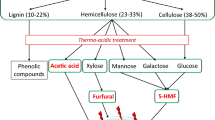
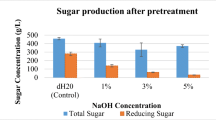
Abstract
Lignocellulosic biomass as a cheap and abundant raw material offers great advantages in terms of biotechnological applications. The current study aimed to produce bioethanol from mulberry pomace by newly isolated xylose/glucose co-fermenter Hanseniaspora uvarum. For this purpose, the xylose consumption capacity of the yeast was first tested using the synthetic xylose-containing medium as a carbon source, and Hanseniaspora uvarum effectively fermented xylose into ethanol. Afterward, Hansenispora uvarum was used for the fermentation of the liquid hydrolysate obtained by dilute acid pretreatment of mulberry pomace. Furthermore, a subsequent enzymatic hydrolysis of the pretreated mulberry pomace was performed to investigate its effect. For this purpose, initial biomass concentration (50–500 g/L) for fermentation was optimized in two different mulberry pomace media containing nitrogen sources or mineral salts. Enzymatic hydrolysis of pretreated mulberry pomace was performed at 400 g/L initial biomass loading with or without soluble soy protein addition, and a significant increase in fermentable sugar concentrations was observed. The highest sugar concentration was observed as 159.6 g/L when 400 g/L biomass loading was used during dilute acid pretreatment, and subsequent enzymatic hydrolysis was performed using 15 FPU/g cellulose. Moreover, after dilute acid pretreatment and enzymatic hydrolysis, ethanol production of Hanseniaspora uvarum reached 61.3 g/L (0.153 g/gbiomass) which is a sufficient amount for efficient distillation according to literature. Results show that xylose/glucose co-fermenter Hanseniaspora uvarum and mulberry pomace can be considered for second-generation bioethanol production.




Similar content being viewed by others
Data Availability
Not applicable.
References
Lugani Y, Rai R, Prabhu AA et al (2020) Recent advances in bioethanol production from lignocelluloses: a comprehensive review with a focus on enzyme engineering and designer biocatalysts. Biofuel Res J 7:1267–1295. https://doi.org/10.18331/BRJ2020.7.4.5
Safaripour M, Ghanbari A, Seyedabadi E, Pourhashem G (2021) Investigation of environmental impacts of bioethanol production from wheat straw in Kermanshah, Iran. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-021-01676-7
Jambo SA, Abdulla R, Mohd Azhar SH et al (2016) A review on third generation bioethanol feedstock. Renew Sustain Energy Rev 65:756–769. https://doi.org/10.1016/j.rser.2016.07.064
Lennartsson PR, Erlandsson P, Taherzadeh MJ (2014) Integration of the first and second generation bioethanol processes and the importance of by-products. Bioresour Technol 165:3–8. https://doi.org/10.1016/j.biortech.2014.01.127
Christiansen K, Raman DR, Hu G, Anex R (2018) First-order estimates of the costs, input-output energy analysis, and energy returns on investment of conventional and emerging biofuels feedstocks. Biofuel Res J 5:894–899. https://doi.org/10.18331/brj2018.5.4.4
Yu HT, Chen BY, Li BY et al (2018) Efficient pretreatment of lignocellulosic biomass with high recovery of solid lignin and fermentable sugars using Fenton reaction in a mixed solvent. Biotechnol Biofuels 11:1–11. https://doi.org/10.1186/s13068-018-1288-4
Haven MØ, Jørgensen H (2013) Adsorption of β-glucosidases in two commercial preparations onto pretreated biomass and lignin. Biotechnol Biofuels 6:1–14. https://doi.org/10.1186/1754-6834-6-165
