Abstract
In the present study, eleven flexible models were employed to describe the effect of different medium compositions (DMC) on ethanol fermentation in repeated-batch biofilm reactors with carob extract. Residual-sum of square, root-mean-square-error, mean-absolute-error, determination coefficient, bias factor, accuracy factor, F test, and objective function were used to compare the models. Findings indicated that corresponding with the prediction of the experimental data of substrate concentration (S), the best-selected models were the Baranyi model (media A and C), Weibull model (medium B), and re-modified Gompertz model (R-MGM) (medium D). It was also found that in the estimation of the observed biomass concentration (X) data, Baranyi model (medium A), Weibull model (medium B), and Stannard model (media C and D) gave well-directed results according to the model comparison, validation, and fitting results. As related to ethanol concentration (P), the predicted data with the re-modified Richards model (R-MRM) (media A and B), re-modified logistic model (R-MLM) (medium C), and Baranyi model (medium D) were showed good agreement with the experimental p values. To validate the best-selected models, an independent set of the experimental data for each medium was used and it was found that the independent experimental values were highly compatible with the selected models. Consequently, the best-selected models can serve as universal equations to fit satisfactorily the experimental S, X, and P curves. These models can also be used for further improvement of the carob extract–based bioethanol production process.

Similar content being viewed by others
Abbreviations
- μ :
-
1/h; Specific growth rate
- °Bx:
-
-; Brix
- AF:
-
-; Accuracy factor
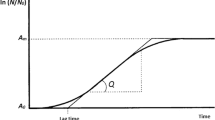
- A m :
-
g/L; Upper asymptote
- A o :
-
g/L; Lower asymptote
- A t :
-
g/L; The predicted biomass, ethanol, and sugar concentration at time t
- BF:
-
-; Bias factor
- DF:
-
-; Degree of freedom
- d :
-
-; Dimensionless design parameter
- DMC:
-
-; Different medium compositions
- e :
-
-; Euler number, 2.718
- h o :
-
g/LA; parameter calculating the initial physiology state of the cells
- k :
-
-; A parameter governing the rate at which the response variable approaches its potential maximum
- MAE:
-
g/L; Mean-absolute-error
- MMF:
-
-; Morgan-Mercer-Flodin model
- n :
-
-; Number of observations
- P :
-
g/L; Ethanol concentration
- ΔP :
-
g/L; Ethanol produced
- PCS:
-
-; Plastic composite support
- P max :
-
g/L; Maximum ethanol concentration
- Q :
-
g/L/h; Maximum ethanol production, sugar consumption, and biomass production rate
- Q P :
-
g/L/h; Maximum ethanol production rate
- Q S :
-
g/L/h; Maximum sugar consumption rate
- Q X :
-
g/L/h; Maximum biomass production rate
- R 2 :
-
-; Determination coefficient
- R-MGM:
-
-; Re-modified Gompertz model
- R-MLM:
-
-; Re-modified logistic model
- R-MRM:
-
-; Re-modified Richards model
- RMSE:
-
g/L; Root-mean-square-error
- RSS:
-
g/L; Residual sum of squares
- S :
-
g/L; Substrate concentration
- ΔS :
-
g/L; Sugar consumed
- S max :
-
g/L; Maximum sugar concentration
- SUY:
-
%; Sugar utilization yield
- t :
-
h; Sampling time
- t d :
-
h; Doubling time
- T L :
-
h; The point where At is equal to Am/2
- v :
-
-; Dimensionless shape parameter
- X :
-
g/L; Biomass concentration
- ΔX :
-
g/L; Biomass produced
- X max :
-
g/L; Maximum biomass production
- x i :
-
g/L; Experimental value at timet
- Y P/S :
-
%; Ethanol yield
- y i :
-
g/L; Predicted value at time t
- a :
-
-; Lag phase transition coefficient
- β :
-
-; Growth displacement
