Abstract
Background
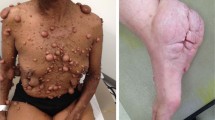
Neurofibromatosis type 1 (NF1) is an autosomal dominant genetic disorder that affects nearly 1 in 3000 infants. Neurofibromin inactivation and NF1 gene mutations are involved in various aspects of neuronal function regulation, including neuronal development induction, electrophysiological activity elevation, growth factor expression, and neurotransmitter release. NF1 patients often exhibit a predisposition to tumor development, especially in the nervous system, resulting in the frequent occurrence of peripheral nerve sheath tumors and gliomas. Recent evidence suggests that nerves play a role in the development of multiple tumor types, prompting researchers to investigate the nerve as a vital component in and regulator of the initiation and progression of NF1-related nervous system tumors.
Conclusion
In this review, we summarize existing evidence about the specific effects of NF1 mutation on neurons and emerging research on the role of nerves in neurological tumor development, promising a new set of selective and targeted therapies for NF1-related tumors.


Similar content being viewed by others
References
P.J. Cimino, D.H. Gutmann, Neurofibromatosis type 1, Handbook of clinical neurology 148, 799–811 (2018) https://doi.org/10.1016/b978-0-444-64076-5.00051-x
J.M. Friedman, Epidemiology of neurofibromatosis type 1. Am. J. Med. Genet. 89, 1–6 (1999)
E. Rad, A.R. Tee, Neurofibromatosis type 1: Fundamental insights into cell signalling and cancer. Semin. Cell Dev. Biol. 52, 39–46 (2016). https://doi.org/10.1016/j.semcdb.2016.02.007
G.A. Mashour, P.H. Driever, M. Hartmann, S.N. Drissel, T. Zhang, B. Scharf, U. Felderhoff-Müser, S. Sakuma, R.E. Friedrich, R.L. Martuza, V.F. Mautner, A. Kurtz, Circulating growth factor levels are associated with tumorigenesis in neurofibromatosis type 1. Clin. cancer research: official J. Am. Association Cancer Res. 10, 5677–5683 (2004). https://doi.org/10.1158/1078-0432.Ccr-03-0769
D.H. Gutmann, R.E. Ferner, R.H. Listernick, B.R. Korf, P.L. Wolters, K.J. Johnson, Neurofibromatosis type 1. Nat. Rev. Dis. Primers 3, 17004 (2017). https://doi.org/10.1038/nrdp.2017.4
T. Tucker, P. Wolkenstein, J. Revuz, J. Zeller, J.M. Friedman, Association between benign and malignant peripheral nerve sheath tumors in NF1, Neurology 65, 205–211 (2005) https://doi.org/10.1212/01.wnl.0000168830.79997.13
B.N. Somatilaka, A. Sadek, R.M. McKay, L.Q. Le, Malignant peripheral nerve sheath tumor: models, biology, and translation. Oncogene 41, 2405–2421 (2022). https://doi.org/10.1038/s41388-022-02290-1
O.O. Seminog, M.J. Goldacre, Risk of benign tumours of nervous system, and of malignant neoplasms, in people with neurofibromatosis: population-based record-linkage study. Br. J. Cancer 108, 193–198 (2013). https://doi.org/10.1038/bjc.2012.535
G. Blanchard, M.P. Lafforgue, L. Lion-François, I. Kemlin, D. Rodriguez, P. Castelnau, M. Carneiro, P. Meyer, F. Rivier, S. Barbarot, Y. Chaix, Systematic MRI in NF1 children under six years of age for the diagnosis of optic pathway gliomas. Study and outcome of a French cohort. Eur. J. Pediatr. neurology: EJPN : official J. Eur. Pediatr. Neurol. Soc. 20, 275–281 (2016). https://doi.org/10.1016/j.ejpn.2015.12.002
C.J. Campen, D.H. Gutmann, Optic Pathway Gliomas in Neurofibromatosis Type 1. J. Child. Neurol. 33, 73–81 (2018). https://doi.org/10.1177/0883073817739509
F. D’Angelo, M. Ceccarelli, L. Tala, J. Garofano, V. Zhang, F.P. Frattini, G. Caruso, K.D. Lewis, L. Alfaro, G. Bauchet, D. Berzero, M. Cachia, L. Cangiano, J. Capelle, F. de Groot, F. DiMeco, W. Ducray, G. Farah, S. Finocchiaro, C. Goutagny, C. Kamiya-Matsuoka, H. Lavarino, V. Loiseau, C.E. Lorgis, I. Marras, D.H. McCutcheon, S. Nam, V. Ronchi, R. Saletti, J. Seizeur, M. Slopis, F. Suñol, P. Vandenbos, D. Varlet, C. Vidaud, V. Watts, D.E. Tabar, S.K. Reuss, D. Kim, K. Meyronet, H. Mokhtari, K.P. Salvador, M. Bhat, M. Eoli, A. Sanson, Lasorella, A. Iavarone, The molecular landscape of glioma in patients with Neurofibromatosis 1. Nat. Med. 25, 176–187 (2019). https://doi.org/10.1038/s41591-018-0263-8
A.H. Zahalka, P.S. Frenette, Nerves in cancer, Nat. Rev. Cancer 20, 143–157 (2020). https://doi.org/10.1038/s41568-019-0237-2
S. Deborde, T. Omelchenko, A. Lyubchik, Y. Zhou, S. He, W.F. McNamara, N. Chernichenko, S.Y. Lee, F. Barajas, C.H. Chen, R.L. Bakst, E. Vakiani, S. He, A. Hall, R.J. Wong, Schwann cells induce cancer cell dispersion and invasion. J. Clin. Investig. 126, 1538–1554 (2016). https://doi.org/10.1172/jci82658
C. Hutchings, J.A. Phillips, M.B.A. Djamgoz, Nerve input to tumours: Pathophysiological consequences of a dynamic relationship. Biochim. Biophys. Acta Rev. Cancer 1874, 188411 (2020). https://doi.org/10.1016/j.bbcan.2020.188411
H.D. Reavis, H.I. Chen, R. Drapkin, Tumor Innervation: Cancer Has Some Nerve. Trends Cancer 6, 1059–1067 (2020). https://doi.org/10.1016/j.trecan.2020.07.005
C. Magnon, S.J. Hall, J. Lin, X. Xue, L. Gerber, S.J. Freedland, P.S. Frenette, Autonomic nerve development contributes to prostate cancer progression. Sci. (New York N Y ) 341, 1236361 (2013). https://doi.org/10.1126/science.1236361
H.C. Ko, V. Gupta, W.F. Mourad, K.S. Hu, L.B. Harrison, P.M. Som, R.L. Bakst, A contouring guide for head and neck cancers with perineural invasion. Pract. Radiat. Oncol. 4, e247–e258 (2014). https://doi.org/10.1016/j.prro.2014.02.001
G. Rademakers, N. Vaes, S. Schonkeren, A. Koch, K.A. Sharkey, V. Melotte, The role of enteric neurons in the development and progression of colorectal cancer, Biochim. Biophys. Acta Rev. Cancer 1868, 420–434 (2017). https://doi.org/10.1016/j.bbcan.2017.08.003
S. Faulkner, P. Jobling, B. March, C.C. Jiang, H. Hondermarck, Tumor Neurobiology and the War of Nerves in Cancer. Cancer Discov. 9, 702–710 (2019). https://doi.org/10.1158/2159-8290.Cd-18-1398
C. Jiang, R.M. McKay, L.Q. Le, Tumorigenesis in neurofibromatosis type 1: role of the microenvironment. Oncogene 40, 5781–5787 (2021). https://doi.org/10.1038/s41388-021-01979-z
N.H. Boyd, A.N. Tran, J.D. Bernstock, T. Etminan, A.B. Jones, G.Y. Gillespie, G.K. Friedman, A.B. Hjelmeland, Glioma stem cells and their roles within the hypoxic tumor microenvironment. Theranostics 11, 665–683 (2021). https://doi.org/10.7150/thno.41692
Y.P. Hsueh, Neurofibromin signaling and synapses. J. Biomed. Sci. 14, 461–466 (2007). https://doi.org/10.1007/s11373-007-9158-2
