Abstract
Purpose
To explore the predictive merit of MFAP2+ cancer associated fibroblasts (CAFs) infiltration for clinical outcomes and adjuvant chemotherapy or immunotherapy responsiveness in gastric cancer (GC).
Methods
In this study, several independent cohorts were included respectively to dissect the relationship of clinical outcomes, therapeutic responses and tumor microenvironment with different MFAP2+ CAFs infiltration. Drug sensitivity analysis was conducted to predict the relationship between MFAP2+ CAFs infiltration and targeted drug response. Kaplan–Meier curves and the log-rank test were used to compare clinical outcomes of patients with different MFAP2+ CAFs infiltration.
Results
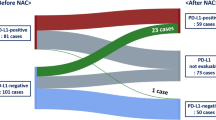
High MFAP2+ CAFs infiltration yielded inferior prognosis in terms of overall survival, progress free survival and recurrence free survival in GC. Patients with low MFAP2+ CAFs infiltration were more likely to gain benefit from adjuvant therapy. Moreover, low MFAP2+ CAFs infiltration could predict a promising response to immunotherapy in GC patients. MFAP2+ CAFs with immunosuppressive features were highly relevant to immune evasive contexture characterized by the dysfunction of CD8+ T cells. We found that MFAP2+ CAFs communicated with T cells, B cells and Macrophages through releasing macrophage migration inhibitor factor (MIF), which further suggested that MFAP2+ CAFs might promote therapeutic resistance through regulating T cells dysfunction and M2 macrophages polarization.
Conclusion
Immunosuppressive MFAP2+ CAFs constructed an immune evasive tumor microenvironment characterized by incapacitated immune effector cells, consequently predicting inferior clinical outcomes and response on adjuvant therapy and immunotherapy in patients with GC. The potential of immunosuppressive MFAP2+ CAFs as a therapeutic target for GC deserved thoroughly exploration.






Similar content being viewed by others
Data availability
Publicly available datasets were analyzed in this study. These data could be found here: UCSC XENA (https://xenabrowser.net/datapages/), GEO (https://www.ncbi.nlm.nih.gov/geo/), Tumor Immune Dysfunction and Exclusion database (http://tide.dfci.harvard.edu) and The Genomics of Drug Sensitivity in Cancer database (https://www.cancerrxgene.org/). Other raw data supporting the conclusions of this article will be available from the corresponding author upon reasonable request.
Abbreviations
- MFAP2 :
-
Microfibril Associated Protein 2
- GC :
-
Gastric cancer
- CAFs :
-
Cancer associated fibroblasts
- ICB :
-
Immune checkpoint blockade
- myCAFs :
-
Myofibroblastic cancer associated fibroblasts
- iCAFs :
-
Inflammatory cancer associated fibroblasts
- TCGA :
-
The Cancer Genome Atlas
- FPKM :
-
Fragments per kilobase million
- TPM :
-
Transcripts per kilobase millions
- ACRG :
-
Asian Cancer Research Group
- SMC :
-
Samsung Medical Center
- TIDE :
-
Tumor Immune Dysfunction and Exclusion
- SD :
-
Stable disease
- PD :
-
Progressive disease
- PR :
-
Partial response
- CR :
-
Complete response
- ScRNA-seq :
-
Single-cell RNA sequencing
- GDSC :
-
Genomics of Drug Sensitivity in Cancer
