Abstract
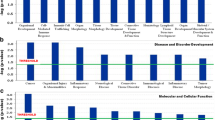
Polychlorinated biphenyls (PCBs) are persistent organic compounds that have been banned since 1970s, but continue to contaminate the environment. PCBs are categorized into two structural groups: coplanar and non-coplanar PCBs. The coplanar PCBs are dioxin-like potent toxic compounds. To evaluate their effects on humans, we chose a coplanar PCB77 for data analysis. We performed meta- analysis by integrating datasets via the Rank Product method, and identified 375 up- and 66 down- regulated differentially expressed genes (DEGs). Notably, up-regulated genes were significantly associated with liver and kidney diseases. Using gene ontology enrichment, we found that the up-regulated DEGs were significantly enriched in the apoptotic process (false discovery rate, FDR=1.62e-10) and response to unfolded protein (FDR=7.65e-10). Protein-protein interaction networks identified the hub proteins containing HSP90AB1 and HSPA5. These findings suggest that our DEGs may provide a robust set of genetic markers for PCB77.
Similar content being viewed by others
References
Tanabe, S., Kannan, N., Subramanian, A., Watanabe, S. & Tatsukawa, R. Highly toxic coplanar PCBs: occurrence, source, persistency and toxic implications to wildlife and humans. Environ. Pollut. 47, 147–163 (1987).
Morelle, R. ‘Shocking’ levels of PCB chemicals in UK killer whale Lulu, http://www.bbc.com/news/science-environment-39738582 (2017).
Moysich, K. B. et al. Polychlorinated biphenyls, cytochrome P4501A1 polymorphism, and postmenopausal breast cancer risk. Cancer Epidemiol. Biomarkers Prev. 8, 41–44 (1999).
Pavuk, M. et al. Environmental exposure to PCBs and cancer incidence in eastern Slovakia. Chemosphere 54, 1509–1520 (2004).
Safe, S. H. Polychlorinated biphenyls (PCBs): environmental impact, biochemical and toxic responses, and implications for risk assessment. Crit. Rev. Toxicol. 24, 87–149 (1994).
Brown, D. P. Mortality of workers exposed to polychlorinated biphenyls-an update. Arch. Environ. Health 42, 333–339 (1987).
Arsenescu, V., Arsenescu, R. I., King, V., Swanson, H. & Cassis, L. A. Polychlorinated biphenyl-77 induces adipocyte differentiation and proinflammatory adipokines and promotes obesity and atherosclerosis. Environ. Health Perspect. 116, 761–768 (2008).
Singleton, D. W. & Khan, S. A. Xenoestrogen exposure and mechanisms of endocrine disruption. Front. Biosci. 8, s110–s118 (2003).
Colborn, T., Vom Saal, F. S. & Soto, A. M. Developmental effects of endocrine-disrupting chemicals in wildlife and humans. Environ. Health Perspect. 101, 378 (1993).
Colciago, A. et al. Chronic treatment with polychlorinated biphenyls (PCB) during pregnancy and lactation in the rat Part 2: Effects on reproductive parameters, on sex behavior, on memory retention and on hypothalamic expression of aromatase and 5alpha-reductases in the offspring. Toxicol. Appl. Pharmacol. 239, 46–54 (2009).
Erickson, M. D. & Kaley, R. G., 2nd. Applications of polychlorinated biphenyls. Environ. Sci. Pollut. Res. Int. 18, 135–151 (2011).
Safe, S. et al. PCBs: structure-function relationships and mechanism of action. Environ. Health Perspect. 60, 47 (1985).
Ghavami, R. & Sajadi, S. M. Semi-empirical topological method for prediction of the relative retention time of polychlorinated biphenyl congeners on 18 different HR GC columns. Chromatographia 72, 523–533 (2010).
Leek, J. T. et al. Tackling the widespread and critical impact of batch effects in high-throughput data. Nat. Rev. Genet. 11, 733–739 (2010).
Bachour, G., Failing, K., Georgii, S., Elmadfa, I. & Brunn, H. Species and organ dependence of PCB contamination in fish, foxes, roe deer, and humans. Arch. Environ. Contam. Toxicol. 35, 666–673 (1998).
Choi, J. K., Yu, U., Kim, S. & Yoo, O. J. Combining multiple microarray studies and modeling interstudy variation. Bioinformatics 19 Suppl 1, i84–90 (2003).
Hong, F. et al. RankProd: a bioconductor package for detecting differentially expressed genes in meta-analysis. Bioinformatics 22, 2825–2827 (2006).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl. Acad. Sci. U S A 102, 15545–15550 (2005).
Ok, S., Kang, J. S. & Kim, K. M. Testicular antioxidant mechanism of cultivated wild ginseng extracts. Mol. Cell Tox. 12, 149–158 (2016).
Lauby-Secretan, B. et al. Carcinogenicity of polychlorinated biphenyls and polybrominated biphenyls. Lancet. Oncol. 14, 287 (2013).
Mandal, P. K. Dioxin: a review of its environmental effects and its aryl hydrocarbon receptor biology. J. Comp. Physiol. B 175, 221–230 (2005).
Chang, X. et al. Ligand-independent regulation of transforming growth factor beta1 expression and cell cycle progression by the aryl hydrocarbon receptor. Mol. Cell Biol. 27, 6127–6139 (2007).