A.B. Trovó-Marqui, E.H. Tajara, Neurofibromin: a general outlook. Clin. Genet. 70, 1–13 (2006). https://doi.org/10.1111/j.1399-0004.2006.00639.x
J.A. Brown, K.A. Diggs-Andrews, S.M. Gianino, D.H. Gutmann, Neurofibromatosis-1 heterozygosity impairs CNS neuronal morphology in a cAMP/PKA/ROCK-dependent manner, Mol. Cell. Neurosci. 49, 13–22 (2012) https://doi.org/10.1016/j.mcn.2011.08.008
B. Rico, H.E. Beggs, D. Schahin-Reed, N. Kimes, A. Schmidt, L.F. Reichardt, Control of axonal branching and synapse formation by focal adhesion kinase. Nat. Neurosci. 7, 1059–1069 (2004). https://doi.org/10.1038/nn1317
E. Robles, T.M. Gomez, Focal adhesion kinase signaling at sites of integrin-mediated adhesion controls axon pathfinding. Nat. Neurosci. 9, 1274–1283 (2006). https://doi.org/10.1038/nn1762
M. Endo, T. Yamashita, Inactivation of Ras by p120GAP via focal adhesion kinase dephosphorylation mediates RGMa-induced growth cone collapse. J. neuroscience: official J. Soc. Neurosci. 29, 6649–6662 (2009). https://doi.org/10.1523/jneurosci.0927-09.2009
S. Woo, D.J. Rowan, T.M. Gomez, Retinotopic mapping requires focal adhesion kinase-mediated regulation of growth cone adhesion. J. neuroscience: official J. Soc. Neurosci. 29, 13981–13991 (2009). https://doi.org/10.1523/jneurosci.4028-09.2009
F. Kweh, M. Zheng, E. Kurenova, M. Wallace, V. Golubovskaya, W.G. Cance, Neurofibromin physically interacts with the N-terminal domain of focal adhesion kinase. Mol. Carcinog. 48, 1005–1017 (2009). https://doi.org/10.1002/mc.20552
T. Ozawa, N. Araki, S. Yunoue, H. Tokuo, L. Feng, S. Patrakitkomjorn, T. Hara, Y. Ichikawa, K. Matsumoto, K. Fujii, H. Saya, The neurofibromatosis type 1 gene product neurofibromin enhances cell motility by regulating actin filament dynamics via the Rho-ROCK-LIMK2-cofilin pathway. J. Biol. Chem. 280, 39524–39533 (2005). https://doi.org/10.1074/jbc.M503707200
P.I. Tsai, M. Wang, H.H. Kao, Y.J. Cheng, J.A. Walker, R.H. Chen, C.T. Chien, Neurofibromin mediates FAK signaling in confining synapse growth at Drosophila neuromuscular junctions. J. neuroscience: official J. Soc. Neurosci. 32, 16971–16981 (2012). https://doi.org/10.1523/jneurosci.1756-12.2012
Y.L. Lin, Y.P. Hsueh, Neurofibromin interacts with CRMP-2 and CRMP-4 in rat brain. Biochem. Biophys. Res. Commun. 369, 747–752 (2008). https://doi.org/10.1016/j.bbrc.2008.02.095
S. Patrakitkomjorn, D. Kobayashi, T. Morikawa, M.M. Wilson, N. Tsubota, A. Irie, T. Ozawa, M. Aoki, N. Arimura, K. Kaibuchi, H. Saya, N. Araki, Neurofibromatosis type 1 (NF1) tumor suppressor, neurofibromin, regulates the neuronal differentiation of PC12 cells via its associating protein, CRMP-2. J. Biol. Chem. 283, 9399–9413 (2008). https://doi.org/10.1074/jbc.M708206200
M. Bergoug, M. Doudeau, F. Godin, C. Mosrin, B. Vallée, H. Bénédetti, Neurofibromin Struct. Funct. Regul. Cells 9, (2020) https://doi.org/10.3390/cells9112365
M.E. Önger, B. Delibaş, A.P. Türkmen, E. Erener, B.Z. Altunkaynak, S. Kaplan, The role of growth factors in nerve regeneration. Drug discoveries & therapeutics 10, 285–291 (2017). https://doi.org/10.5582/ddt.2016.01058
K. Cichowski, S. Santiago, M. Jardim, B.W. Johnson, T. Jacks, Dynamic regulation of the Ras pathway via proteolysis of the NF1 tumor suppressor. Genes Dev. 17, 449–454 (2003). https://doi.org/10.1101/gad.1054703
Y.Y. Zhang, T.A. Vik, J.W. Ryder, E.F. Srour, T. Jacks, K. Shannon, D.W. Clapp, Nf1 regulates hematopoietic progenitor cell growth and ras signaling in response to multiple cytokines. J. Exp. Med. 187, 1893–1902 (1998). https://doi.org/10.1084/jem.187.11.1893
K.S. Vogel, C.I. Brannan, N.A. Jenkins, N.G. Copeland, L.F. Parada, Loss of neurofibromin results in neurotrophin-independent survival of embryonic sensory and sympathetic neurons. Cell 82, 733–742 (1995). https://doi.org/10.1016/0092-8674(95)90470-0
S.L. Carroll, M.S. Stonecypher, Tumor suppressor mutations and growth factor signaling in the pathogenesis of NF1-associated peripheral nerve sheath tumors: II. The role of dysregulated growth factor signaling. J. Neuropathol. Exp. Neurol. 64, 1–9 (2005). https://doi.org/10.1093/jnen/64.1.1
Y. Kawachi, H. Maruyama, Y. Ishitsuka, Y. Fujisawa, J. Furuta, Y. Nakamura, E. Ichikawa, M. Furumura, F. Otsuka, NF1 gene silencing induces upregulation of vascular endothelial growth factor expression in both Schwann and non-Schwann cells. Exp. Dermatol. 22, 262–265 (2013). https://doi.org/10.1111/exd.12115
T.A. Mitsiadis, M. Salmivirta, T. Muramatsu, H. Muramatsu, H. Rauvala, E. Lehtonen, M. Jalkanen, I. Thesleff, Expression of the heparin-binding cytokines, midkine (MK) and HB-GAM (pleiotrophin) is associated with epithelial-mesenchymal interactions during fetal development and organogenesis. Dev. (Cambridge England) 121, 37–51 (1995). https://doi.org/10.1242/dev.121.1.37
T. Muramatsu, Midkine and pleiotrophin: two related proteins involved in development, survival, inflammation and tumorigenesis. J. BioChem. 132, 359–371 (2002). https://doi.org/10.1093/oxfordjournals.jbchem.a003231
I. Atallah, A.M. Cieza Rivera, O.M. Rivero Lezcano, L. Tascón-González, C. González-Cortés, C. Diez, T. Tascón, Fernández-Villa, V. Martín, Increased serum concentrations of estrogen-induced growth factors Midkine and FGF2 in NF1 patients with plexiform neurofibroma. Am. J. translational Res. 14, 3180–3188 (2022)
X. Guo, Y. Pan, M. Xiong, S. Sanapala, C. Anastasaki, O. Cobb, S. Dahiya, D.H. Gutmann, Midkine activation of CD8(+) T cells establishes a neuron-immune-cancer axis responsible for low-grade glioma growth. Nat. Commun. 11, 2177 (2020). https://doi.org/10.1038/s41467-020-15770-3
A. Moutal, E.T. Dustrude, R. Khanna, Sensitization of Ion Channels Contributes to Central and Peripheral Dysfunction in Neurofibromatosis Type 1, Mol. Neurobiol. 54, 3342–3349 (2017) https://doi.org/10.1007/s12035-016-9907-1
Y. Wang, J.M. Brittain, S.M. Wilson, C.M. Hingtgen, R. Khanna, Altered calcium currents and axonal growth in NF1 haploinsufficient mice, Transl. Neurosci. 1, 106–114 (2010). https://doi.org/10.2478/v10134-010-0025-8
C.M. Johannessen, E.E. Reczek, M.F. James, H. Brems, E. Legius, K. Cichowski, The NF1 tumor suppressor critically regulates TSC2 and mTOR. Proc. Natl. Acad. Sci. U.S.A. 102, 8573–8578 (2005). https://doi.org/10.1073/pnas.0503224102
M. Rosner, M. Hanneder, N. Siegel, A. Valli, C. Fuchs, M. Hengstschläger, The mTOR pathway and its role in human genetic diseases. Mutat. Res. 659, 284–292 (2008). https://doi.org/10.1016/j.mrrev.2008.06.001
