract using an aqueous-two phase system (ATPS) method. The use of HFBI fusion partner increased the expression level of the Nb42 recombinant protein while simultaneously facilitated its subsequent purification using an inexpensive non-chromatographic method known as ATPS. As a conclusion, the application of ER-targeted hydrophobin fusion strategy with the use of special features of plant viral vectors, can improve the value of plant-based transient expression platform for the production of important proteins with the industrial and therapeutic application.







Similar content being viewed by others
Abbreviations
- VEGF:
-
Vascular endothelial growth factor
- HFBI:
-
Hydrophobin I
- ER:
-
Endoplasmic reticulum
- ATPS:
-
Aqueous-two phase system
- MAbs:
-
Monoclonal antibodies
References
Arazi T et al (2001) Engineering zucchini yellow mosaic potyvirus as a non-pathogenic vector for expression of heterologous proteins in cucurbits. J Biotechnol 87:67–82
Behdani M et al (2012) Generation and characterization of a functional Nanobody against the vascular endothelial growth factor receptor-2; angiogenesis cell receptor. Mol Immunol 50:35–41
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buyel JF, Fischer R (2014) Downstream processing of biopharmaceutical proteins produced in plants: the pros and cons of flocculants. Bioengineered 5:138–142
Buyel J, Twyman R, Fischer R (2015) Extraction and downstream processing of plant-derived recombinant proteins. Biotechnol Adv 33:902–913
Buyel J, Twyman R, Fischer R (2017) Very-large-scale production of antibodies in plants: the biologization of manufacturing. Biotechnol Adv 35:458–465
Chen Q (2008) Expression and purification of pharmaceutical proteins in plants. Biol Eng 1:291–321
Conley AJ, Joensuu JJ, Menassa R, Brandle JE (2009) Induction of protein body formation in plant leaves by elastin-like polypeptide fusions. BMC Biol 7:48
Conley AJ, Joensuu JJ, Richman A, Menassa R (2011) Protein body-inducing fusions for high-level production and purification of recombinant proteins in plants. Plant Biotechnol J 9:419–433
Conrad U et al (2011) ELPylated anti-human TNF therapeutic single-domain antibodies for prevention of lethal septic shock. Plant Biotechnol J 9:22–31
Cutt JR, Dixon DC, Carr JP, Klessig DF (1988) Isolation and nuleotide sequence of cDNA clones for the pathogenesis-related proteins PR1a, PR1b and PRIc of Nicotiana tabacum cv. Xanthi nc induced by TMV infection. Nucleic Acids Res 16:9861
Ebrahimizadeh W, Gargari SLMM, Javidan Z, Rajabibazl M (2015) Production of novel VHH nanobody inhibiting angiogenesis by targeting binding site of VEGF. Appl Biochem Biotech 176:1985–1995
Ellis LM, Hicklin DJ (2008) VEGF-targeted therapy: mechanisms of anti-tumour activity. Nat Rev Cancer 8:579–591
Fiedler U, Phillips J, Artsaenko O, Conrad U (1997) Optimization of scFv antibody production in transgenic plants. Immunotechnology 3:205–216
Gerasimova S, Smirnova O, Kochetov A, Shumnyi V (2016) Production of recombinant proteins in plant cells. Russ J Plant Physiol 63:26–37
Gleba Y, Marillonnet S, Klimyuk V (2004) Engineering viral expression vectors for plants: the ‘full virus’ and the ‘deconstructed virus’ strategies. Curr Opin Plant Biol 7:182–188
Gleba Y, Klimyuk V, Marillonnet S (2007) Viral vectors for the expression of proteins in plants. Curr Opin Biotechnol 18:134–141
Gleba YY, Tusé D, Giritch A (2013) Plant viral vectors for delivery by Agrobacterium. In: Palmer K, Gleba Y (eds) Plant Viral Vectors. Springer, Berlin, pp 155–192
Gutiérrez SP, Saberianfar R, Kohalmi SE, Menassa R (2013) Protein body formation in stable transgenic tobacco expressing elastin-like polypeptide and hydrophobin fusion proteins. BMC Biotechnol 13:40
Hsu C-H, Lin S-S, Liu F-L, Su W-C, Yeh S-D (2004) Oral administration of a mite allergen expressed by zucchini yellow mosaic virus in cucurbit species downregulates allergen-induced airway inflammation and IgE synthesis. J Allergy Clin Immun 113:1079–1085
Huang Z, Dry I, Webster D, Strugnell R, Wesselingh S (2001) Plant-derived measles virus hemagglutinin protein induces neutralizing antibodies in mice. Vaccine 19:2163–2171
Huang Z et al (2010) High-level rapid production of full-size monoclonal antibodies in plants by a single-vector DNA replicon system. Biotechnol Bioeng 106:9
Ismaili A, Jalali-Javaran M, Rasaee MJ, Rahbarizadeh F, Forouzandeh-Moghadam M, Memari HR (2007) Production and characterization of anti-(mucin MUC1) single-domain antibody in tobacco (Nicotiana tabacum cultivar Xanthi). Biotechnol Appl Biochem 47:11–19
Jacquet N, Navarre C, Desmecht D, Boutry M (2014) Hydrophobin fusion of an influenza virus hemagglutinin allows high transient expression in Nicotiana benthamiana, easy purification and immune response with neutralizing activity. PLoS ONE 9:e115944