- δ :
-
-; Allometric constant
- λ :
-
h; Lag time
- Φ value:
-
-; Objective function
References
Machineni L (2019) Lignocellulosic biofuel production: review of alternatives. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-019-00445-x
Germec M, Turhan I, Karhan M, Demirci A (2015) Ethanol production via repeated-batch fermentation from carob pod extract by using Saccharomyces cerevisiae in biofilm reactor. Fuel 161:304–311
Abbas A, Ansumali S (2010) Global potential of rice husk as a renewable feedstock for ethanol biofuel production. BioEnergy Research 3(4):328–334. https://doi.org/10.1007/s12155-010-9088-0
Germec M, Kartal FK, Bilgic M, Ilgin M, Ilhan E, Güldali H, Isci A, Turhan I (2016) Ethanol production from rice hull using Pichia stipitis and optimization of acid pretreatment and detoxification processes. Biotechnol Prog 32(4):872–882
Germec M, Turhan I (2018) Ethanol production from acid-pretreated and detoxified tea processing waste and its modeling. Fuel 231:101–109
Germec M, Turhan I, Yatmaz E, Tetik N, Karhan M (2016) Fermentation of acid-pretreated tea processing waste for ethanol production using Saccharomyces cerevisiae. Sci Bull Series F Biotechnol 20:269–274
Germec M, Ozcan A, Yilmazer C, Tas N, Onuk Z, Demirel F, Turhan I (2017) Ethanol fermentation from microwave-assisted acid pretreated raw materials by Scheffersomyces stipitis. AgroLife Sci J 6(1):112–118
Germec M, Turhan I (2018) Ethanol production from acid-pretreated and detoxified rice straw as sole renewable resource. Biomass Convers Biorefinery 8(3):607–619
Medouze D, Pan CL, Cheng KC (2018) Evaluation of ethanol production from Ulva lactuca hydrolysate. Taiwan J Agr Chem Food Sci 56(3/4):69–76
Wu WH, Hung WC, Lo KY, Chen YH, Wan HP, Cheng KC (2016) Bioethanol production from taro waste using thermo-tolerant yeast Kluyveromyces marxianus K21. Bioresour Technol 201:27–32
Cardona CA, Sánchez ÓJ (2007) Fuel ethanol production: process design trends and integration opportunities. Bioresour Technol 98(12):2415–2457
Sarris D, Papanikolaou S (2016) Biotechnological production of ethanol: biochemistry, processes and technologies. Eng Life Sci 16(4):307–329
Ercan Y, Irfan T, Mustafa K (2013) Optimization of ethanol production from carob pod extract using immobilized Saccharomyces cerevisiae cells in a stirred tank bioreactor. Bioresour Technol 135:365–371
Germec M, Karhan M, Demirci A, Turhan I (2018) Ethanol production in a biofilm reactor with non-sterile carob extract media and its modeling. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects 40(22):2726–2734
Germec M, Turhan I, Demirci A, Karhan M (2016) Effect of media sterilization and enrichment on ethanol production from carob extract in a biofilm reactor. Energy Sources, Part A: Recovery, Utilization, and Environmental Effects 38(21):3268–3272
Oziyci HR, Tetik N, Turhan I, Yatmaz E, Ucgun K, Akgul H, Gubbuk H, Karhan M (2014) Mineral composition of pods and seeds of wild and grafted carob (Ceratonia siliqua L.) fruits. Sci Hortic 167:149–152
Turhan I, Bialka KL, Demirci A, Karhan M (2010) Ethanol production from carob extract by using Saccharomyces cerevisiae. Bioresour Technol 101(14):5290–5296
Yatmaz E, Turhan I (2018) Carob as a carbon source for fermentation technology. Biocatalysis Agr Biotechnol 16:200–208
Ho K, Pometto A, Hinz PN, Demirci A (1997) Nutrient leaching and end product accumulation in plastic composite supports for L-(+)-lactic acid biofilm fermentation. Appl Environ Microbiol 63(7):2524–2532
Demirci A, Pometto A III, Ho KG (1997) Ethanol production by Saccharomyces cerevisiae in biofilm reactors. J Ind Microbiol Biotechnol 19(4):299–304
