Abstract
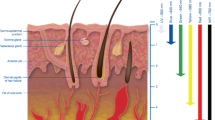
Numerous designs and methods have been examined to improve penetration depth (PD), but there is a need for research to explore the potential increase in PD through uniform heating, a compact applicator, and low input power. This paper presents metasurface based hyperthermia lens applicator with water bolus for uniform heating of cancerous tissues. The proposed applicator consists of a stacked spiral antenna and a spiral-shaped frequency selective surface as a superstrate. The spiral antenna and superstrate are optimized on a low cost FR4 substrate having a size of 32 × 32 × 3.27mm3 and 10 × 10 × 1.6mm3 (size of the unit cell), respectively. The proposed applicator is simulated with heterogeneous phantom (skin, fat, and muscle layers) and with the Gustav voxel model with and without a water bolus layer. The number of unit cells in the superstrate is optimized to direct the maximum energy toward the tumor location. The performance study of the applicator is carried out in terms of specific absorption rate, PD, and effective field size. Further, thermal analysis is carried out with 1.9 W of input power at the antenna port, and the highest 44.7 °C temperature rise is obtained. The cancerous tissue’s (tumor) surrounding temperature is between 41 and 45 °C, which is adequate for efficient hyperthermia treatment. Finally, the proposed metasurface hyperthermia lens applicator is fabricated and experimentally validated in a mimicked phantom’s presence.







Similar content being viewed by others
References
Cabuy E. Electrochemical therapy in cancer treatment. Reliab Cancer Ther. 2012;6:1–20.
Kroesen M, Mulder HT, van Holthe JML, Aangeenbrug AA, Mens JWM, van Doorn HC, et al. Confirmation of thermal dose as a predictor of local control in cervical carcinoma patients treated with state-of-the-art radiation therapy and hyperthermia. Radiother Oncol. 2019;140:150–8. https://doi.org/10.1016/j.radonc.2019.06.021.
Bakker A, van der Zee J, van Tienhoven G, Kok HP, Rasch CRN, Crezee H. Temperature and thermal dose during radiotherapy and hyperthermia for recurrent breast cancer are related to clinical outcome and thermal toxicity: a systematic review. Int J Hyperth. 2019;36:1024–39. https://doi.org/10.1080/02656736.2019.1665718.
Kok HP, Cressman ENK, Ceelen W, Brace CL, Ivkov R, Grüll H, et al. Heating technology for malignant tumors: a review. Int J Hyperth. 2020;37:711–41. https://doi.org/10.1080/02656736.2020.1779357.
Tumors D. Radiofrequency capacitive hyperthermia for deep-seated tumors. Cancer. 1987;60:121–7. https://doi.org/10.1002/1097-0142(19870701)60:1%3c128::AID-CNCR2820600124%3e3.0.CO;2-V.
Zhu L, Altman MB, Laszlo A, Straube W, Zoberi I, Hallahan DE, et al. Ultrasound hyperthermia technology for radiosensitization. Ultrasound Med Biol. 2019;45:1025–43. https://doi.org/10.1016/j.ultrasmedbio.2018.12.007.
Singh S, Sahu B, Singh SP. Conformal microstrip slot antenna with an AMC reflector for hyperthermia. J Electromagn Waves Appl. 2016;30:1603–19. https://doi.org/10.1080/09205071.2016.1207568.
van Vulpen M, Raaymakers BW, Lagendijk JJW, Crezee J, de Leeuw AAC, van Moorselaar JRA, et al. Three-dimensional controlled interstitial hyperthermia combined with radiotherapy for locally advanced prostate carcinoma—a feasibility study. Int J Radiat Oncol. 2002;53:116–26. https://doi.org/10.1016/S0360-3016(01)02828-0.
Guy AW. Electromagnetic fields and relative heating patterns due to a rectangular aperture source in direct contact with bilayered biological tissue. IEEE Trans Microw Theory Tech. 1968;16:214–23. https://doi.org/10.1109/TMTT.1968.1127485.
Singh S, Singh SP. Water-loaded metal diagonal horn applicator for hyperthermia. IET Microw Antennas Propag. 2015;9:814–21. https://doi.org/10.1049/iet-map.2014.0699.
Tanabe E, McEuen AH, Caslow S, Norris CS, Samulski T V., Fessenden P. Microstrip spiral antenna for local hyperthermia. In: IEEE MTT-S international microwave symposium digest, pp. 133–134. https://doi.org/10.1109/mwsym.1984.1131715.
Yong-xing DU. The design and simulation of two-armed spiral antenna for microwave hyperthermia 2011, pp. 3–6.
Koo YS, Kazemi R, Liu Q, Phillips JC, Fathy AE. Development of a high SAR conformal antenna for hyperthermia tumors treatment. IEEE Trans Antennas Propag. 2014;62:5830–40. https://doi.org/10.1109/TAP.2014.2357419.
Korkmaz E, Isik O, Nassor MA. A compact microstrip spiral antenna embedded in water bolus for hyperthermia applications. Int J Antennas Propag. 2013. https://doi.org/10.1155/2013/954986.
Curto S, McEvoy P, Bao X, Ammann MJ. Compact patch antenna for electromagnetic interaction with human tissue at 434 MHz. IEEE Trans Antennas Propag. 2009;57:2564–71. https://doi.org/10.1109/TAP.2009.2027040.