J. Bockaert, P. Marin, mTOR in Brain Physiology and Pathologies. Physiol. Rev. 95, 1157–1187 (2015). https://doi.org/10.1152/physrev.00038.2014
P.Y. Martin, S. Doly, A.M. Hamieh, E. Chapuy, V. Canale, M. Drop, S. Chaumont-Dubel, X. Bantreil, F. Lamaty, A.J. Bojarski, P. Zajdel, A. Eschalier, P. Marin, C. Courteix, mTOR activation by constitutively active serotonin6 receptors as new paradigm in neuropathic pain and its treatment. Prog. Neurobiol. 193, 101846 (2020). https://doi.org/10.1016/j.pneurobio.2020.101846
E. Doucet, K. Grychowska, P. Zajdel, J. Bockaert, P. Marin, C. Bécamel, Blockade of Serotonin 5-HT(6) Receptor Constitutive Activity Alleviates Cognitive Deficits in a Preclinical Model of Neurofibromatosis Type 1, Int. J. Mol. Sci. 22, (2021) https://doi.org/10.3390/ijms221810178
M.J. Ribeiro, I.R. Violante, I. Bernardino, R.A. Edden, M. Castelo-Branco, Abnormal relationship between GABA, neurophysiology and impulsive behavior in neurofibromatosis type 1. Cortex 64, 194–208 (2015) https://doi.org/10.1016/j.cortex.2014.10.019
I.R. Violante, M. Patricio, I. Bernardino, J. Rebola, A.J. Abrunhosa, N. Ferreira, M. Castelo-Branco, GABA deficiency in NF1: A multimodal [11 C]-flumazenil and spectroscopy study. Neurology 87, 897–904 (2016). https://doi.org/10.1212/wnl.0000000000003044
R.M. Costa, N.B. Federov, J.H. Kogan, G.G. Murphy, J. Stern, M. Ohno, R. Kucherlapati, T. Jacks, A.J. Silva, Mechanism for the learning deficits in a mouse model of neurofibromatosis type 1. Nature 415, 526–530 (2002). https://doi.org/10.1038/nature711
Y. Cui, R.M. Costa, G.G. Murphy, Y. Elgersma, Y. Zhu, D.H. Gutmann, L.F. Parada, I. Mody, A.J. Silva, Neurofibromin regulation of ERK signaling modulates GABA release and learning. Cell 135, 549–560 (2008). https://doi.org/10.1016/j.cell.2008.09.060
S. Hilfiker, V.A. Pieribone, A.J. Czernik, H.T. Kao, G.J. Augustine, P. Greengard, Synapsins as regulators of neurotransmitter release. Philos. Trans. R. Soc. Lond. B Biol. Sci. 354, 269–279 (1999). https://doi.org/10.1098/rstb.1999.0378
I.R. Violante, M.J. Ribeiro, R.A. Edden, P. Guimarães, I. Bernardino, J. Rebola, G. Cunha, E. Silva, M. Castelo-Branco, GABA deficit in the visual cortex of patients with neurofibromatosis type 1: genotype-phenotype correlations and functional impact. Brain: a journal of neurology 136, 918–925 (2013). https://doi.org/10.1093/brain/aws368
Y. Wang, G.D. Nicol, D.W. Clapp, C.M. Hingtgen, Sensory neurons from Nf1 haploinsufficient mice exhibit increased excitability. J. Neurophysiol. 94, 3670–3676 (2005). https://doi.org/10.1152/jn.00489.2005
J.H. Duan, Y. Wang, D. Duarte, M.R. Vasko, G.D. Nicol, C.M. Hingtgen, Ras signaling pathways mediate NGF-induced enhancement of excitability of small-diameter capsaicin-sensitive sensory neurons from wildtype but not Nf1+/- mice. Neurosci. Lett. 496, 70–74 (2011). https://doi.org/10.1016/j.neulet.2011.03.083
K.E. Hodgdon, C.M. Hingtgen, G.D. Nicol, Dorsal root ganglia isolated from Nf1+/- mice exhibit increased levels of mRNA expression of voltage-dependent sodium channels. Neuroscience 206, 237–244 (2012). https://doi.org/10.1016/j.neuroscience.2011.12.045
J.H. Duan, K.E. Hodgdon, C.M. Hingtgen, G.D. Nicol, N-type calcium current, Cav2.2, is enhanced in small-diameter sensory neurons isolated from Nf1+/- mice. Neuroscience 270, 192–202 (2014). https://doi.org/10.1016/j.neuroscience.2014.04.021
C. Anastasaki, J. Mo, J.K. Chen, J. Chatterjee, Y. Pan, S.M. Scheaffer, O. Cobb, M. Monje, L.Q. Le, D.H. Gutmann, Neuronal hyperexcitability drives central and peripheral nervous system tumor progression in models of neurofibromatosis-1. Nat. Commun. 13, 2785 (2022). https://doi.org/10.1038/s41467-022-30466-6
J.S. Nix, J. Blakeley, F.J. Rodriguez, An update on the central nervous system manifestations of neurofibromatosis type 1. Acta Neuropathol. 139, 625–641 (2020). https://doi.org/10.1007/s00401-019-02002-2
M. Bayat, A. Bayat, Neurological manifestations of neurofibromatosis: a review. Neurol. Sci. 41, 2685–2690 (2020) https://doi.org/10.1007/s10072-020-04400-x
K.I. Ly, J.O. Blakeley, The diagnosis and management of neurofibromatosis Type 1. Med. Clin. North Am. 103, 1035–1054 (2019) https://doi.org/10.1016/j.mcna.2019.07.004
A. Cannon, M.J. Chen, P. Li, K.P. Boyd, A. Theos, D.T. Redden, B. Korf, Cutaneous neurofibromas in Neurofibromatosis type I: a quantitative natural history study. Orphanet J. Rare Dis. 13, 31 (2018). https://doi.org/10.1186/s13023-018-0772-z
L. Dugoff, E. Sujansky, Neurofibromatosis type 1 and pregnancy. Am. J. Med. Genet. 66, 7–10 (1996) https://doi.org/10.1002/(sici)1096-8628(19961202)66:1(7::Aid-ajmg2)3.0.Co;2-r
R.E. Ferner, Neurofibromatosis 1. Eur. J. Hum. Genet 15, 131–138 (2007). https://doi.org/10.1038/sj.ejhg.5201676
D.G. Evans, M.E. Baser, J. McGaughran, S. Sharif, E. Howard, A. Moran, Malignant peripheral nerve sheath tumours in neurofibromatosis 1. J. Med. Genet. 39, 311–314 (2002). https://doi.org/10.1136/jmg.39.5.311
P. de Robles, K.M. Fiest, A.D. Frolkis, T. Pringsheim, C. Atta, C.St Germaine-Smith, L. Day, D. Lam, N. Jette, The worldwide incidence and prevalence of primary brain tumors: a systematic review and meta-analysis. Neuro-oncology 17, 776–783 (2015). https://doi.org/10.1093/neuonc/nou283
A. Rosenfeld, R. Listernick, J. Charrow, S. Goldman, Neurofibromatosis type 1 and high-grade tumors of the central nervous system. Child’s Nerv. system: ChNS : official J. Int. Soc. Pediatr. Neurosurg. 26, 663–667 (2010). https://doi.org/10.1007/s00381-009-1024-2
D.H. Gutmann, S.A. Rasmussen, P. Wolkenstein, M.M. MacCollin, A. Guha, P.D. Inskip, K.N. North, M. Poyhonen, P.H. Birch, J.M. Friedman, Gliomas presenting after age 10 in individuals with neurofibromatosis type 1 (NF1). Neurology 59, 759–761 (2002). https://doi.org/10.1212/wnl.59.5.759
H.S. Venkatesh, T.B. Johung, V. Caretti, A. Noll, Y. Tang, S. Nagaraja, E.M. Gibson, C.W. Mount, J. Polepalli, S.S. Mitra, P.J. Woo, R.C. Malenka, H. Vogel, M. Bredel, P. Mallick, and M. Monje, Neuronal Activity Promotes Glioma Growth through Neuroligin-3 Secretion. Cell 161, 803–816 (2015). https://doi.org/10.1016/j.cell.2015.04.012
H.S. Venkatesh, L.T. Tam, P.J. Woo, J. Lennon, S. Nagaraja, S.M. Gillespie, J. Ni, D.Y. Duveau, P.J. Morris, J.J. Zhao, C.J. Thomas, M. Monje, Targeting neuronal activity-regulated neuroligin-3 dependency in high-grade glioma. Nature 549, 533–537 (2017). https://doi.org/10.1038/nature24014