- IC50 :
-
Half-maximal inhibitory concentration
- ROC :
-
Receiver operating characteristic
- AUC :
-
The area under the curve
- MSI :
-
Microsatellite instability
- GS :
-
Genome stable
- EBV :
-
Epstein-Barr virus
- CIN :
-
Chromosomal instability
- EMT :
-
Epithelial mesenchymal transformation
- Pan-F TBRS :
-
Panfibroblast TGFβ response characteristics
- OS :
-
Overall survival
- RFS :
-
Recurrence free survival
- PFS :
-
Progress free survival
- GEO :
-
Gene Expression Omnibus
- GSEA :
-
Gene set enrichment analysis
- MIF :
-
Macrophage migration inhibitor factor
- TRG :
-
Tumor regression grade
References
H. Sung, J. Ferlay, R.L. Siegel, M. Laversanne, I. Soerjomataram, A. Jemal, F. Bray, CA Cancer J Clin 71, 209–249 (2021). https://doi.org/10.3322/caac.21660
S.S. Joshi, B.D. Badgwell, CA Cancer J Clin 71, 264–279 (2021). https://doi.org/10.3322/caac.21657
J.A. Ajani, T.A. D’Amico, D.J. Bentrem, J. Chao, D. Cooke, C. Corvera, P. Das, P.C. Enzinger, T. Enzler, P. Fanta, F. Farjah, H. Gerdes, M.K. Gibson, S. Hochwald, W.L. Hofstetter, D.H. Ilson, R.N. Keswani, S. Kim, L.R. Kleinberg, S.J. Klempner, J. Lacy, Q.P. Ly, K.A. Matkowskyj, M. McNamara, M.F. Mulcahy, D. Outlaw, H. Park, K.A. Perry, J. Pimiento, G.A. Poultsides, S. Reznik, R.E. Roses, V.E. Strong, S. Su, H.L. Wang, G. Wiesner, C.G. Willett, D. Yakoub, H. Yoon, N. McMillian, L.A. Pluchino, J Natl Compr Canc Netw 20, 167–192 (2022). https://doi.org/10.6004/jnccn.2022.0008
Y. Zhang, Z. Zhang, Cell Mol Immunol 17, 807–821 (2020). https://doi.org/10.1038/s41423-020-0488-6
D.C. Hinshaw, L.A. Shevde, Cancer Res 79, 4557–4566 (2019). https://doi.org/10.1158/0008-5472.CAN-18-3962
M. Nurmik, P. Ullmann, F. Rodriguez, S. Haan, E. Letellier, Int J Cancer 146, 895–905 (2020). https://doi.org/10.1002/ijc.32193
M.Q. Gao, B.G. Kim, S. Kang, Y.P. Choi, H. Park, K.S. Kang, N.H. Cho, J Cell Sci 123, 3507–3514 (2010). https://doi.org/10.1242/jcs.072900
S. Su, J. Chen, H. Yao, J. Liu, S. Yu, L. Lao, M. Wang, M. Luo, Y. Xing, F. Chen, D. Huang, J. Zhao, L. Yang, D. Liao, F. Su, M. Li, Q. Liu, E. Song, Cell 172, 841-856 e816 (2018). https://doi.org/10.1016/j.cell.2018.01.009
S. Zhu, L. Ye, S. Bennett, H. Xu, D. He, J. Xu, J Cell Physiol 236, 41–48 (2021). https://doi.org/10.1002/jcp.29893
F. Segade, N. Suganuma, J.C. Mychaleckyj, R.P. Mecham, Int J Biochem Cell Biol 39, 2303–2313 (2007). https://doi.org/10.1016/j.biocel.2007.06.017
J.K. Wang, W.J. Wang, H.Y. Cai, B.B. Du, P. Mai, L.J. Zhang, W. Ma, Y.G. Hu, S.F. Feng, G.Y. Miao, Onco Targets Ther 11, 4001–4017 (2018). https://doi.org/10.2147/OTT.S160831
L.W. Yao, L.L. Wu, L.H. Zhang, W. Zhou, L. Wu, K. He, J.C. Ren, Y.C. Deng, D.M. Yang, J. Wang, G.G. Mu, M. Xu, J. Zhou, G.A. Xiang, Q.S. Ding, Y.N. Yang, H.G. Yu, Oncogenesis 9, 17 (2020). https://doi.org/10.1038/s41389-020-0198-z
X. Zhu, Y. Cheng, F. Wu, H. Sun, W. Zheng, W. Jiang, J. Shi, S. Ma, H. Cao, Technol Cancer Res Treat 19, 1533033820977524 (2020). https://doi.org/10.1177/1533033820977524
I. Gomez de Segura, P. Ahechu, J. Gomez-Ambrosi, A. Rodriguez, B. Ramirez, S. Becerril, X. Unamuno, A. Mentxaka, J. Baixauli, V. Valenti, R. Moncada, C. Silva, G. Fruhbeck, V. Catalan. Int J Mol Sci 22, 8485 (2021). https://doi.org/10.3390/ijms22168485
