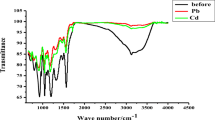
Abstract
The study represents the synthesis and characterization of two new pyrazine-based polymers to remove mercury(II) ions from wastewater solutions. The polymers were synthesized via the polycondensation of 2,6-diaminopyrazine (MXR-1) or 2,3-diaminopyrazine (MXR-2) with terepthaldehyde and glacial acetic acid as a catalyst. The polymerization reactions revealed the formation of a polyamine (MXR-1) and a polyimine (MXR-2). The SEM–EDX analysis showed a sphere-like structure for MXR-1 with a surface area of 56 m2/g, whereas MXR-2 showed a sheet-like structure with a surface area of 26 m2/g, the higher surface area of MXR-1 showed better adsorption capacity compared to MXR-2. The efficiency of the new polymers was tested to remove mercury ions at low concentrations from wastewater solutions. The effect of concentration on the adsorption capacity of MXR-1 to mercury ions showed that the experimental data fitted both Langmuir and Freundlich isotherm models, indicating homogeneous and heterogenous adsorption, respectively, whereas MXR-2 fitted the Freundlich isotherm model. The effect of time on the adsorption capacity study revealed that the adsorption is chemisorption in nature by pseudo-second-order kinetic model for both MXR-1 and MXR-2 polymers. The thermodynamic properties revealed that the adsorption process is endothermic in nature. The new polymers were tested on spiked wastewater samples and showed the superiority of MXR-1 polymer over MXR-2 with higher efficiency in the removal of mercury from wastewater solutions with a 95% removal efficiency. The study reveals the potency of MXR-1 polymer as an adsorbent for wastewater purification.
Graphic Abstract











Similar content being viewed by others
References
Ahmad, M.; Wang, J.; Xu, J.; Yang, Z.; Zhang, Q.; Zhang, B.: Novel synthetic method for magnetic sulphonated tubular trap for efficient mercury removal from wastewater. J. Colloid Interface Sci. 565, 523–535 (2020). https://doi.org/10.1016/j.jcis.2020.01.024
Ng, A.; Weerakoon, D.; Lim, E.; Padhye, L.P.: Fate of environmental pollutants. Water Environ. Res. 91(10), 1294–1325 (2019). https://doi.org/10.1002/wer.1225
Hasanpour, M.; Hatami, M.: Application of three dimensional porous aerogels as adsorbent for removal of heavy metal ions from water/wastewater: a review study. Adv. Coll. Interface. Sci. 284, 102247 (2020). https://doi.org/10.1016/j.cis.2020.102247
Leopold, K.; Foulkes, M.; Worsfold, P.: Methods for the determination and speciation of mercury in natural waters—a review. Anal. Chim. Acta 663(2), 127–138 (2010). https://doi.org/10.1016/j.aca.2010.01.048
Kurwadkar, S.: Occurrence and distribution of organic and inorganic pollutants in groundwater. Water Environ. Res. 91(10), 1001–1008 (2019). https://doi.org/10.1002/wer.1166
Pirrone, N.; Cinnirella, S.; Feng, X.; Finkelman, R.B.; Friedli, H.R.; Leaner, J.; Mason, R.; Mukherjee, A.B.; Stracher, G.B.; Streets, D.G.; Telmer, K.: Global mercury emissions to the atmosphere from anthropogenic and natural sources. Atmos. Chem. Phys. 10(13), 5951–5964 (2010). https://doi.org/10.5194/acp-10-5951-2010
Enrico, M.; Mere, A.; Zhou, H.; Loriau, M.; Tessier, E.; Bouyssiere, B.: Methods for total and speciation analysis of mercury in the petroleum industry. Energy Fuels 34(11), 13307–13320 (2020). https://doi.org/10.1021/acs.energyfuels.0c02730
Davidson, P.W.; Cory-Slechta, D.A.; Thurston, S.W.; Huang, L.-S.; Shamlaye, C.F.; Gunzler, D.; Watson, G.; van Wijngaarden, E.; Zareba, G.; Klein, J.D.; Clarkson, T.W.; Strain, J.J.; Myers, G.J.: Fish consumption and prenatal methylmercury exposure: cognitive and behavioral outcomes in the main cohort at 17 years from the Seychelles child development study. Neurotoxicology 32(6), 711–717 (2011). https://doi.org/10.1016/j.neuro.2011.08.003