H.S. Venkatesh, W. Morishita, A.C. Geraghty, D. Silverbush, S.M. Gillespie, M. Arzt, L.T. Tam, C. Espenel, A. Ponnuswami, L. Ni, P.J. Woo, K.R. Taylor, A. Agarwal, A. Regev, D. Brang, H. Vogel, S. Hervey-Jumper, D.E. Bergles, M.L. Suvà, R.C. Malenka, M. Monje, Electrical and synaptic integration of glioma into neural circuits. Nature 573, 539–545 (2019). https://doi.org/10.1038/s41586-019-1563-y
V. Venkataramani, D.I. Tanev, C. Strahle, A. Studier-Fischer, L. Fankhauser, T. Kessler, C. Körber, M. Kardorff, M. Ratliff, R. Xie, H. Horstmann, M. Messer, S.P. Paik, J. Knabbe, F. Sahm, F.T. Kurz, A.A. Acikgöz, F. Herrmannsdörfer, A. Agarwal, D.E. Bergles, A. Chalmers, H. Miletic, S. Turcan, C. Mawrin, D. Hänggi, H.K. Liu, W. Wick, F. Winkler, T. Kuner, Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573, 532–538 (2019). https://doi.org/10.1038/s41586-019-1564-x
A. Comba, S.M. Faisal, P.J. Dunn, A.E. Argento, T.C. Hollon, W.N. Al-Holou, M.L. Varela, D.B. Zamler, G.L. Quass, P.F. Apostolides, C. Abel 2nd, C.E. Brown, P.E. Kish, A. Kahana, C.G. Kleer, S. Motsch, M.G. Castro, P.R. Lowenstein, Spatiotemporal analysis of glioma heterogeneity reveals COL1A1 as an actionable target to disrupt tumor progression. Nat. Commun. 13, 3606 (2022). https://doi.org/10.1038/s41467-022-31340-1
A. Comba, S.M. Faisal, M.L. Varela, T. Hollon, W.N. Al-Holou, Y. Umemura, F.J. Nunez, S. Motsch, M.G. Castro, P.R. Lowenstein, Uncovering Spatiotemporal Heterogeneity of High-Grade Gliomas: From Disease Biology to Therapeutic Implications. Front. Oncol. 11, 703764 (2021). https://doi.org/10.3389/fonc.2021.703764
A. Mega, M. Hartmark Nilsen, L.W. Leiss, N.P. Tobin, H. Miletic, L. Sleire, C. Strell, S. Nelander, C. Krona, D. Hägerstrand, P. Enger, M. Nistér, A. Östman, Astrocytes enhance glioblastoma growth. Glia 68, 316–327 (2020). https://doi.org/10.1002/glia.23718
D. Hambardzumyan, D.H. Gutmann, H. Kettenmann, The role of microglia and macrophages in glioma maintenance and progression. Nat. Neurosci. 19, 20–27 (2016). https://doi.org/10.1038/nn.4185
J. Wei, P. Chen, P. Gupta, M. Ott, D. Zamler, C. Kassab, K.P. Bhat, M.A. Curran, J.F. de Groot, A.B. Heimberger, Immune biology of glioma-associated macrophages and microglia: functional and therapeutic implications. Neuro-oncology 22, 180–194 (2020). https://doi.org/10.1093/neuonc/noz212
J.C. Carlson, M. Cantu Gutierrez, B. Lozzi, E. Huang-Hobbs, W.D. Turner, B. Tepe, Y. Zhang, A.M. Herman, G. Rao, C.J. Creighton, J.D. Wythe, B. Deneen, Identification of diverse tumor endothelial cell populations in malignant glioma. Neuro-oncology 23, 932–944 (2021). https://doi.org/10.1093/neuonc/noaa297
D. Cahill, S. Turcan, Origin of Gliomas. Semin. Neurol. 38, 5–10 (2018). https://doi.org/10.1055/s-0037-1620238
A.E.S. Watson, K. Goodkey, T. Footz, A. Voronova, Regulation of CNS precursor function by neuronal chemokines. Neurosci. Lett. 715, 134533 (2020). https://doi.org/10.1016/j.neulet.2019.134533
Z.Q. Li, Z.Y. Yan, F.J. Lan, Y.Q. Dong, Y. Xiong, Suppression of NLRP3 inflammasome attenuates stress-induced depression-like behavior in NLGN3-deficient mice. Biochem. Biophys. Res. Commun. 501, 933–940 (2018) https://doi.org/10.1016/j.bbrc.2018.05.085
C. Davey, A. Tallafuss, P. Washbourne, Differential expression of neuroligin genes in the nervous system of zebrafish, Dev. Dyn. 239, 703–714 (2010) https://doi.org/10.1002/dvdy.22195
R. Liu, X.P. Qin, Y. Zhuang, Y. Zhang, H.B. Liao, J.C. Tang, M.X. Pan, F.F. Zeng, Y. Lei, R.X. Lei, S. Wang, A.C. Liu, J. Chen, Z.F. Zhang, D. Zhao, S.L. Wu, R.Z. Liu, Z.F. Wang, Q. Wan, Glioblastoma recurrence correlates with NLGN3 levels. Cancer Med. 7, 2848–2859 (2018). https://doi.org/10.1002/cam4.1538
V. Serafim Junior, G.M.M. Fernandes, J.G. Oliveira-Cucolo, E.C. Pavarino, E.M. Goloni-Bertollo, Role of Tropomyosin-related kinase B receptor and brain-derived neurotrophic factor in cancer. Cytokine 136, 155270 (2020). https://doi.org/10.1016/j.cyto.2020.155270
J. Xiong, L. Zhou, Y. Lim, M. Yang, Y.H. Zhu, Z.W. Li, F.H. Zhou, Z.C. Xiao, X.F. Zhou, Mature BDNF promotes the growth of glioma cells in vitro. Oncol. Rep. 30, 2719–2724 (2013). https://doi.org/10.3892/or.2013.2746
S.H. Jiang, L.P. Hu, X. Wang, J. Li, Z.G. Zhang, Neurotransmitters: emerging targets in cancer. Oncogene 39, 503–515 (2020). https://doi.org/10.1038/s41388-019-1006-0
T.W. Hodo, M.T.P. de Aquino, A. Shimamoto, A. Shanker, Critical Neurotransmitters in the Neuroimmune Network. Front. Immunol. 11, 1869 (2020). https://doi.org/10.3389/fimmu.2020.01869
K.L. Ligon, E. Huillard, S. Mehta, S. Kesari, H. Liu, J.A. Alberta, R.M. Bachoo, M. Kane, D.N. Louis, R.A. Depinho, D.J. Anderson, C.D. Stiles, D.H. Rowitch, Olig2-regulated lineage-restricted pathway controls replication competence in neural stem cells and malignant glioma. Neuron 53, 503–517 (2007). https://doi.org/10.1016/j.neuron.2007.01.009
A.J. Walker, T. Card, T.E. Bates, K. Muir, Tricyclic antidepressants and the incidence of certain cancers: a study using the GPRD. Br. J. Cancer 104, 193–197 (2011). https://doi.org/10.1038/sj.bjc.6605996
J. Li, S. Zhu, D. Kozono, K. Ng, D. Futalan, Y. Shen, J.C. Akers, T. Steed, D. Kushwaha, M. Schlabach, B.S. Carter, C.H. Kwon, F. Furnari, W. Cavenee, S. Elledge, C.C. Chen, Genome-wide shRNA screen revealed integrated mitogenic signaling between dopamine receptor D2 (DRD2) and epidermal growth factor receptor (EGFR) in glioblastoma. Oncotarget 5, 882–893 (2014). https://doi.org/10.18632/oncotarget.1801
M. Tonini, L. Cipollina, E. Poluzzi, F. Crema, G.R. Corazza, F. De Ponti, Review article: clinical implications of enteric and central D2 receptor blockade by antidopaminergic gastrointestinal prokinetics. Aliment. Pharmacol. Ther. 19, 379–390 (2004). https://doi.org/10.1111/j.1365-2036.2004.01867.x
A.S. Chi, R.S. Tarapore, M.D. Hall, N. Shonka, S. Gardner, Y. Umemura, A. Sumrall, Z. Khatib, S. Mueller, C. Kline, W. Zaky, S. Khatua, S.P. Weathers, Y. Odia, T.N. Niazi, D. Daghistani, I. Cherrick, D. Korones, M.A. Karajannis, X.T. Kong, J. Minturn, A. Waanders, I. Arillaga-Romany, T. Batchelor, P.Y. Wen, K. Merdinger, L. Schalop, M. Stogniew, J.E. Allen, W. Oster, M.P. Mehta, Pediatric and adult H3 K27M-mutant diffuse midline glioma treated with the selective DRD2 antagonist ONC201. J. Neurooncol. 145, 97–105 (2019). https://doi.org/10.1007/s11060-019-03271-3