Z.N. Liu, Y.K. Wang, L. Zhang, Y.N. Jia, S. Fei, X.J. Ying, Y. Zhang, S.X. Li, Y. Sun, Z.Y. Li, J.F. Ji, World J Gastrointest Oncol 13, 2161–2179 (2021). https://doi.org/10.4251/wjgo.v13.i12.2161
R. Cristescu, J. Lee, M. Nebozhyn, K.M. Kim, J.C. Ting, S.S. Wong, J. Liu, Y.G. Yue, J. Wang, K. Yu, X.S. Ye, I.G. Do, S. Liu, L. Gong, J. Fu, J.G. Jin, M.G. Choi, T.S. Sohn, J.H. Lee, J.M. Bae, S.T. Kim, S.H. Park, I. Sohn, S.H. Jung, P. Tan, R. Chen, J. Hardwick, W.K. Kang, M. Ayers, D. Hongyue, C. Reinhard, A. Loboda, S. Kim, A. Aggarwal, Nat Med 21, 449–456 (2015). https://doi.org/10.1038/nm.3850
J. Lee, I. Sohn, I.G. Do, K.M. Kim, S.H. Park, J.O. Park, Y.S. Park, H.Y. Lim, T.S. Sohn, J.M. Bae, M.G. Choi, D.H. Lim, B.H. Min, J.H. Lee, P.L. Rhee, J.J. Kim, D.I. Choi, I.B. Tan, K. Das, P. Tan, S.H. Jung, W.K. Kang, S. Kim, PLoS One 9, e90133 (2014). https://doi.org/10.1371/journal.pone.0090133
S.C. Oh, B.H. Sohn, J.H. Cheong, S.B. Kim, J.E. Lee, K.C. Park, S.H. Lee, J.L. Park, Y.Y. Park, H.S. Lee, H.J. Jang, E.S. Park, S.C. Kim, J. Heo, I.S. Chu, Y.J. Jang, Y.J. Mok, W. Jung, B.H. Kim, A. Kim, J.Y. Cho, J.Y. Lim, Y. Hayashi, S. Song, E. Elimova, J.S. Estralla, J.H. Lee, M.S. Bhutani, Y. Lu, W. Liu, J. Lee, W.K. Kang, S. Kim, S.H. Noh, G.B. Mills, S.Y. Kim, J.A. Ajani, J.S. Lee, Nat Commun 9, 1777 (2018). https://doi.org/10.1038/s41467-018-04179-8
S.T. Kim, R. Cristescu, A.J. Bass, K.M. Kim, J.I. Odegaard, K. Kim, X.Q. Liu, X. Sher, H. Jung, M. Lee, S. Lee, S.H. Park, J.O. Park, Y.S. Park, H.Y. Lim, H. Lee, M. Choi, A. Talasaz, P.S. Kang, J. Cheng, A. Loboda, J. Lee, W.K. Kang, Nat Med 24, 1449–1458 (2018). https://doi.org/10.1038/s41591-018-0101-z
T.N. Gide, C. Quek, A.M. Menzies, A.T. Tasker, P. Shang, J. Holst, J. Madore, S.Y. Lim, R. Velickovic, M. Wongchenko, Y. Yan, S. Lo, M.S. Carlino, A. Guminski, R.P.M. Saw, A. Pang, H.M. McGuire, U. Palendira, J.F. Thompson, H. Rizos, I.P.D. Silva, M. Batten, R.A. Scolyer, G.V. Long, J.S. Wilmott, Cancer Cell 35, 238-255 e236 (2019). https://doi.org/10.1016/j.ccell.2019.01.003
D. Maeser, R.F. Gruener and R.S. Huang, Brief Bioinform 22, bbab260 (2021). https://doi.org/10.1093/bib/bbab260
R. Kanzaki, K. Pietras, Cancer Sci 111, 2708–2717 (2020). https://doi.org/10.1111/cas.14537
O. Serra, M. Galan, M.M. Ginesta, M. Calvo, N. Sala, R. Salazar, Cancer Treat Rev 77, 29–34 (2019). https://doi.org/10.1016/j.ctrv.2019.05.005
G. Knight, C.C. Earle, R. Cosby, N. Coburn, Y. Youssef, R. Malthaner, R.K. Wong, G. Gastrointestinal Cancer Disease Site, Gastric Cancer, Gastric Cancer 16, 28–40 (2013). https://doi.org/10.1007/s10120-012-0148-3
G. Group, X. Paoletti, K. Oba, T. Burzykowski, S. Michiels, Y. Ohashi, J.P. Pignon, P. Rougier, J. Sakamoto, D. Sargent, M. Sasako, E. Van Cutsem, M. Buyse, JAMA 303(1729), 1737 (2010). https://doi.org/10.1001/jama.2010.534
R. Kalluri, Nat Rev Cancer 16, 582–598 (2016). https://doi.org/10.1038/nrc.2016.73
Y. Shi, L. Du, L. Lin, Y. Wang, Nat Rev Drug Discov 16, 35–52 (2017). https://doi.org/10.1038/nrd.2016.193