Javaran VJ, Shafeinia A, Javaran MJ, Gojani EG, Mirzaee M (2017) Transient expression of recombinant tissue plasminogen activator (rt-PA) gene in cucurbit plants using viral vector. Biotechnol Lett 39:607–612
Joensuu JJ, Conley AJ, Lienemann M, Brandle JE, Linder MB, Menassa R (2010) Hydrophobin fusions for high-level transient protein expression and purification in Nicotiana benthamiana. Plant Physiol 152:622–633
Joensuu JJ, Conley AJ, Linder MB, Menassa R (2012) Bioseparation of recombinant proteins from plant extract with hydrophobin fusion technology. In: Lorence A (ed) Recombinant gene expression: reviews and protocols, 3rd edn. Springer, New York, pp 527–534
Kang M, Seo J-K, Choi H, Choi H-S, Kim K-H (2016) Establishment of a simple and rapid gene delivery system for cucurbits by using engineered of Zucchini yellow mosaic virus. Plant Pathol J 32:70
Kazemi-Lomedasht F et al (2015) Inhibition of angiogenesis in human endothelial cell using VEGF specific nanobody. Mol Immunol 65:58–67
Ko K, Steplewski Z, Glogowska M, Koprowski H (2005) Inhibition of tumor growth by plant-derived mAb. Proc Natl Acad Sci 102:7026–7030
Kopertekh L, Schiemann J (2017) Transient production of recombinant pharmaceutical proteins in plants: evolution and perspectives. Curr Med Chem 24:1–16
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lai H, He J, Engle M, Diamond MS, Chen Q (2012) Robust production of virus-like particles and monoclonal antibodies with geminiviral replicon vectors in lettuce. Plant Biotechnol J 10:95–104
Lentz EM, Garaicoechea L, Alfano EF, Parreño V, Wigdorovitz A, Bravo-Almonacid FF (2012) Translational fusion and redirection to thylakoid lumen as strategies to improve the accumulation of a camelid antibody fragment in transplastomic tobacco. Planta 236:703–714
Lico C, Chen Q, Santi L (2008) Viral vectors for production of recombinant proteins in plants. J Cell Physiol 216:366–377
Linder MB, Qiao M, Laumen F, Selber K, Hyytiä T, Nakari-Setälä T, Penttilä ME (2004) Efficient purification of recombinant proteins using hydrophobins as tags in surfactant-based two-phase systems. Biochemistry 43:11873–11882
López-Moya JJ, Fernández-Fernández MR, Cambra M, García JA (2000) Biotechnological aspects of plum pox virus. J Biotechnol 76:121–136
McLean MD et al (2012) Purification of the therapeutic antibody trastuzumab from genetically modified plants using safflower protein A-oleosin oilbody technology. Transgenic Res 21:1291–1301
Mirzaee M, Jalali-Javaran M, Moieni A, Zeinali S, Behdani M, Shams-Bakhsh M, Modarresi M (2017) Anti-VEGFR2 nanobody expression in lettuce using an infectious Turnip mosaic virus vector. J Plant Biochem Biotechnol 27:167–174
Riechmann JL, Laín S, García JA (1992) Highlights and prospects of potyvirus molecular biology. J Gen Virol 73:1–16
Shiboleth YM, Arazi T, Wang Y, Gal-On A (2001) A new approach for weed control in a cucurbit field employing an attenuated potyvirus-vector for herbicide resistance. J Biotechnol 92:37–46
Sil B, Jha S (2014) Plants: the future pharmaceutical factory. Am J Plant Sci 5:319
Simmons H, Dunham J, Zinn K, Munkvold G, Holmes E, Stephenson A (2013) Zucchini yellow mosaic virus (ZYMV, Potyvirus): vertical transmission, seed infection and cryptic infections. Virus Res 176:259–264
Steeland S, Vandenbroucke RE, Libert C (2016) Nanobodies as therapeutics: big opportunities for small antibodies. Drug Discov Today 21:1076–1113
Tschofen M, Knopp D, Hood E, Stöger E (2016) Plant molecular farming: much more than medicines. Annu Rev Anal Chem 9:271–294
Vardakou M, Sainsbury F, Rigby N, Mulholland F, Lomonossoff GP (2012) Expression of active recombinant human gastric lipase in Nicotiana benthamiana using the CPMV-HT transient expression system. Protein Expr Purif 81:69–74
Wilken LR, Nikolov ZL (2012) Recovery and purification of plant-made recombinant proteins. Biotechnol Adv 30:419–433
Winichayakul S, Pernthaner A, Scott R, Vlaming R, Roberts N (2009) Head-to-tail fusions of camelid antibodies can be expressed in planta and bind in rumen fluid. Biotechnol Appl Biochem 53:111–122
Youssoufian H, Hicklin DJ, Rowinsky EK (2007) Review: monoclonal antibodies to the vascular endothelial growth factor receptor-2 in cancer therapy. Clin Cancer Res 13:5544s–5548s
Zhang J, Yang PL, Gray NS (2009) Targeting cancer with small molecule kinase inhibitors. Nat Rev Cancer 9:28–39
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Soleimanizadeh, M., Bagheri, A., Jalali Javaran, M. et al. Enhanced expression and purification of anti-VEGF nanobody in cucurbit plants. J. Plant Biochem. Biotechnol. 28, 263–270 (2019). https://doi.org/10.1007/s13562-018-0471-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-018-0471-9