I. Arrillaga-Romany, A.S. Chi, J.E. Allen, W. Oster, P.Y. Wen, T.T. Batchelor, A phase 2 study of the first imipridone ONC201, a selective DRD2 antagonist for oncology, administered every three weeks in recurrent glioblastoma. Oncotarget 8, 79298–79304 (2017). https://doi.org/10.18632/oncotarget.17837
J.E. Allen, G. Krigsfeld, P.A. Mayes, L. Patel, D.T. Dicker, A.S. Patel, N.G. Dolloff, E. Messaris, K.A. Scata, W. Wang, J.Y. Zhou, G.S. Wu, and W.S. El-Deiry, Dual inactivation of Akt and ERK by TIC10 signals Foxo3a nuclear translocation, TRAIL gene induction, and potent antitumor effects. Sci. Transl. Med. 5, 171ra117 (2013). https://doi.org/10.1126/scitranslmed.3004828
A. Arce-Sillas, E. Sevilla-Reyes, D.D. Álvarez-Luquín, A. Guevara-Salinas, M.C. Boll, C.A. Pérez-Correa, A.V. Vivas-Almazan, U. Rodríguez-Ortiz, C. Castellanos Barba, M. Hernandez, G. Fragoso, E. Sciutto, G. Cárdenas, and L.V. Adalid-Peralta, Expression of dopamine receptors in immune regulatory cells. Neuroimmunomodulation 26, 159–166 (2019). https://doi.org/10.1159/000501187
F. McKenna, P.J. McLaughlin, B.J. Lewis, G.C. Sibbring, J.A. Cummerson, D. Bowen-Jones, R.J. Moots, Dopamine receptor expression on human T- and B-lymphocytes, monocytes, neutrophils, eosinophils and NK cells: a flow cytometric study. J. Neuroimmunol. 132, 34–40 (2002). https://doi.org/10.1016/s0165-5728(02)00280-1
J.M. Beaulieu, R.R. Gainetdinov, The physiology, signaling, and pharmacology of dopamine receptors. Pharmacol. Rev. 63, 182–217 (2011). https://doi.org/10.1124/pr.110.002642
K. Bhat, M. Saki, F. Cheng, L. He, L. Zhang, A. Ioannidis, D. Nathanson, J. Tsang, S.J. Bensinger, P.L. Nghiemphu, T.F. Cloughesy, L.M. Liau, H.I. Kornblum, F. Pajonk, Dopamine Receptor Antagonists, Radiation, and Cholesterol Biosynthesis in Mouse Models of Glioblastoma. J. Natl Cancer Inst. 113, 1094–1104 (2021). https://doi.org/10.1093/jnci/djab018
L.F. Mohammad-Zadeh, L. Moses, S.M. Gwaltney-Brant, Serotonin: a review. J. Vet. Pharmacol. Ther. 31, 187–199 (2008). https://doi.org/10.1111/j.1365-2885.2008.00944.x
D. Sarrouilhe, J. Clarhaut, N. Defamie, M. Mesnil, Serotonin and cancer: what is the link? Curr. Mol. Med. 15, 62–77 (2015). https://doi.org/10.2174/1566524015666150114113411
G. Fouquet, T. Coman, O. Hermine, F. Côté, Serotonin, hematopoiesis and stem cells. Pharmacol. Res. 140, 67–74 (2019). https://doi.org/10.1016/j.phrs.2018.08.005
D. Hoyer, D.E. Clarke, J.R. Fozard, P.R. Hartig, G.R. Martin, E.J. Mylecharane, P.R. Saxena, P.P. Humphrey, International Union of Pharmacology classification of receptors for 5-hydroxytryptamine (Serotonin). Pharmacol. Rev. 46, 157–203 (1994)
A. Merzak, S. Koochekpour, M.P. Fillion, G. Fillion, G.J. Pilkington, Expression of serotonin receptors in human fetal astrocytes and glioma cell lines: a possible role in glioma cell proliferation and migration, Brain research. Mol. Brain Res. 41, 1–7 (1996). https://doi.org/10.1016/0169-328x(96)00058-7
B. Abadi, Y. Shahsavani, M. Faramarzpour, N. Rezaei, H.R. Rahimi, Antidepressants with anti-tumor potential in treating glioblastoma: A narrative review. Fundam. Clin. Pharmacol. 36, 35–48 (2022). https://doi.org/10.1111/fcp.12712
K.H. Liu, S.T. Yang, Y.K. Lin, J.W. Lin, Y.H. Lee, J.Y. Wang, C.J. Hu, E.Y. Lin, S.M. Chen, C.K. Then, S.C. Shen, Fluoxetine, an antidepressant, suppresses glioblastoma by evoking AMPAR-mediated calcium-dependent apoptosis. Oncotarget 6, 5088–5101 (2015). https://doi.org/10.18632/oncotarget.3243
V.C. Chen, Y.H. Hsieh, L.J. Chen, T.C. Hsu, B.S. Tzang, Escitalopram oxalate induces apoptosis in U-87MG cells and autophagy in GBM8401 cells. J. Cell. Mol. Med. 22, 1167–1178 (2018). https://doi.org/10.1111/jcmm.13372
C. Shi, N. Lamba, L.J. Zheng, D. Cote, Q.R. Regestein, C.M. Liu, Q. Tran, S. Routh, T.R. Smith, R.A. Mekary, M.L.D. Broekman, Depression and survival of glioma patients: A systematic review and meta-analysis. Clin. Neurol. Neurosurg. 172, 8–19 (2018). https://doi.org/10.1016/j.clineuro.2018.06.016
S. Otto-Meyer, R. DeFaccio, C. Dussold, E. Ladomersky, L. Zhai, K.L. Lauing, L.R. Bollu, C. Amidei, R.V. Lukas, D.M. Scholtens, D.A. Wainwright, A retrospective survival analysis of Glioblastoma patients treated with selective serotonin reuptake inhibitors. Brain Behav. Immun. Health 2, (2020) https://doi.org/10.1016/j.bbih.2019.100025
A. Jussofie, V. Reinhardt, R. Kalff, GABA binding sites: their density, their affinity to muscimol and their behaviour against neuroactive steroids in human gliomas of different degrees of malignancy. J. Neural Transm. Gen. Sections 96, 233–241 (1994). https://doi.org/10.1007/bf01294790
A. Blanchart, R. Fernando, M. Häring, N. Assaife-Lopes, R.A. Romanov, M. Andäng, T. Harkany, P. Ernfors, Endogenous GAB(AA) receptor activity suppresses glioma growth. Oncogene 36, 777–786 (2017). https://doi.org/10.1038/onc.2016.245
S. Han, Y. Liu, S.J. Cai, M. Qian, J. Ding, M. Larion, M.R. Gilbert, C. Yang, IDH mutation in glioma: molecular mechanisms and potential therapeutic targets. Br. J. Cancer 122, 1580–1589 (2020). https://doi.org/10.1038/s41416-020-0814-x
L. Dang, K. Yen, E.C. Attar, IDH mutations in cancer and progress toward development of targeted therapeutics. Annals of oncology: official journal of the European Society for Medical Oncology 27, 599–608 (2016). https://doi.org/10.1093/annonc/mdw013
T. Gong, X. Zhang, X. Wei, S. Yuan, M.G. Saleh, Y. Song, R.A. Edden, G. Wang, GSH and GABA decreases in IDH1-mutated low-grade gliomas detected by HERMES spectral editing at 3 T in vivo. Neurochem. Int. 141, 104889 (2020). https://doi.org/10.1016/j.neuint.2020.104889
A. Rubio-Casillas, A. Fernández-Guasti, The dose makes the poison: from glutamate-mediated neurogenesis to neuronal atrophy and depression. Rev. Neurosci. 27, 599–622 (2016). https://doi.org/10.1515/revneuro-2015-0066
N. Nakamichi, T. Takarada, Y. Yoneda, Neurogenesis mediated by gamma-aminobutyric acid and glutamate signaling. J. Pharmacol. Sci. 110, 133–149 (2009). https://doi.org/10.1254/jphs.08r03cr
Z. Pei, K.C. Lee, A. Khan, G. Erisnor, H.Y. Wang, Pathway analysis of glutamate-mediated, calcium-related signaling in glioma progression. Biochem. Pharmacol. 176, 113814 (2020). https://doi.org/10.1016/j.bcp.2020.113814