A. Cats, E.P.M. Jansen, N.C.T. van Grieken, K. Sikorska, P. Lind, M. Nordsmark, E. Meershoek-Klein Kranenbarg, H. Boot, A.K. Trip, H.A.M. Swellengrebel, H.W.M. van Laarhoven, H. Putter, J.W. van Sandick, M.I. van Berge Henegouwen, H.H. Hartgrink, H. van Tinteren, C.J.H. van de Velde, M. Verheij, Lancet Oncol 19, 616–628 (2018). https://doi.org/10.1016/s1470-2045(18)30132-3
S.H. Park, T.S. Sohn, J. Lee, D.H. Lim, M.E. Hong, K.M. Kim, I. Sohn, S.H. Jung, M.G. Choi, J.H. Lee, J.M. Bae, S. Kim, S.T. Kim, J.O. Park, Y.S. Park, H.Y. Lim, W.K. Kang, J Clin Oncol 33, 3130–3136 (2015). https://doi.org/10.1200/JCO.2014.58.3930
C.D. Coldren, B.A. Helfrich, S.E. Witta, M. Sugita, R. Lapadat, C. Zeng, A. Baron, W.A. Franklin, F.R. Hirsch, M.W. Geraci, P.A. Bunn Jr., Mol Cancer Res 4, 521–528 (2006). https://doi.org/10.1158/1541-7786.MCR-06-0095
J.S. O’Donnell, M.W.L. Teng, M.J. Smyth, Nat Rev Clin Oncol 16, 151–167 (2019). https://doi.org/10.1038/s41571-018-0142-8
R. Shi, Z. Zhang, A. Zhu, X. Xiong, J. Zhang, J. Xu, M.S. Sy, C. Li, Int J Cancer 151, 665–683 (2022). https://doi.org/10.1002/ijc.33985
P. Jiang, S. Gu, D. Pan, J. Fu, A. Sahu, X. Hu, Z. Li, N. Traugh, X. Bu, B. Li, J. Liu, G.J. Freeman, M.A. Brown, K.W. Wucherpfennig, X.S. Liu, Nat Med 24, 1550–1558 (2018). https://doi.org/10.1038/s41591-018-0136-1
Q. Zhou, X. Yan, J. Gershan, R.J. Orentas, B.D. Johnson, J Immunol 181, 1877–1886 (2008). https://doi.org/10.4049/jimmunol.181.3.1877
M. Mittelbronn, M. Platten, P. Zeiner, Y. Dombrowski, B. Frank, C. Zachskorn, P.N. Harter, M. Weller, J. Wischhusen, Acta Neuropathol 122, 353–365 (2011). https://doi.org/10.1007/s00401-011-0858-3
K.D. Simpson, D.J. Templeton, J.V. Cross, J Immunol 189, 5533–5540 (2012). https://doi.org/10.4049/jimmunol.1201161
T. Vierbuchen, A. Ostermeier, Z.P. Pang, Y. Kokubu, T.C. Sudhof, M. Wernig, Nature 463, 1035–1041 (2010). https://doi.org/10.1038/nature08797
E. Sahai, I. Astsaturov, E. Cukierman, D.G. DeNardo, M. Egeblad, R.M. Evans, D. Fearon, F.R. Greten, S.R. Hingorani, T. Hunter, R.O. Hynes, R.K. Jain, T. Janowitz, C. Jorgensen, A.C. Kimmelman, M.G. Kolonin, R.G. Maki, R.S. Powers, E. Pure, D.C. Ramirez, R. Scherz-Shouval, M.H. Sherman, S. Stewart, T.D. Tlsty, D.A. Tuveson, F.M. Watt, V. Weaver, A.T. Weeraratna, Z. Werb, Nat Rev Cancer 20, 174–186 (2020). https://doi.org/10.1038/s41568-019-0238-1
G. Biffi, D.A. Tuveson, Physiol Rev 101, 147–176 (2021). https://doi.org/10.1152/physrev.00048.2019
S. Hu, H. Lu, W. Xie, D. Wang, Z. Shan, X. Xing, X.M. Wang, J. Fang, W. Dong, W. Dai, J. Guo, Y. Zhang, S. Wen, X.Y. Guo, Q. Chen, F. Bai, Z. Wang, J Clin Invest (2022). https://doi.org/10.1172/JCI157649
X. Li, Z. Sun, G. Peng, Y. Xiao, J. Guo, B. Wu, X. Li, W. Zhou, J. Li, Z. Li, C. Bai, L. Zhao, Q. Han, R.C. Zhao, X. Wang, Theranostics 12, 620–638 (2022). https://doi.org/10.7150/thno.60540
X.L. Zhang, L.P. Hu, Q. Yang, W.T. Qin, X. Wang, C.J. Xu, G.A. Tian, X.M. Yang, L.L. Yao, L. Zhu, H.Z. Nie, Q. Li, Q. Xu, Z.G. Zhang, Y.L. Zhang, J. Li, Y.H. Wang, S.H. Jiang, Oncogene 40, 3959–3973 (2021). https://doi.org/10.1038/s41388-021-01827-0
