Abstract
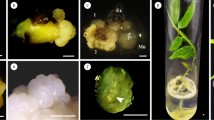
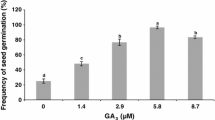
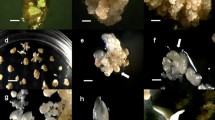
An efficient protocol for a complete plant regeneration by somatic embryogenesis was developed with Smooth Cayenne pineapple (Ananas comosus L.). Previous works showed that this species is responsive to somatic embryogenesis. In the present work the influence of components of culture medium in the induction, development and conversion of somatic embryos was investigate in order to establish a somatic embryogenesis protocol. Nodular callus (83.67%) was initiated from leaf explants of young plants on CIM3 medium. The highest frequency (37.6%) of embryogenic callus induction was obtained from 4-week-old calluses on EIM3 medium supplemented with 3.0 mg/l picloram. The highly organized callus induction and the development of somatic embryos were achieved after the transfer of callus clumps onto EIM3 medium containing 1.0 mg/l BAP + 0.1 mg/l NAA. The frequency of somatic embryo formation was of 39.5 ± 2.45 embryos per callus. Up to 97% of the somatic embryos were converted into complete plants within 4 week on MSB medium with 1.0 mg/l BAP + 0.05 mg/l GA3 + 500 mg/l glutamine. The continuation of the elongation of the shoots occurred on this medium). Shoots obtained from all the above methods were rooted in MSB medium with activated charcoal. Complete plantlets were transferred onto specially made polyethylene bags containing soil mixture and transferred to the greenhouse. Survival rate of the plantlets under ex vitro conditions was 98% and maximum average number of plantlets (80 ± 0.6). The well-developed plantlets were transferred to an open field where the plants produced normal fruits.

Similar content being viewed by others
Abbreviations
- BAP:
-
6-benzylaminopurine
- GA3 :
-
Gibberellic acid
- IBA:
-
Indole-3-butyric acid
- KIN:
-
Kinetin
- MS:
-
Murashige and Skoog
- NAA:
-
Naphthalene acetic acid
- picloram:
-
4-amino-3,5,6-trichloropicolinic acid
- 2,4-D:
-
2,4-dichlorophenoxyacetic acid
References
Arnold SV, Sabala I, Bozhkov P, Dyachok J, Filonova L (2002) Developmental pathways of somatic embryogenesis. Plant Cell Tiss Organ Cult 69:233–249
Chakroun A, Jemmali A, Ben Hamed K, Abdelli C, Druart P (2007) Effet du nitrate d’ammonium sur le développement et l’activité des enzymes anti-oxydantes du fraisier (Fragaria x ananassa L.) micropropagé. Biotechnol Agron Soc Environ 11(2):89–95
Dal Vesco LL, Guerra MP (2001) The effectiveness of nitrogen sources in Feijoa somatic embryogenesis. Plant Cell Tiss Organ Cult 64:19–25
Daquinta MA, Cisneros A, Rodriguez Y, Escalona M, Perez MC, Luna I, Borroto CG (1997) Somatic embryogenesis in pineapple (Ananas comosus L.) Merr.) In: Martin-Prevel P, Hugon R (eds) Proceeding of the Second International Pineapple Symposium. Acta Hort 425:251–257
Devi YS, Mujib A, Kundu SC (1997) Efficient regenerative potential from long term culture of pineapple. Phytomorphology 47:255–259
Dublin P, Enjalric F, Lardet L, Carron MP, Trolinder N, Pannetier C (1991) Estate crops. In: Debergh PC, Zimmerman RH (eds) Micropropagation technology and application. Kluwer, Dordrecht, pp 337–361
Filner P (1966) Regulation of nitrate reductase in cultured tobacco cells. Biochem Biophys Acta 118:299–310
Firoozabady E, Moy Y (2004) Regeneration of pineapple plants via somatic embryogenesis and organogenesis. In Vitro Cell Dev Biol Plant 40:67–74. doi:10.1079/IVP2003494
Firoozabady E, Heckert M, Gutterson N (2006) Transformation and regeneration of pineapple. Plant Cell Tiss Organ Cult 84:1–16. doi:10.1007/s11240-005-1371-y
Fuentes-Cerda CFJ, Monforte-Gonzalez M, Mendez-Zeel M, Rojas-Herrera R, Loyola-Vargas VM (2006) Modification of the embryogenic response of Coffea arabica by the nitrogen source. Biotechnol Lett 23:1341–1343
Fukunaga Y, King J (1982) The effect of L-amino acid on the growth and nitrate reductase activity in cultured cells of Datura innoxia. Plant Sci Lett 24:45–54
Gangopadhyay G, Roy SK, Basu GS, Mukherjee KK (2009) Agrobacterium-mediated genetic transformation of pineapple var. Queen using a novel encapsulation-based antibiotic selection technique. Plant Cell Tiss Organ Cult 97:295–302
George EF (1993) Plant Propagation by tissue culture: Part I: the technology. Exegetics Ltd, Edington, pp 273–291
Gona KK, Omid K (2008) Picloram-induced somatic embryogenesis in leaves of strawberry (Fragaria ananassa L.). Acta Biol Cracov Bot 50:69–72