Izmirlioglu G, Demirci A (2016) Ethanol production in biofilm reactors from potato waste hydrolysate and optimization of growth parameters for Saccharomyces cerevisiae. Fuel 181:643–651
Izmirlioglu G, Demirci A (2017) Simultaneous saccharification and fermentation of ethanol from potato waste by co-cultures of Aspergillus niger and Saccharomyces cerevisiae in biofilm reactors. Fuel 202:260–270
Bhowmick AR, Bhattacharya S (2014) A new growth curve model for biological growth: some inferential studies on the growth of Cirrhinus mrigala. Math Biosci 254:28–41
Chou IC, Voit EO (2009) Recent developments in parameter estimation and structure identification of biochemical and genomic systems. Math Biosci 219(2):57–83205
de Jesus S, Maciel Filho R (2010) Modeling growth of microalgae Dunaliella salina under different nutritional conditions. Am J Biochem Biotechnol 6(4):279–283
Farliahati M, Mohamed M, Rosfarizan M, Tri Puspaningsih N, Ariff A (2009) Kinetics of xylanase fermentation by recombinant Escherichia coli DH5α in shake flask culture. Am J Biochem Biotechnol 5(3):110–118
Germec M, Cheng K-C, Karhan M, Demirci A, Turhan I (2019) Application of mathematical models to ethanol fermentation in biofilm reactor with carob extract. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-019-00425-1
Germec M, Karhan M, Bialka KL, Demirci A, Turhan I (2018) Mathematical modeling of lactic acid fermentation in bioreactor with carob extract. Biocatalysis Agr Biotechnol 14:254–263
Germec M, Kartal FK, Guldali H, Bilgic M, Isci A, Turhan I (2016) Obtaining growth curves for Scheffersomyces stipitis strains and their modeling. Sci Bull Series F Biotechnol 20:263–268
Sasikumar E, Viruthagiri T (2008) Optimization of process conditions using response surface methodology (RSM) for ethanol production from pretreated sugarcane bagasse: kinetics and modeling. BioEnergy Res 1:239–247
Sriyudthsak K, Shiraishi F (2010) Investigation of the performance of fermentation processes using a mathematical model including effects of metabolic bottleneck and toxic product on cells. Math Biosci 228(1):1–9
Sablani SS, Datta AK, Rahman MS, Mujumdar AS (2006) Handbook of food and bioprocess modeling techniques. CRC Press
Srivastava N, Rawat R, Singh Oberoi H, Ramteke PW (2015) A review on fuel ethanol production from lignocellulosic biomass. Int J Green Energy 12(9):949–960
Dodić JM, Vučurović DG, Dodić SN, Grahovac JA, Popov SD, Nedeljković NM (2012) Kinetic modelling of batch ethanol production from sugar beet raw juice. Appl Energy 99:192–197
Gabardo S, Pereira GF, Rech R, Ayub MAZ (2015) The modeling of ethanol production by Kluyveromyces marxianus using whey as substrate in continuous A-Stat bioreactors. J Ind Microbiol Biotechnol 42(9):1243–1253
Putra MD, Abasaeed AE, Atiyeh HK, Al-Zahrani SM, Gaily MH, Sulieman AK, Zeinelabdeen MA (2015) Kinetic modeling and enhanced production of fructose and ethanol from date fruit extract. Chem Eng Commun 202(12):1618–1627
Zwietering M, Jongenburger I, Rombouts F, Van't Riet K (1990) Modeling of the bacterial growth curve. Appl Environ Microbiol 56(6):1875–1881
Stannard C, Williams A, Gibbs P (1985) Temperature/growth relationships for psychrotrophic food-spoilage bacteria. Food Microbiol 2(2):115–122
Weibull W (1951) A statistical distribution function of wide applicability. J Appl Mech 9:293–297
Morgan PH, Mercer LP, Flodin NW (1975) General model for nutritional responses of higher organisms. Proc Natl Acad Sci 72(11):4327–4331
Dantigny P, Nanguy SP-M, Judet-Correia D, Bensoussan M (2011) A new model for germination of fungi. Int J Food Microbiol 146(2):176–181