J. Lee, J. Song, E.S. Kwon, S. Jo, M.K. Kang, Y.J. Kim, Y. Hwang, H. Bae, T.H. Kang, S. Chang, H.J. Cho, S.C. Kim, S. Kim, S.S. Koh, Exp Mol Med 48, e261 (2016). https://doi.org/10.1038/emm.2016.87
S. Qin, J.H. Zheng, Z.H. Xia, J. Qian, C.L. Deng, S.L. Yang, Biomed Pharmacother 113, 108594 (2019). https://doi.org/10.1016/j.biopha.2019.01.055
L. Fang, Y. Che, C. Zhang, J. Huang, Y. Lei, Z. Lu, N. Sun, J. He, Cell Death Discov 7, 32 (2021). https://doi.org/10.1038/s41420-021-00410-6
S. Ciavarella, A. Laurenzana, S. De Summa, B. Pilato, A. Chilla, R. Lacalamita, C. Minoia, F. Margheri, A. Iacobazzi, A. Rana, F. Merchionne, G. Fibbi, M. Del Rosso, A. Guarini, S. Tommasi, S. Serrati, BMC Cancer 17, 215 (2017). https://doi.org/10.1186/s12885-017-3183-y
G. Friedman, O. Levi-Galibov, E. David, C. Bornstein, A. Giladi, M. Dadiani, A. Mayo, C. Halperin, M. Pevsner-Fischer, H. Lavon, S. Mayer, R. Nevo, Y. Stein, N. Balint-Lahat, I. Barshack, H.R. Ali, C. Caldas, E. Nili-Gal-Yam, U. Alon, I. Amit, R. Scherz-Shouval, Nat Cancer 1, 692–708 (2020). https://doi.org/10.1038/s43018-020-0082-y
T. Sakai, K. Aokage, S. Neri, H. Nakamura, S. Nomura, K. Tane, T. Miyoshi, M. Sugano, M. Kojima, S. Fujii, T. Kuwata, A. Ochiai, A. Iyoda, M. Tsuboi, G. Ishii, Lung Cancer 126, 64–71 (2018). https://doi.org/10.1016/j.lungcan.2018.10.021
J. Suzuki, K. Aokage, S. Neri, T. Sakai, H. Hashimoto, Y. Su, S. Yamazaki, H. Nakamura, K. Tane, T. Miyoshi, M. Sugano, M. Kojima, S. Fujii, T. Kuwata, A. Ochiai, M. Tsuboi, G. Ishii, Lung Cancer 153, 1–10 (2021). https://doi.org/10.1016/j.lungcan.2020.12.020
H. Su, N. Na, X. Zhang, Y. Zhao, Inflamm Res 66, 209–216 (2017). https://doi.org/10.1007/s00011-016-0995-1
M. de Barbosa Souza Rizzo, M. de Brasilino Carvalho, E.J. Kim, B.E. Rendon, J.T. Noe, A. Darlene Wise, R.A. Mitchell, QJM 111(769), 778 (2018). https://doi.org/10.1093/qjmed/hcy163
F. Garrido, N. Aptsiauri, E.M. Doorduijn, A.M. Garcia Lora, T. van Hall, Curr Opin Immunol 39, 44–51 (2016). https://doi.org/10.1016/j.coi.2015.12.007
Funding
This study was sponsored by Natural Science Foundation of Shanghai (21ZR1414600), Shanghai Pujiang Program (2019PJD007), Shanghai Sailing Program (20YF1409200) and National Natural Science Foundation of China (82002545). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of manuscript.
Author information
Authors and Affiliations
Contributions
LXW and LCC conceived and designed this study. WRY, SJQ, LXJ, HSY analyzed the data and drafted the manuscript. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The studies involving human participants were reviewed and approved by Clinical Research Ethics Committee of Fudan University Shanghai Cancer Center, and the patients/participants offered their written informed consent to participate in this study.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information


Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wei, R., Song, J., Liu, X. et al. Immunosuppressive MFAP2+ cancer associated fibroblasts conferred unfavorable prognosis and therapeutic resistance in gastric cancer. Cell Oncol. 47, 55–68 (2024). https://doi.org/10.1007/s13402-023-00849-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-023-00849-y