Bhagia S, Dhir R, Kumar R, Wyman CE (2018) Deactivation of cellulase at the air-liquid ınterface ıs the main cause of ıncomplete cellulose conversion at low enzyme loadings. Sci Rep 8:1–12. https://doi.org/10.1038/s41598-018-19848-3
Luo X, Liu J, Zheng P et al (2019) Promoting enzymatic hydrolysis of lignocellulosic biomass by inexpensive soy protein. Biotechnol Biofuels 12:1–13. https://doi.org/10.1186/s13068-019-1387-x
Walker GM, Basso TO (2020) Mitigating stress in industrial yeasts. Fungal Biol 124:387–397. https://doi.org/10.1016/j.funbio.2019.10.010
Tse TJ, Wiens DJ (2021) Production of bioethanol—a review of factors affecting ethanol yield. Fermantation 7:1–18. https://doi.org/10.3390/fermentation7040268
Koppram R, Tomás-Pejó E, Xiros C, Olsson L (2014) Lignocellulosic ethanol production at high-gravity: challenges and perspectives. Trends Biotechnol 32:46–53. https://doi.org/10.1016/j.tibtech.2013.10.003
Ye G, Zeng D, Zhang S et al (2018) Ethanol production from mixtures of sugarcane bagasse and Dioscorea composita extracted residue with high solid loading. Bioresour Technol 257:23–29. https://doi.org/10.1016/j.biortech.2018.02.008
Imran M, Khan H, Shah M et al (2010) Chemical composition and antioxidant activity of certain Morus species. J Zhejiang Univ Sci B 11:973–980. https://doi.org/10.1631/jzus.B1000173
Minhas MA, Begum A, Hamid S et al (2016) Evaluation of antibiotic and antioxidant activity of morus nigra (black mulberry) extracts against soil borne, food borne and clinical human pathogens. Pak J Zool 48:1381–1388
Yuan Q, Zhao L (2017) The Mulberry (Morus alba L.) Fruit – a review of characteristic components and health benefits. J Agric Food Chem 65:10383–10394. https://doi.org/10.1021/acs.jafc.7b03614
Aidynova R, Arslan N, Aydoğan MN (2020) Use of mulberry pomace as substrate for cıtrıc acıd productıon by Aspergillus niger MT-4. Trak Univ J Nat Sci 21:159–165. https://doi.org/10.23902/trkjnat.670859
Baeyens J, Kang Q, Appels L et al (2015) Challenges and opportunities in improving the production of bio-ethanol. Prog Energy Combust Sci 47:60–88. https://doi.org/10.1016/j.pecs.2014.10.003
Goshima T, Tsuji M, Inoue H et al (2013) Bioethanol production from lignocellulosic biomass by a novel Kluyveromyces marxianus strain. Biosci Biotechnol Biochem 77:1505–1510. https://doi.org/10.1271/bbb.130173
Zaky AS, Greetham D, Tucker GA, Du C (2018) The establishment of a marine focused biorefinery for bioethanol production using seawater and a novel marine yeast strain. Sci Rep 8:1–14. https://doi.org/10.1038/s41598-018-30660-x
Ben Atitallah I, Ntaikou I, Antonopoulou G et al (2020) Evaluation of the non-conventional yeast strain Wickerhamomyces anomalus (Pichia anomala) X19 for enhanced bioethanol production using date palm sap as renewable feedstock. Renew Energy 154:71–81. https://doi.org/10.1016/j.renene.2020.03.010
Pramateftaki PV, Kouvelis VN, Lanaridis P, Typas MA (2006) The mitochondrial genome of the wine yeast Hanseniaspora uvarum: a unique genome organization among yeast/fungal counterparts. FEMS Yeast Res 6:77–90. https://doi.org/10.1111/j.1567-1364.2005.00018.x
Pietrafesa A, Capece A, Pietrafesa R et al (2020) Saccharomyces cerevisiae and Hanseniaspora uvarum mixed starter cultures: ınfluence of microbial/physical interactions on wine characteristics. Yeast 37:609–621. https://doi.org/10.1002/yea.3506
