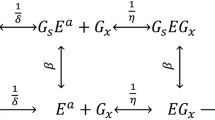Abstract
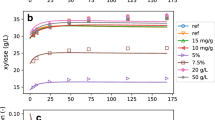
Though a large number of simple to complex models are theorized in the kinetic description of cellulose hydrolysis, the reason for their selection in specific studies remains obscure. Considering different combinations of substrate biodegradability-accessibility, hydrolysis step, and the fate of enzyme (adsorption/deactivation) in a hierarchical approach, 36 dynamic model structures of different complexity were tested in the present study for microcrystalline cellulose hydrolysis at different substrate/enzyme loadings. The quality of the candidate models was assessed based on R2, adjusted R2, Aike information criteria (AIC), and statistical analysis. The results suggest that consideration of substrate accessibility can influence model fitness only when the substrate is biodegradable. However, the selection of hydrolysis kinetics and enzyme fate is remained as significant. A simple 4-parameter model assuming complete biodegradable-accessible substrate, homogeneous Michaelis–Menten (MM), and enzyme deactivation is selected (R2 = 0.946, adj. R2 = 0.920), even though a few complex models offered marginally better R2. Further, dynamic relative sensitivity analysis revealed that reducing sugar profile was sensitive to the entire set of parameters. Though the complex models reveal the cellulose hydrolysis dynamics on a greater extent, such an attempt should be performed with caution unless experimental data on other process variables (apart from reducing sugar) are made available in regression. Estimation of parameters relevant to enzyme inactivation or biomass adsorption from independent batch experiments can be invaluable in deciphering complex hydrolysis kinetics.



Similar content being viewed by others
Data availability
Experimental data in this study are available on request to the corresponding author.
References
Batista Meneses D, Montes de Oca-Vásquez G, Vega-Baudrit JR et al (2022) Pretreatment methods of lignocellulosic wastes into value-added products: recent advances and possibilities. Biomass Convers Biorefinery 12:547–564. https://doi.org/10.1007/s13399-020-00722-0
Nill J, Karuna N, Jeoh T (2018) The impact of kinetic parameters on cellulose hydrolysis rates. Process Biochem 74:108–117. https://doi.org/10.1016/j.procbio.2018.07.006
Liang C, Gu C, Raftery J et al (2019) Development of modified HCH-1 kinetic model for long-term enzymatic cellulose hydrolysis and comparison with literature models. Biotechnol Biofuels 12:34. https://doi.org/10.1186/s13068-019-1371-5
Ciesielski PN, Pecha MB, Lattanzi AM et al (2020) Advances in multiscale modeling of lignocellulosic biomass. ACS Sustain Chem Eng 8:3512–3531. https://doi.org/10.1021/acssuschemeng.9b07415
Jeoh T, Cardona MJ, Karuna N et al (2017) Mechanistic kinetic models of enzymatic cellulose hydrolysis—a review. Biotechnol Bioeng 114:1369–1385. https://doi.org/10.1002/bit.26277
Gan Q, Allen S, Taylor G (2003) Kinetic dynamics in heterogeneous enzymatic hydrolysis of cellulose: an overview, an experimental study and mathematical modelling. Process Biochem 38:1003–1018. https://doi.org/10.1016/S0032-9592(02)00220-0
Pratto B, de Souza RBA, Sousa R, da Cruz AJG (2016) Enzymatic hydrolysis of pretreated sugarcane straw: kinetic study and semi-mechanistic modeling. Appl Biochem Biotechnol 178:1430–1444. https://doi.org/10.1007/s12010-015-1957-8
Chan K, Ko C, Chang K, Leu S (2021) Construction of a structural enzyme adsorption/kinetics model to elucidate additives associated lignin–cellulase interactions in complex bioconversion system. Biotechnol Bioeng 118:4065–4075. https://doi.org/10.1002/bit.27883
Sin G, Meyer AS, Gernaey KV (2010) Assessing reliability of cellulose hydrolysis models to support biofuel process design—identifiability and uncertainty analysis. Comput Chem Eng 34:1385–1392. https://doi.org/10.1016/j.compchemeng.2010.02.012
Sousa R Jr, Carvalho ML, Giordano RLC, Giordano RC (2011) Recent trends in the modeling of cellulose hydrolysis. Braz J Chem Eng 28:545–564. https://doi.org/10.1590/S0104-66322011000400001