Gonzalez-Arnao MT, Panta A, Roca WM, Escobar RH, Engelmann F (2008) Development and large-scale application of cryopreservation techniques for shoot and somatic embryo cultures of tropical crops. Plant Cell Tiss Organ Cult 92:1–13
Greer MS, Kovalchuk I, Eud F (2009) Ammonium nitrate improves direct somatic embryogenesis and biolistic transformation of Triticum aestiva. New Biotechnol 26:44–52
Ikram UH (2005) Callus proliferation and somatic embryogenesis in cotton (Gossypium hirsutum L.). Afr J Biotechnol 4:206–209
Ikram UH, Yusuf Z (2004) Effect of nitrates on embryo induction efficiency in cotton (Gossypium hirsutum L.). Afr J Biotechnol 6:319–323
Jain A, Rout GR, Raina SN (2002) Somatic embryogenesis and plant regeneration from callus cultures of Phlox paniculta Linn. Sci Hortic 94:137–143
Le Van TH, Takamura T, Tanaka M (2004) Callus formation and plant regeneration from callus through somatic embryo structures in Cymbidium orchid. Plant Sci 166:1443–1449. doi:10.1016/j.plantsci.2004.01.023
Manoj KR, Akhtar N, Jaiswal VS (2007) Somatic embryogenesis and plant regeneration in Psidium guajava L. cv. Banarasi local. Sci Hortic 113:129–133. doi:10.1016/j.scienta.2007.02.010
Murashige T, Skoog F (1962) Revised medium for rapid growth and bioassays with tobacco tissue culture. Physiol Plant 15:473–497
Nomura K, Komamime A (1995) Physiological and biochemical aspect of somatic embryogenesis. Curr Plant Sci Biotechnol Agric 20:249–265
Odutayo O, Akinrimisi FB, Ogunbosoye I, Oso RT (2005) Multiple shoot induction from embryo derived callus cultures of cowpea (Vigna unguiculata L.) Walp. Afr J Biotechnol 4(11):1214–1216
Rahman KW, Ahmed MB, Rahman MM, Amin MN, Hossain MS, Ahmed R (2005) Large scale plant regeneration in vitro from leaf derived callus cultures of pineapple [Ananas comosus (L.) Merr. cv. Giant Kew]. Int J Bot 1(2):128–132
Samson NP, Campa C, Le Gal L, Noirot M, Thomas G, Lokeswari TS, de Kochko A (2006) Effect of primary culture medium composition on high frequency somatic embryogenesis in different Coffea species. Plant Cell Tiss Organ Cult 86:37–45
Selvaraj N, Vasudevan A, Manickavasagam M, Ganapathi A (2006) in vitro organogenesis and plant formation in cucumber. Biol Plant 50(1):123–126
Smith MK, Ko HL, Hamill SD, Sanewski GM (2002) Pineapple Transformation: managing somaclonal variation. Acta Hortic 575:69–74
Sripaoraya S, Marchant R, Brian PJ, Davey MR (2003) Plant regeneration by somatic embryogenesis and organogenesis in commercial pineapple (Ananas comosus L.). In Vitro Cell Dev Biol Plant 39:450–454. doi:10.1079/IVP2003445
Stamp JA, Henshaw GG (1987) Somatic embryogenesis from clonal leaf tissues of cassava. Ann Bot 59:445–450
Thiruvengadam M, Mohamed SV, Yang CH, Jayabalan N (2006) Development of an embryogenic suspension culture of bitter melon (Momordica charantia L.). Sci Hortic 109:123–129. doi:10.1016/j.scienta.2006.03.012
Thomas DT (2008) The role of activated charcoal in plant tissue culture. Biotechnol Adv 26:618–631
Wu YJ, Huang XL, Chen QZ, Li XJ, Engelmann F (2007) Induction and cryopreservation of embryogenic cultures from nucelli and immature cotyledon cuts of mango (Mangifera indica L. var Zihua). Plant Cell Rep 26:161–168
Yadava R, Chawla HS (2002) Role of genotypes, growth regulators and amino acids on callus induction and plant regeneration from different developmental stages of inflorescence in wheat. Indian J Genet Plant Breed 62(1):55–60
Zaidi MA, Narayanan M, Sardana R, Taga I, Postel S, Johns R, McNulty M, Mottiar Y, Mao J, Loi E, Altosaar I (2006) Optimizing tissues culture media for efficient transformation of different indica rice genotype. Agron Res 4(2):563–575
Zhang CX, Li Q, Kong L (2007) Induction, development and maturation of somatic embryos in Bunge’s pine (Pinus bungeana Zucc. ex Endl.). Plant Cell Tiss Organ Cult 91:273–280
Acknowledgments
Authors are thankful to the National Centre for Agronomic Research of Ivory Coast for supplying pineapple suckers. We are grateful to Dr. Alison Pawlus for critically reading the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yapo, E.S., Kouakou, T.H., Kone, M. et al. Regeneration of pineapple (Ananas comosus L.) plant through somatic embryogenesis. J. Plant Biochem. Biotechnol. 20, 196–204 (2011). https://doi.org/10.1007/s13562-011-0046-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13562-011-0046-5