Matraxia M, Alfonzo A, Prestianni R et al (2021) Non-conventional yeasts from fermented honey by-products: focus on Hanseniaspora uvarum strains for craft beer production. Food Microbiol 99:103806. https://doi.org/10.1016/j.fm.2021.103806
Barros EM, Rodrigues THS, Pinheiro ADT et al (2014) A yeast ısolated from cashew apple juice and ıts ability to produce first- and second-generation ethanol. Appl Biochem Biotechnol 174:2762–2776. https://doi.org/10.1007/s12010-014-1224-4
Hong YA, Park HD (2013) Role of non-Saccharomyces yeasts in Korean wines produced from Campbell Early grapes: potential use of Hanseniaspora uvarum as a starter culture. Food Microbiol 34:207–214. https://doi.org/10.1016/j.fm.2012.12.011
Glass NL, Donaldson GC (1995) Development of primer sets designed for use with the PCR to amplify conserved genes from filamentous ascomycetes. Appl Environ Microbiol 61:1323–1330. https://doi.org/10.1128/aem.61.4.1323-1330.1995
Wistara NJ, Pelawi R, Fatriasari W (2016) The effect of lignin content and freeness of pulp on the bioethanol productivity of Jabon wood. Waste and Biomass Valorization 7:1141–1146. https://doi.org/10.1007/s12649-016-9510-8
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31:426–428. https://doi.org/10.1021/ac60147a030
Aksu Z, Dönmez G (2000) The use of molasses in copper(II) containing wastewaters: effects on growth and copper(II) bioaccumulation properties of Kluyveromyces marxianus. Process Biochem 36:451–458. https://doi.org/10.1016/S0032-9592(00)00234-X
Kim TH, Lee YY (2007) Pretreatment of corn stover by soaking in aqueous ammonia at moderate temperatures. Appl Biochem Biotechnol 136:81–82
Roca C, Olsson L (2003) Increasing ethanol productivity during xylose fermentation by cell recycling of recombinant Saccharomyces cerevisiae. Appl Microbiol Biotechnol 60:560–563. https://doi.org/10.1007/s00253-002-1147-9
Günan Yücel H, Aksu Z (2015) Ethanol fermentation characteristics of Pichia stipitis yeast from sugar beet pulp hydrolysate: use of new detoxification methods. Fuel 158:793–799. https://doi.org/10.1016/j.fuel.2015.06.016
Petravić-Tominac V, Zechner-Krpan V, Berković K et al (2011) Rheological properties, water-holding and oil-binding capacities of particulate β-glucans isolated from spent Brewer’s yeast by three different procedures. Food Technol Biotechnol 49:56–64
Jagtap SS, Rao CV (2018) Microbial conversion of xylose into useful bioproducts. Appl Microbiol Biotechnol 102:9015–9036. https://doi.org/10.1007/s00253-018-9294-9
Langenberg A, Bink FJ, Wolff L et al (2017) Glycolytic functions are conserved in the genome of the wine yeast. Appl Environ Microbiol 83:1–20
Thomson JM, Gaucher EA, Burgan MF et al (2005) Resurrecting ancestral alcohol dehydrogenases from yeast. Nat Genet 37:630–635. https://doi.org/10.1038/ng1553
Raamsdonk LM, Diderich JA, Kuiper A et al (2002) Erratum: co-consumption of sugars or ethanol and glucose in a Saccharomyces cervisiae strain deleted in the HXK2 gene (Yeast (2001) vol. 18 (11) (1023-1033)). Yeast 19:183. https://doi.org/10.1002/yea.836
de Moura Ferreira MA, da Silveira FA, da Silveira WB (2022) Ethanol stress responses in Kluyveromyces marxianus: current knowledge and perspectives. Appl Microbiol Biotechnol 106:1341–1353. https://doi.org/10.1007/s00253-022-11799-0
Sanchez A, Hernández-Sánchez P, Puente R (2019) Hydration of lignocellulosic biomass. Modelling and experimental validation. Ind Crop Prod 131:70–77. https://doi.org/10.1016/j.indcrop.2019.01.029