Chopra, M., Gahrs, M., Haben, M., Michels, C. & Schrenk, D. Inhibition of apoptosis by 2,3,7,8-tetrachlorodibenzo-p-dioxin depends on protein biosynthesis. Cell Biol. Toxicol. 26, 391–401 (2010).
Andrysik, Z. et al. Aryl hydrocarbon receptor-mediated disruption of contact inhibition is associated with connexin43 downregulation and inhibition of gap junctional intercellular communication. Arch. Toxicol. 87, 491–503 (2013).
Carlson, E. A. et al. Divergent transcriptomic responses to aryl hydrocarbon receptor agonists between rat and human primary hepatocytes. Toxicol. Sci. 112, 257–272 (2009).
De, S. et al. PCB congener specific oxidative stress response by microarray analysis using human liver cell line. Environ. Int. 36, 907–917 (2010).
Sweeney, M. H. & Mocarelli, P. Human health effects after exposure to 2,3,7,8-TCDD. Food Addit. Contam. 17, 303–316 (2000).
Pavek, P. & Dvorak, Z. Xenobiotic-induced transcriptional regulation of xenobiotic metabolizing enzymes of the cytochrome P450 superfamily in human extrahepatic tissues. Curr. Drug Metab. 9, 129–143 (2008).
Misu, H. et al. A liver-derived secretory protein, selenoprotein P, causes insulin resistance. Cell Metab. 12, 483–495 (2010).
Frades, I. et al. Integrative genomic signatures of hepatocellular carcinoma derived from nonalcoholic Fatty liver disease. PLoS One 10, e0124544 (2015).
Wotschofsky, Z. et al. Integrated microRNA and mRNA Signature Associated with the Transition from the Locally Confined to the Metastasized Clear Cell Renal Cell Carcinoma Exemplified by miR-146-5p. PLoS One 11, e0148746 (2016).
Nakagawa, S. et al. Molecular markers of tubulointerstitial fibrosis and tubular cell damage in patients with chronic kidney disease. PLoS One 10, e0136994 (2015).
Barouki, R. & Morel, Y. Repression of cytochrome P450 1A1 gene expression by oxidative stress: mechanisms and biological implications. Biochem. Pharmacol. 61, 511–516 (2001).
Hestermann, E. V., Stegeman, J. J. & Hahn, M. E. Relative contributions of affinity and intrinsic efficacy to aryl hydrocarbon receptor ligand potency. Toxicol. Appl. Pharmacol. 168, 160–172 (2000).
Tilg, H., Moschen, A. R. & Roden, M. NAFLD and diabetes mellitus. Nat. Rev. Gastroenterol. Hepatol. 14, 32–42 (2017).
Cox, M. B. & Miller, C. A., 3rd. Cooperation of heat shock protein 90 and p23 in aryl hydrocarbon receptor signaling. Cell Stress Chaperones 9, 4–20 (2004).
Mannelli, P., Peindl, K. S. & Wu, L. T. Pharmacological enhancement of naltrexone treatment for opioid dependence: a review. Subst. Abuse Rehabil. 2011, 113–123 (2011).
Murray, I. A., Patterson, A. D. & Perdew, G. H. Aryl hydrocarbon receptor ligands in cancer: friend and foe. Nat. Rev. Cancer 14, 801–814 (2014).
Moennikes, O. et al. A constitutively active dioxin/aryl hydrocarbon receptor promotes hepatocarcinogenesis in mice. Cancer Res. 64, 4707–4710 (2004).
Kampinga, H. H. et al. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones 14, 105–111 (2009).
Lee, A. S. The ER chaperone and signaling regulator GRP78/BiP as a monitor of endoplasmic reticulum stress. Methods 35, 373–381 (2005).
Barrett, T. et al. NCBI GEO: archive for functional genomics data sets-update. Nucleic Acids Res. 41, D991–995 (2013).
Carvalho, B. S. & Irizarry, R. A. A framework for oligonucleotide microarray preprocessing. Bioinformatics 26, 2363–2367 (2010).
Bolstad, B. M., Irizarry, R. A., Astrand, M. & Speed, T. P. A comparison of normalization methods for high density oligonucleotide array data based on variance and bias. Bioinformatics 19, 185–193 (2003).
Jung, J., Mok, C., Lee, W. & Jang, W. Meta-analysis of microarray and RNA-Seq gene expression datasets for carcinogenic risk assessment of Bisphenol A. Mol. Cell Tox. 13, 239–249 (2017).
Parker, H. S., Corrada Bravo, H. & Leek, J. T. Removing batch effects for prediction problems with frozen surrogate variable analysis. Peer J 2, e561 (2014).
Ritchie, M. E. et al. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43, e47 (2015).
Szklarczyk, D. et al. STRING v10: protein-protein interaction networks, integrated over the tree of life. Nucleic Acids Res. 43, D447–452 (2015).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
13530_2017_317_MOESM1_ESM.pdf
Meta-analysis of microarray datasets for the risk assessment of coplanar polychlorinated biphenyl 77 (PCB77) on human health
Rights and permissions
About this article
Cite this article
Jung, J., Hah, K., Lee, W. et al. Meta-analysis of microarray datasets for the risk assessment of coplanar polychlorinated biphenyl 77 (PCB77) on human health. Toxicol. Environ. Health Sci. 9, 161–168 (2017). https://doi.org/10.1007/s13530-017-0317-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13530-017-0317-1