Baranyi J, Roberts TA (1994) A dynamic approach to predicting bacterial growth in food. Int J Food Microbiol 23(3–4):277–294
Huang L (2013) Optimization of a new mathematical model for bacterial growth. Food Control 32(1):283–288
Fitzhugh H (1976) Analysis of growth curves and strategies for altering their shape. J Anim Sci 42(4):1036–1051
Cone JW, van Gelder AH, Visscher GJ, Oudshoorn L (1996) Influence of rumen fluid and substrate concentration on fermentation kinetics measured with a fully automated time related gas production apparatus. Anim Feed Sci Technol 61(1):113–128
Mahdinia E, Mamouri SJ, Puri VM, Demirci A, Berenjian A (2019) Modeling of vitamin K (Menaquinoe-7) fermentation by Bacillus subtilis natto in biofilm reactors. Biocatalysis Agr Biotechnol 17:196–202
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Chem 31(3):426–428
Chai T, Draxler RR (2014) Root mean square error (RMSE) or mean absolute error (MAE)?–arguments against avoiding RMSE in the literature. Geosci Model Dev 7(3):1247–1250
Germec M, Bader NB, Turhan I (2018) Dilute acid and alkaline pretreatment of spent tea leaves to determine the potential of carbon sources. Biomass Convers Biorefinery 8(3):529–544
Ross T (1996) Indices for performance evaluation of predictive models in food microbiology. J Appl Microbiol 81(5):501–508
Skandamis PN, Nychas G-JE (2000) Development and evaluation of a model predicting the survival of Escherichia coli O157: H7 NCTC 12900 in homemade eggplant salad at various temperatures, pHs, and oregano essential oil concentrations. Appl Environ Microbiol 66(4):1646–1653
Ross T (1999) Predictive food microbiology models in the meat industry. Meat and Livestock Australia
Ross T, Dalgaard P, Tienungoon S (2000) Predictive modelling of the growth and survival of Listeria in fishery products. Int J Food Microbiol 62(3):231–245
Feng J, Zhang J-S, Jia W, Yang Y, Liu F, Lin C-C (2014) An unstructured kinetic model for the improvement of triterpenes production by Ganoderma lucidum G0119 based on nitrogen source effect. Biotechnol Bioprocess Eng 19(4):727–732
Marquardt DW (1963) An algorithm for least-squares estimation of nonlinear parameters. J Soc Ind Appl Math 11(2):431–441
Çelekli A, Yavuzatmaca M (2009) Predictive modeling of biomass production by Spirulina platensis as function of nitrate and NaCl concentrations. Bioresour Technol 100(5):1847–1851
Wasungu K, Simard R (1982) Growth characteristics of bakers’ yeast in ethanol. Biotechnol Bioeng 24(5):1125–1134
Suresh S, Srivastava V, Sakthivel S, Arisutha S (2018) Kinetic modeling of ethanol production for substrate–microbe system. In: Biorefining of Biomass to Biofuels. Springer, pp 361–372
Shuler ML, Kargi F (2017) Major metabolic pathways. In: ShuRiver ML, Kargi F (eds) Bioprocess engineering: basic concepts, 2nd edn. Prentice Hall, Upper Saddle River
Germec M, Gürler HN, Özcan A, Erkan SB, Karahalil E, Turhan I (2019) Medium optimization and kinetic modeling for the production of Aspergillus niger inulinase. Bioprocess Biosyst Eng 43:217–232. https://doi.org/10.1007/s00449-019-02219-1
Funding
This work was supported by the Akdeniz University Research Foundation (Grant number 2014.02.0121.020).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Germec, M., Karhan, M., Demirci, A. et al. Implementation of flexible models to bioethanol production from carob extract–based media in a biofilm reactor. Biomass Conv. Bioref. 11, 2983–2999 (2021). https://doi.org/10.1007/s13399-020-00612-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-020-00612-5