Phuttaro C, Sawatdeenarunat C, Surendra KC et al (2019) Anaerobic digestion of hydrothermally-pretreated lignocellulosic biomass: ınfluence of pretreatment temperatures, inhibitors and soluble organics on methane yield. Bioresour Technol 284:128–138. https://doi.org/10.1016/j.biortech.2019.03.114
López-Linares JC, Romero I, Cara C et al (2014) Bioethanol production from rapeseed straw at high solids loading with different process configurations. Fuel 122:112–118. https://doi.org/10.1016/j.fuel.2014.01.024
Sindhu R, Binod P, Pandey A (2016) A novel sono-assisted acid pretreatment of chili post harvest residue for bioethanol production. Bioresour Technol 213:58–63. https://doi.org/10.1016/j.biortech.2016.02.079
Germec M, Demirel F, Tas N et al (2017) Microwave-assisted dilute acid pretreatment of different agricultural bioresources for fermentable sugar production. Cellulose 24:4337–4353. https://doi.org/10.1007/s10570-017-1408-5
Mohd Zakria R, Gimbun J, Asras MFF, Chua GK (2017) Magnesium sulphate and Β-alanine enhanced the ability of Kluyveromyces marxianus producing bioethanol using oil palm trunk sap. Biofuels 8:595–603. https://doi.org/10.1080/17597269.2016.1242690
Slininger PJ, Dien BS, Gorsich SW, Liu ZL (2006) Nitrogen source and mineral optimization enhance D-xylose conversion to ethanol by the yeast Pichia stipitis NRRL Y-7124. Appl Microbiol Biotechnol 72:1285–1296. https://doi.org/10.1007/s00253-006-0435-1
Hu K, Jin GJ, Xu YH et al (2019) Enhancing wine ester biosynthesis in mixed Hanseniaspora uvarum/Saccharomyces cerevisiae fermentation by nitrogen nutrient addition. Food Res Int 123:559–566. https://doi.org/10.1016/j.foodres.2019.05.030
Liu R, Li J, Shen F (2008) Refining bioethanol from stalk juice of sweet sorghum by immobilized yeast fermentation. Renew Energy 33:1130–1135. https://doi.org/10.1016/j.renene.2007.05.046
Shahirah MNN, Gimbun J, Pang SF et al (2015) Influence of nutrient addition on the bioethanol yield from oil palm trunk sap fermented by Saccharomyces cerevisiae. J Ind Eng Chem 23:213–217. https://doi.org/10.1016/j.jiec.2014.08.018
Birch RM, Walker GM (2000) Influence of magnesium ions on heat shock and ethanol stress responses of Saccharomyces cerevisiae. Enzyme Microb Technol 26:678–687. https://doi.org/10.1016/S0141-0229(00)00159-9
Demiray E, Kut A, Ertuğrul Karatay S, Dönmez G (2021) Usage of soluble soy protein on enzymatically hydrolysis of apple pomace for cost-efficient bioethanol production. Fuel 289:1–9. https://doi.org/10.1016/j.fuel.2020.119785
Lee I, Yu JH (2020) The production of fermentable sugar and bioethanol from acacia wood by optimizing dilute sulfuric acid pretreatment and post treatment. Fuel 275:117943. https://doi.org/10.1016/j.fuel.2020.117943
Funding
This study was supported by the Ankara University Research Foundation. Project Number: 21L0430012.
Author information
Authors and Affiliations
Contributions
Hüseyin Kaan Kabadayı conducted the experiments. Ekin Demiray conducted the experiments and wrote the original draft. Sevgi Ertuğrul Karatay conceived and designed research, reviewed and edited the manuscript, and provided funding. Gönül Dönmez provided funding.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kabadayı, H.K., Demiray, E., Karatay, S.E. et al. Bioethanol production from mulberry pomace by newly ısolated non-conventional yeast Hanseniaspora uvarum. Biomass Conv. Bioref. 14, 10611–10620 (2024). https://doi.org/10.1007/s13399-023-04340-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-023-04340-4