Montecchia F. Microstrip-antenna design for hyperthermia treatment of superficial tumors. IEEE Trans Biomed Eng. 1992;39:580–8. https://doi.org/10.1109/10.141196.
Choi WC, Lim S, Yoon YJ. Evaluation of transmit-array lens antenna for deep-seated hyperthermia tumor treatment. IEEE Antennas Wirel Propag Lett. 2020;19:866–70. https://doi.org/10.1109/LAWP.2020.2982676.
Tao Y, Wang G. Conformal hyperthermia of superficial tumor with left-handed metamaterial lens applicator. IEEE Trans Biomed Eng. 2012;59:3525–30. https://doi.org/10.1109/TBME.2012.2218108.
Velázquez-Ahumada MC, Freire MJ, Marqués R. Metamaterial focusing device for microwave hyperthermia. Microw Opt Technol Lett. 2011;53:2868–72. https://doi.org/10.1002/mop.26434.
Jaffar NA, Lias K, Madzhi NK, Buniyamin N. Improving the performance of applicators for use in hyperthermia cancer treatment procedure by the introduction of LHM lens. Int J Electr Electron Syst Res. 2019;14:24–9.
Gong Y, Wang G. Superficial tumor hyperthermia with flat left-handed metamaterial lens. Prog Electromagn Res. 2009;98:389–405. https://doi.org/10.2528/PIER09091401.
Available from: http://www.cst.com (n.d.).
Sharma N, Singh HS, Khanna R, Kaur A, Agarwal M. Development of deeply focused microwave lens applicator for efficient hyperthermia treatment. Optik. 2022;259:168946. https://doi.org/10.1016/j.ijleo.2022.168946.
Tao Y, Wang G. Influence of source offset on breast tumor hyperthermia with Γ-shaped LHM lens applicator. In: 2010 International conference on microwave and millimeter wave technology ICMMT 2010, pp. 1859–61. https://doi.org/10.1109/ICMMT.2010.5524876.
Wu T, Consulting A, Cost L. Frequency selective surfaces. New York: Inc New York; 2016.
Razi ZM, Rezaei P, Valizade A. A novel design of Fabry- Perot antenna using metamaterial superstrate for gain and bandwidth enhancement international. AEUE Int J Electron Commun. 2015;69:1525–32. https://doi.org/10.1016/j.aeue.2015.05.012.
Foroozesh A, Shafai L. Investigation into the effects of the patch-type FSS superstrate on the high-gain cavity resonance antenna design. IEEE Trans Antennas Propag. 2010;58:258–70. https://doi.org/10.1109/TAP.2009.2037702.
Holloway CL, Dienstfrey A, Kuester EF, Hara JFO, Azad AK, Taylor AJ. A discussion on the interpretation and characterization of metafilms/metasurfaces: the two-dimensional equivalent of metamaterials. Metamaterials. 2009;3:100–12. https://doi.org/10.1016/j.metmat.2009.08.001.
Holloway CL, Kuester EF, Gordon JA, Hara JO, Booth J, Smith DR. An overview of the theory and applications of metasurfaces: the two-dimensional equivalents of metamaterials. IEEE Antennas Propag Mag. 2012;54:10–35.
Pfeiffer C, Grbic A. Metamaterial Huygens’ surfaces: tailoring wave fronts with reflectionless sheets. Phys Rev Lett. 2013;110:197401. https://doi.org/10.1103/PhysRevLett.110.197401.
Feresidis AP, Vardaxoglou JC. High gain planar antenna using optimised partially reflective surfaces. IEEE Proc Microw Antennas Propag. 2001;148:345–50.
Available from https://itis.swiss/virtual-population/tissue-properties/database/dielectric-properties.
Neuman DG, Stauffer PR, Jacobsen S, Rossetto F. SAR pattern perturbations from resonance effects in water bolus layers used with superficial microwave hyperthermia applicators. Int J Hyperth. 2002;18:180–93. https://doi.org/10.1080/02656730110119198.
Pennes HH. Analysis of tissue and arterial blood temperatures in the resting human forearm. J Appl Physiol. 1948;85:5–34. https://doi.org/10.1152/jappl.1998.85.1.5.
Karacolak T, Hood AZ, Topsakal E. Design of a dual-band implantable antenna and development of skin mimicking gels for continuous glucose monitoring. IEEE Trans Microw Theory Tech. 2008;56:1001–8.
Sani A, Rajab M, Foster R, Hao Y. Antennas and propagation of implanted RFIDs for pervasive healthcare applications. Proc IEEE. 2010;98:1648–55. https://doi.org/10.1109/JPROC.2010.2051010.
Acknowledgements
The authors would like to take an opportunity to thank the TIET-VT Center of Excellence in Emerging Materials (CEEMS), TIET for facilitating the laboratory and providing financial support to carry out this research.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
NS: Problem definition, analysis, write-up, and review. HSS: Analysis, write-up, and review. RK: Funding acquisition. AK: Review. MA: Write-up and review.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Consent for publication
Not applicable.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, N., Singh, H.S., Khanna, R. et al. Development of metasurface based hyperthermia lens applicator for heating of cancerous tissues. Biomed. Eng. Lett. 14, 1–12 (2024). https://doi.org/10.1007/s13534-023-00300-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13534-023-00300-z