A. Reiner, J. Levitz, Glutamatergic Signaling in the Central Nervous System: Ionotropic and Metabotropic Receptors in Concert. Neuron 98, 1080–1098 (2018). https://doi.org/10.1016/j.neuron.2018.05.018
S.S. Shin, J. Namkoong, B.A. Wall, R. Gleason, H.J. Lee, S. Chen, Oncogenic activities of metabotropic glutamate receptor 1 (Grm1) in melanocyte transformation. Pigment Cell Melanoma Res. 21, 368–378 (2008). https://doi.org/10.1111/j.1755-148X.2008.00452.x
F. Gardoni, J. Stanic, D. Scheggia, A. Benussi, B. Borroni and M. Di Luca, NMDA and AMPA Receptor Autoantibodies in Brain Disorders: From Molecular Mechanisms to Clinical Features. Cells 10 (2021) https://doi.org/10.3390/cells10010077
S. Ishiuchi, K. Tsuzuki, Y. Yoshida, N. Yamada, N. Hagimura, H. Okado, A. Miwa, H. Kurihara, Y. Nakazato, M. Tamura, T. Sasaki, S. Ozawa, Blockage of Ca(2+)-permeable AMPA receptors suppresses migration and induces apoptosis in human glioblastoma cells. Nat. Med. 8, 971–978 (2002). https://doi.org/10.1038/nm746
C.M. Niswender, P.J. Conn, Metabotropic glutamate receptors: physiology, pharmacology, and disease. Annu. Rev. Pharmacol. Toxicol. 50, 295–322 (2010). https://doi.org/10.1146/annurev.pharmtox.011008.145533
T. Takano, J.H. Lin, G. Arcuino, Q. Gao, J. Yang, M. Nedergaard, Glutamate release promotes growth of malignant gliomas. Nat. Med. 7, 1010–1015 (2001). https://doi.org/10.1038/nm0901-1010
L.J. Yu, B.A. Wall, J. Wangari-Talbot, S. Chen, Metabotropic glutamate receptors in cancer. Neuropharmacology 115, 193–202 (2017). https://doi.org/10.1016/j.neuropharm.2016.02.011
L. Iacovelli, A. Arcella, G. Battaglia, S. Pazzaglia, E. Aronica, P. Spinsanti, A. Caruso, E. De Smaele, A. Saran, A. Gulino, M. D’Onofrio, F. Giangaspero, F. Nicoletti, Pharmacological activation of mGlu4 metabotropic glutamate receptors inhibits the growth of medulloblastomas. J. neuroscience: official J. Soc. Neurosci. 26, 8388–8397 (2006). https://doi.org/10.1523/jneurosci.2285-06.2006
C. Zhang, X.R. Yuan, H.Y. Li, Z.J. Zhao, Y.W. Liao, X.Y. Wang, J. Su, S.S. Sang, Q. Liu, Anti-cancer effect of metabotropic glutamate receptor 1 inhibition in human glioma U87 cells: involvement of PI3K/Akt/mTOR pathway. Cell. Physiol. Biochem. 35, 419–432 (2015) https://doi.org/10.1159/000369707
M. D’Onofrio, A. Arcella, V. Bruno, R.T. Ngomba, G. Battaglia, V. Lombari, G. Ragona, A. Calogero, F. Nicoletti, Pharmacological blockade of mGlu2/3 metabotropic glutamate receptors reduces cell proliferation in cultured human glioma cells. J. Neurochem. 84, 1288–1295 (2003). https://doi.org/10.1046/j.1471-4159.2003.01633.x
M.S.L. Pereira, F. Klamt, C.C. Thomé, P.V. Worm, D.L. de Oliveira, Metabotropic glutamate receptors as a new therapeutic target for malignant gliomas. Oncotarget 8, 22279–22298 (2017). https://doi.org/10.18632/oncotarget.15299
P.F. Behrens, H. Langemann, R. Strohschein, J. Draeger, J. Hennig, Extracellular glutamate and other metabolites in and around RG2 rat glioma: an intracerebral microdialysis study. J. Neurooncol. 47, 11–22 (2000). https://doi.org/10.1023/a:1006426917654
R. Listernick, R.E. Ferner, G.T. Liu, D.H. Gutmann, Optic pathway gliomas in neurofibromatosis-1: controversies and recommendations. Ann. Neurol. 61, 189–198 (2007). https://doi.org/10.1002/ana.21107
G.C. Daginakatte, D.H. Gutmann, Neurofibromatosis-1 (Nf1) heterozygous brain microglia elaborate paracrine factors that promote Nf1-deficient astrocyte and glioma growth. Hum. Mol. Genet. 16, 1098–1112 (2007). https://doi.org/10.1093/hmg/ddm059
M.E. Freret, D.H. Gutmann, Insights into optic pathway glioma vision loss from mouse models of neurofibromatosis type 1. J. Neurosci. Res. 97, 45–56 (2019). https://doi.org/10.1002/jnr.24250
W.W. Pong, S.B. Higer, S.M. Gianino, R.J. Emnett, D.H. Gutmann, Reduced microglial CX3CR1 expression delays neurofibromatosis-1 glioma formation. Ann. Neurol. 73, 303–308 (2013). https://doi.org/10.1002/ana.23813
A.C. Solga, W.W. Pong, K.Y. Kim, P.J. Cimino, J.A. Toonen, J. Walker, T. Wylie, V. Magrini, M. Griffith, O.L. Griffith, A. Ly, M.H. Ellisman, E.R. Mardis, D.H. Gutmann, RNA Sequencing of Tumor-Associated Microglia Reveals Ccl5 as a Stromal Chemokine Critical for Neurofibromatosis-1 Glioma Growth. Neoplasia 17, 776–788 (2015) https://doi.org/10.1016/j.neo.2015.10.002
C.Yu-Ju Wu, C.H. Chen, C.Y. Lin, L.Y. Feng, Y.C. Lin, K.C. Wei, C.Y. Huang, J.Y. Fang, P.Y. Chen, CCL5 of glioma-associated microglia/macrophages regulates glioma migration and invasion via calcium-dependent matrix metalloproteinase 2. Neuro-oncology 22, 253–266 (2020). https://doi.org/10.1093/neuonc/noz189
Y. Pan, M. Xiong, R. Chen, Y. Ma, C. Corman, M. Maricos, U. Kindler, M. Semtner, Y.H. Chen, S. Dahiya, D.H. Gutmann, Athymic mice reveal a requirement for T-cell-microglia interactions in establishing a microenvironment supportive of Nf1 low-grade glioma growth. Genes Dev. 32, 491–496 (2018). https://doi.org/10.1101/gad.310797.117
Y.H. Chen, L.D. McGowan, P.J. Cimino, S. Dahiya, J.R. Leonard, D.Y. Lee, D.H. Gutmann, Mouse low-grade gliomas contain cancer stem cells with unique molecular and functional properties. Cell Rep. 10, 1899–1912 (2015). https://doi.org/10.1016/j.celrep.2015.02.041
J. Chatterjee, S. Sanapala, O. Cobb, A. Bewley, A.K. Goldstein, E. Cordell, X. Ge, J.R. Garbow, M.J. Holtzman, D.H. Gutmann, Asthma reduces glioma formation by T cell decorin-mediated inhibition of microglia. Nat. Commun. 12, 7122 (2021). https://doi.org/10.1038/s41467-021-27455-6
Y. Pan, J.D. Hysinger, T. Barron, N.F. Schindler, O. Cobb, X. Guo, B. Yalçın, C. Anastasaki, S.B. Mulinyawe, A. Ponnuswami, S. Scheaffer, Y. Ma, K.C. Chang, X. Xia, J.A. Toonen, J.J. Lennon, E.M. Gibson, J.R. Huguenard, L.M. Liau, J.L. Goldberg, M. Monje, D.H. Gutmann, NF1 mutation drives neuronal activity-dependent initiation of optic glioma. Nature 594, 277–282 (2021). https://doi.org/10.1038/s41586-021-03580-6
K.R. Jessen, R. Mirsky, A.C. Lloyd, Schwann Cells: Development and Role in Nerve Repair, Cold Spring Harb. Perspect. Biol. 7, a020487 (2015) https://doi.org/10.1101/cshperspect.a020487
H. Zheng, L. Chang, N. Patel, J. Yang, L. Lowe, D.K. Burns, Y. Zhu, Induction of abnormal proliferation by nonmyelinating schwann cells triggers neurofibroma formation. Cancer cell. 13, 117–128 (2008). https://doi.org/10.1016/j.ccr.2008.01.002
N.M. Joseph, J.T. Mosher, J. Buchstaller, P. Snider, P.E. McKeever, M. Lim, S.J. Conway, L.F. Parada, Y. Zhu, S.J. Morrison, The loss of Nf1 transiently promotes self-renewal but not tumorigenesis by neural crest stem cells. Cancer cell. 13, 129–140 (2008). https://doi.org/10.1016/j.ccr.2008.01.003