Haldar D, Sen D, Gayen K (2018) Enzymatic hydrolysis of banana stems (Musa acuminata): optimization of process parameters and inhibition characterization. Int J Green Energy 15:406–413. https://doi.org/10.1080/15435075.2018.1467834
Jain A, Jain R, Jain S (2020) Quantitative analysis of reducing sugars by 3, 5-dinitrosalicylic acid (DNSA method). In: Basic Techniques in Biochemistry, Microbiology and Molecular Biology. Humana, New York, pp 181–183
Lebaz N, Cockx A, Spérandio M, Morchain J (2015) Population balance approach for the modelling of enzymatic hydrolysis of cellulose. Can J Chem Eng 93:276–284. https://doi.org/10.1002/cjce.22088
Myung JI, Pitt MA (2004) Model comparison methods. Methods Enzymol 383:351–366. https://doi.org/10.1016/S0076-6879(04)83014-3
Akaike H (1974) A new look at the statistical model identification. IEEE Trans Automat Contr 19:716–723. https://doi.org/10.1109/TAC.1974.1100705
Fieberg JR, Vitense K, Johnson DH (2020) Resampling-based methods for biologists. PeerJ 8:e9089. https://doi.org/10.7717/peerj.9089
Anane E, López CDC, Barz T et al (2019) Output uncertainty of dynamic growth models: effect of uncertain parameter estimates on model reliability. Biochem Eng J 150:107247. https://doi.org/10.1016/j.bej.2019.107247
Carvalho ML, Sousa R Jr, Rodríguez-Zúñiga UF et al (2013) Kinetic study of the enzymatic hydrolysis of sugarcane bagasse. Braz J Chem Eng 30:437–447. https://doi.org/10.1590/S0104-66322013000300002
Kumar D, Murthy GS (2013) Stochastic molecular model of enzymatic hydrolysis of cellulose for ethanol production. Biotechnol Biofuels 6:63. https://doi.org/10.1186/1754-6834-6-63
Tervasmäki P, Sotaniemi V, Kangas J et al (2017) A discretized model for enzymatic hydrolysis of cellulose in a fed-batch process. Bioresour Technol 227:112–124. https://doi.org/10.1016/j.biortech.2016.12.054
Yeh A-I, Huang Y-C, Chen SH (2010) Effect of particle size on the rate of enzymatic hydrolysis of cellulose. Carbohydr Polym 79:192–199. https://doi.org/10.1016/j.carbpol.2009.07.049
Kristensen JB, Felby C, Jørgensen H (2009) Yield-determining factors in high-solids enzymatic hydrolysis of lignocellulose. Biotechnol Biofuels 2:11. https://doi.org/10.1186/1754-6834-2-11
Zhang Y, Zhang Y, Song M et al (2021) Combining Michaelis-Menten theory and enzyme deactivation reactions for the kinetic study of enzymatic hydrolysis by different pretreated sugarcane bagasse. Process Biochem 105:72–78. https://doi.org/10.1016/j.procbio.2021.03.023
Zhang Y, Xu J-L, Xu H-J et al (2010) Cellulase deactivation based kinetic modeling of enzymatic hydrolysis of steam-exploded wheat straw. Bioresour Technol 101:8261–8266. https://doi.org/10.1016/j.biortech.2010.06.015
Ye Z, Hatfield KM, Berson RE (2014) Relative extents of activity loss between enzyme–substrate interactions and combined environmental mechanisms. Bioresour Technol 164:143–148. https://doi.org/10.1016/j.biortech.2014.04.080
Liang L, Xue D (2017) Kinetics of cellulose hydrolysis by halostable cellulase from a marine Aspergillus niger at different salinities. Process Biochem 63:163–168. https://doi.org/10.1016/j.procbio.2017.09.004
Joshi M, Seidel-Morgenstern A, Kremling A (2006) Exploiting the bootstrap method for quantifying parameter confidence intervals in dynamical systems. Metab Eng 8:447–455. https://doi.org/10.1016/j.ymben.2006.04.003
Acknowledgements
The authors DH and BM are thankful to Karunya Institute of Technology and Sciences (Deemed to be University), Coimbatore, India, for providing support for this study and to develop knowledge towards subsequent research works.
Author information
Authors and Affiliations
Contributions
The idea for the article was conceived by DH. First draft preparation and analysis was performed by BM. Both the authors critically revised the work.
Corresponding authors
Ethics declarations
Ethics approval and consent to participate
The study does not involve any human participants, human data, or human tissue. Ethics approval is not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Haldar, D., Mahanty, B. Hierarchical model screening on enzymatic hydrolysis of microcrystalline cellulose. Biomass Conv. Bioref. 14, 8503–8512 (2024). https://doi.org/10.1007/s13399-022-02860-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-022-02860-z