S. Parrinello, L.A. Noon, M.C. Harrisingh, P. Wingfield Digby, L.H. Rosenberg, C.A. Cremona, P. Echave, A.M. Flanagan, L.F. Parada, A.C. Lloyd, NF1 loss disrupts Schwann cell-axonal interactions: a novel role for semaphorin 4F. Genes Dev. 22, 3335–3348 (2008). https://doi.org/10.1101/gad.490608
K.J. Radomska, F. Coulpier, A. Gresset, A. Schmitt, A. Debbiche, S. Lemoine, P. Wolkenstein, J.M. Vallat, P. Charnay, P. Topilko, Cellular Origin, Tumor Progression, and Pathogenic Mechanisms of Cutaneous Neurofibromas Revealed by Mice with Nf1 Knockout in Boundary Cap Cells. Cancer Discov. 9, 130–147 (2019). https://doi.org/10.1158/2159-8290.Cd-18-0156
F.L. Rice, G. Houk, J.P. Wymer, S.J.C. Gosline, J. Guinney, J. Wu, N. Ratner, M.P. Jankowski, S. La Rosa, M. Dockum, J.R. Storey, S.L. Carroll, P.J. Albrecht, V.M. Riccardi, The evolution and multi-molecular properties of NF1 cutaneous neurofibromas originating from C-fiber sensory endings and terminal Schwann cells at normal sites of sensory terminations in the skin. PloS one 14, e0216527 (2019) https://doi.org/10.1371/journal.pone.0216527
C.P. Liao, S. Pradhan, Z. Chen, A.J. Patel, R.C. Booker, L.Q. Le, The role of nerve microenvironment for neurofibroma development. Oncotarget 7, 61500–61508 (2016). https://doi.org/10.18632/oncotarget.11133
J. Korfhage, D.B. Lombard, Malignant peripheral nerve sheath tumors: From epigenome to bedside. Mol. Cancer Res. 17, 1417–1428 (2019). https://doi.org/10.1158/1541-7786.Mcr-19-0147
A.W. James, E. Shurell, A. Singh, S.M. Dry, F.C. Eilber, Malignant Peripheral Nerve Sheath Tumor. Surg. Oncol. Clin. N. Am. 25, 789–802 (2016). https://doi.org/10.1016/j.soc.2016.05.009
S.L. Carroll, M.S. Stonecypher, Tumor suppressor mutations and growth factor signaling in the pathogenesis of NF1-associated peripheral nerve sheath tumors. I. The role of tumor suppressor mutations. J. Neuropathol. Exp. Neurol. 63, 1115–1123 (2004). https://doi.org/10.1093/jnen/63.11.1115
M. Rahmatullah, A. Schroering, K. Rothblum, R.C. Stahl, B. Urban, D.J. Carey, Synergistic regulation of Schwann cell proliferation by heregulin and forskolin, Molecular and cellular biology 18, 6245–6252 (1998) https://doi.org/10.1128/mcb.18.11.6245
S.L. Carroll, M.L. Miller, P.W. Frohnert, S.S. Kim, J.A. Corbett, Expression of neuregulins and their putative receptors, ErbB2 and ErbB3, is induced during Wallerian degeneration. J. neuroscience: official J. Soc. Neurosci. 17, 1642–1659 (1997). https://doi.org/10.1523/jneurosci.17-05-01642.1997
R.P. Huijbregts, K.A. Roth, R.E. Schmidt, S.L. Carroll, Hypertrophic neuropathies and malignant peripheral nerve sheath tumors in transgenic mice overexpressing glial growth factor beta3 in myelinating Schwann cells. J. neuroscience: official J. Soc. Neurosci. 23, 7269–7280 (2003). https://doi.org/10.1523/jneurosci.23-19-07269.2003
M.S. Stonecypher, S.J. Byer, W.E. Grizzle, S.L. Carroll, Activation of the neuregulin-1/ErbB signaling pathway promotes the proliferation of neoplastic Schwann cells in human malignant peripheral nerve sheath tumors. Oncogene 24, 5589–5605 (2005). https://doi.org/10.1038/sj.onc.1208730
J.M. Eckert, S.J. Byer, B.J. Clodfelder-Miller, S.L. Carroll, Neuregulin-1 beta and neuregulin-1 alpha differentially affect the migration and invasion of malignant peripheral nerve sheath tumor cells. Glia 57, 1501–1520 (2009). https://doi.org/10.1002/glia.20866
S.J. Kazmi, S.J. Byer, J.M. Eckert, A.N. Turk, R.P. Huijbregts, N.M. Brossier, W.E. Grizzle, F.M. Mikhail, K.A. Roth, S.L. Carroll, Transgenic mice overexpressing neuregulin-1 model neurofibroma-malignant peripheral nerve sheath tumor progression and implicate specific chromosomal copy number variations in tumorigenesis. Am. J. Pathol. 182, 646–667 (2013). https://doi.org/10.1016/j.ajpath.2012.11.017
S.N. Brosius, A.N. Turk, S.J. Byer, N.M. Brossier, L. Kohli, A. Whitmire, F.M. Mikhail, K.A. Roth, S.L. Carroll, Neuregulin-1 overexpression and Trp53 haploinsufficiency cooperatively promote de novo malignant peripheral nerve sheath tumor pathogenesis. Acta Neuropathol. 127, 573–591 (2014). https://doi.org/10.1007/s00401-013-1209-3
K.L. Carraway 3rd and L.C. Cantley, A neu acquaintance for erbB3 and erbB4: a role for receptor heterodimerization in growth signaling. Cell 78, 5–8 (1994). https://doi.org/10.1016/0092-8674(94)90564-9
J.F. Longo, S.N. Brosius, L. Black, S.H. Worley, R.C. Wilson, K.A. Roth, S.L. Carroll, ErbB4 promotes malignant peripheral nerve sheath tumor pathogenesis via Ras-independent mechanisms. Cell. communication and signaling: CCS 17, 74 (2019). https://doi.org/10.1186/s12964-019-0388-5
Z. Wang, ErbB Receptors and Cancer, (Clifton, Methods in molecular biology. N J. ) 1652, 3–35 (2017). https://doi.org/10.1007/978-1-4939-7219-7_1
R. Huang, A. Fujimura, E. Nakata, S. Takihira, H. Inoue, S. Yoshikawa, T. Hiyama, T. Ozaki, A. Kamiya, Adrenergic signaling promotes the expansion of cancer stem-like cells of malignant peripheral nerve sheath tumors. Biochem. Biophys. Res. Commun. 557, 199–205 (2021). https://doi.org/10.1016/j.bbrc.2021.03.172
S. Li, Z. Chen, L.Q. Le, New insights into the neurofibroma tumor cells of origin. Neurooncol. Adv. 2, i13-i22 (2020) https://doi.org/10.1093/noajnl/vdz044
D.Y. Lee, S.M. Gianino, D.H. Gutmann, Innate neural stem cell heterogeneity determines the patterning of glioma formation in children. Cancer cell. 22, 131–138 (2012). https://doi.org/10.1016/j.ccr.2012.05.036
F.C. Yang, D.A. Ingram, S. Chen, Y. Zhu, J. Yuan, X. Li, X. Yang, S. Knowles, W. Horn, Y. Li, S. Zhang, Y. Yang, S.T. Vakili, M. Yu, D. Burns, K. Robertson, G. Hutchins, L.F. Parada, D.W. Clapp, Nf1-dependent tumors require a microenvironment containing Nf1+/-- and c-kit-dependent bone marrow. Cell 135, 437–448 (2008). https://doi.org/10.1016/j.cell.2008.08.041
A. Bui, C. Jiang, R.M. McKay, L.J. Klesse, L.Q. Le, Insights into the Pathogenesis of NF1-Associated Neoplasms, JID innovations: skin science from molecules to population health 1, (2021) https://doi.org/10.1016/j.xjidi.2021.100044
L.B. King, T. Boto, V. Botero, A.M. Aviles, B.M. Jomsky, C. Joseph, J.A. Walker, S.M. Tomchik, Developmental loss of neurofibromin across distributed neuronal circuits drives excessive grooming in Drosophila. PLoS Genet. 16, e1008920 (2020). https://doi.org/10.1371/journal.pgen.1008920
V. Pinna, V. Lanari, P. Daniele, F. Consoli, E. Agolini, K. Margiotti, I. Bottillo, I. Torrente, A. Bruselles, C. Fusilli, A. Ficcadenti, S. Bargiacchi, E. Trevisson, M. Forzan, S. Giustini, C. Leoni, G. Zampino, M.C. Digilio, B. Dallapiccola, M. Clementi, M. Tartaglia, A. De Luca, p.Arg1809Cys substitution in neurofibromin is associated with a distinctive NF1 phenotype without neurofibromas. Eur. J. Hum. genetics: EJHG 23, 1068–1071 (2015). https://doi.org/10.1038/ejhg.2014.243
M. Upadhyaya, S.M. Huson, M. Davies, N. Thomas, N. Chuzhanova, S. Giovannini, D.G. Evans, E. Howard, B. Kerr, S. Griffiths, C. Consoli, L. Side, D. Adams, M. Pierpont, R. Hachen, A. Barnicoat, H. Li, P. Wallace, J.P. Van Biervliet, D. Stevenson, D. Viskochil, D. Baralle, E. Haan, V. Riccardi, P. Turnpenny, C. Lazaro, L. Messiaen, An absence of cutaneous neurofibromas associated with a 3-bp inframe deletion in exon 17 of the NF1 gene (c.2970–2972 delAAT): evidence of a clinically significant NF1 genotype-phenotype correlation. Am. J. Hum. Genet. 80, 140–151 (2007). https://doi.org/10.1086/510781
C. Ejerskov, M. Gaustadnes, J.R. Ostergaard, K. Krogh, K. Thorsen, A.D. Borglum, A. Haagerup, Exploring associations between constipation, severity of neurofibromatosis type 1 and NF1 mutational spectrum. Sci. Rep. 11, 9179 (2021). https://doi.org/10.1038/s41598-021-87686-x
M. Scala, I. Schiavetti, F. Madia, C. Chelleri, G. Piccolo, A. Accogli, A. Riva, V. Salpietro, R. Bocciardi, G. Morcaldi, M. Di Duca, F. Caroli, A. Verrico, C. Milanaccio, G. Viglizzo, M. Traverso, S. Baldassari, P. Scudieri, M. Iacomino, G. Piatelli, C. Minetti, P. Striano, M.L. Garrè, P. De Marco, M.C. Diana, V. Capra, M. Pavanello, F. Zara, Genotype-Phenotype Correlations in Neurofibromatosis Type 1: A Single-Center Cohort Study. Cancers 13 (2021) https://doi.org/10.3390/cancers13081879
H. Kehrer-Sawatzki, D.N. Cooper, Classification of NF1 microdeletions and its importance for establishing genotype/phenotype correlations in patients with NF1 microdeletions. Hum. Genet. 140, 1635–1649 (2021). https://doi.org/10.1007/s00439-021-02363-3
T. Johung, M. Monje, Neuronal activity in the glioma microenvironment. Curr. Opin. Neurobiol. 47, 156–161 (2017). https://doi.org/10.1016/j.conb.2017.10.009
D.P. Radin, S.E. Tsirka, Interactions between Tumor Cells, Neurons, and Microglia in the Glioma Microenvironment, Int. J. Mol. Sci. 21, (2020) https://doi.org/10.3390/ijms21228476
M.L. Broekman, S.L.N. Maas, E.R. Abels, T.R. Mempel, A.M. Krichevsky, X.O. Breakefield, Multidimensional communication in the microenvirons of glioblastoma, Nature reviews. Neurology 14, 482–495 (2018). https://doi.org/10.1038/s41582-018-0025-8
S. Friedman, R. Levy, W. Garrett, D. Doval, Bondarde, S.J.C. Research, Clinical Benefit of INCB7839, a Potent and Selective Inhibitor of ADAM10 and ADAM17, in Combination with Trastuzumab in Metastatic HER2 Positive Breast Cancer Patients. Cancer Res 69, 5056–5056 (2009)
J. Infante, H.A. Burris, N. Lewis, A multicenter phase Ib study of the safety, pharmacokinetics, biological activity and clinical efficacy of INCB7839, a potent and selective inhibitor of ADAM10 and ADAM17. (2007)
M.W. Ronellenfitsch, P.N. Harter, M. Kirchner, C. Heining, B. Hutter, L. Gieldon, J. Schittenhelm, M.U. Schuhmann, M. Tatagiba, G. Marquardt, M. Wagner, V. Endris, C.H. Brandts, V.F. Mautner, E. Schröck, W. Weichert, B. Brors, A. von Deimling, M. Mittelbronn, J.P. Steinbach, D.E. Reuss, H. Glimm, A. Stenzinger, S. Fröhling, Targetable ERBB2 mutations identified in neurofibroma/schwannoma hybrid nerve sheath tumors. J. Clin. Investig. 130, 2488–2495 (2020). https://doi.org/10.1172/jci130787
B. Weiss, B.C. Widemann, P. Wolters, E. Dombi, A. Vinks, A. Cantor, J. Perentesis, E. Schorry, N. Ullrich, D.H. Gutmann, J. Tonsgard, D. Viskochil, B. Korf, R.J. Packer, M.J. Fisher, Sirolimus for progressive neurofibromatosis type 1-associated plexiform neurofibromas: a neurofibromatosis Clinical Trials Consortium phase II study. Neuro-oncology 17, 596–603 (2015). https://doi.org/10.1093/neuonc/nou235
N.J. Ullrich, S.P. Prabhu, A.T. Reddy, M.J. Fisher, R. Packer, S. Goldman, N.J. Robison, D.H. Gutmann, D.H. Viskochil, J.C. Allen, B. Korf, A. Cantor, G. Cutter, C. Thomas, J.P. Perentesis, T. Mizuno, A.A. Vinks, P.E. Manley, S.N. Chi, M.W. Kieran, A phase II study of continuous oral mTOR inhibitor everolimus for recurrent, radiographic-progressive neurofibromatosis type 1-associated pediatric low-grade glioma: a Neurofibromatosis Clinical Trials Consortium study. Neuro-oncology 22, 1527–1535 (2020). https://doi.org/10.1093/neuonc/noaa071
Acknowledgements
Figures 1 and 2 were designed using BioRender.com web resource.
Funding
This work was supported by grants from National Natural Science Foundation of China (82102344; 82172228); Shanghai Rising Star Program supported by Science and Technology Commission of Shanghai Municipality (20QA1405600); Science and Technology Commission of Shanghai Mu-nicipality (19JC1413); Natural Science Foundation of Shanghai (22ZR1422300); “Chenguang Pro-gram” supported by Shanghai Education Development Foundation (SHEDF) (19CG18); Shanghai Municipal Key Clinical Specialty (shslczdzk00901); Innovative research team of high-level local universities in Shanghai (SSMU-ZDCX20180700).
Author information
Authors and Affiliations
Contributions
Conceptualization, ZC W, QF L, and HB Z; methodology, ZC W, QF L, and HB Z; investigation, LL G and MY X; resources, LL G and MY X; data curation, LL G and MY X; writing—original draft preparation, LL G; writing—review and editing, MY X, ZC W, and HB Z; visualization, LL G; supervision, ZC W, QF L, and HB Z; project administration, ZC W, QF L, and HB Z; funding acquisition, ZC W. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethical approval and Consent to participate
Not applicable.
Human Ethics
Not applicable.
Consent for publication
Not applicable.
Availability of supporting data
Not applicable.
Competing interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
# These authors contributed equally to this work.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ge, LL., Xing, MY., Zhang, HB. et al. Role of nerves in neurofibromatosis type 1-related nervous system tumors. Cell Oncol. 45, 1137–1153 (2022). https://doi.org/10.1007/s13402-022-00723-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-022-00723-3