tv1`Stokes-Riner, A., Thurston, S.W., J. Myers, G., Duffy, E.M., Wallace, J., Bonham, M., Robson, P., Shamlaye, C.F., Strain, J.J., Watson, G., Davidson, P.W.: A longitudinal analysis of prenatal exposure to methylmercury and fatty acids in the Seychelles. Neurotoxicol. Teratol. 33(2), 325–328 (2011). https://doi.org/10.1016/j.ntt.2010.11.003
Furieri, L.B.; Fioresi, M.; Junior, R.F.R.; Bartolomé, M.V.; Fernandes, A.A.; Cachofeiro, V.; Lahera, V.; Salaices, M.; Stefanon, I.; Vassallo, D.V.: Exposure to low mercury concentration in vivo impairs myocardial contractile function. Toxicol. Appl. Pharmacol. 255(2), 193–199 (2011). https://doi.org/10.1016/j.taap.2011.06.015
Ramezani, M.S.; Ozdemir, J.; Khosropour, A.R.; Beyzavi, H.: Sulfur-decorated hyper-cross-linked coal tar: a microporous organic polymer for efficient and expeditious mercury removal. ACS Appl. Mater. Interfaces 12(39), 44117–44124 (2020). https://doi.org/10.1021/acsami.0c10617
Štandeker, S.; Veronovski, A.; Novak, Z.; Knez, Ž: Silica aerogels modified with mercapto functional groups used for Cu(II) and Hg(II) removal from aqueous solutions. Desalination 269(1), 223–230 (2011). https://doi.org/10.1016/j.desal.2010.10.064
Bibby, A.; Mercier, L.: Mercury(II) ion adsorption behavior in thiol-functionalized mesoporous silica microspheres. Chem. Mater. 14(4), 1591–1597 (2002). https://doi.org/10.1021/cm0112082
Khajeh, M.; Laurent, S.; Dastafkan, K.: Nanoadsorbents: classification, preparation, and applications (with emphasis on aqueous media). Chem. Rev. 113(10), 7728–7768 (2013). https://doi.org/10.1021/cr400086v
Ramalingam, B.; Parandhaman, T.; Choudhary, P.; Das, S.K.: Biomaterial functionalized graphene-magnetite nanocomposite: a novel approach for simultaneous removal of anionic dyes and heavy-metal ions. ACS Sustain. Chem. Eng. 6(5), 6328–6341 (2018). https://doi.org/10.1021/acssuschemeng.8b00139
Zeng, H.; Wang, L.; Zhang, D.; Wang, F.; Sharma, V.K.; Wang, C.: Amido-functionalized carboxymethyl chitosan/montmorillonite composite for highly efficient and cost-effective mercury removal from aqueous solution. J. Colloid Interface Sci. 554, 479–487 (2019). https://doi.org/10.1016/j.jcis.2019.07.029
Adly, M.S.; El-Dafrawy, S.M.; Ibrahim, A.A.; El-Hakam, S.A.; El-Shall, M.S.: Efficient removal of heavy metals from polluted water with high selectivity for Hg(ii) and Pb(ii) by a 2-imino-4-thiobiuret chemically modified MIL-125 metal–organic framework. RSC Adv. 11(23), 13940–13950 (2021). https://doi.org/10.1039/D1RA00927C
Awad, F.S.; Bakry, A.M.; Ibrahim, A.A.; Lin, A.; El-Shall, M.S.: Thiol- and amine-incorporated UIO-66-NH2 as an efficient adsorbent for the removal of Mercury(II) and phosphate ions from aqueous solutions. Ind. Eng. Chem. Res. 60(34), 12675–12688 (2021). https://doi.org/10.1021/acs.iecr.1c01892
Foroutan, R.; Peighambardoust, S.J.; Ahmadi, A.; Akbari, A.; Farjadfard, S.; Ramavandi, B.: Adsorption mercury, cobalt, and nickel with a reclaimable and magnetic composite of hydroxyapatite/Fe3O4/polydopamine. J. Environ. Chem. Eng. 9(4), 105709 (2021). https://doi.org/10.1016/j.jece.2021.105709
Sang, Y.; Cao, Y.; Wang, L.; Yan, W.; Chen, T.; Huang, J.; Liu, Y.-N.: N-rich porous organic polymers based on Schiff base reaction for CO2 capture and mercury(II) adsorption. J. Colloid Interface Sci. 587, 121–130 (2021). https://doi.org/10.1016/j.jcis.2020.12.002
Tan, M.X.; Sum, Y.N.; Ying, J.Y.; Zhang, Y.: A mesoporous poly-melamine-formaldehyde polymer as a solid sorbent for toxic metal removal. Energy Environ. Sci. 6(11), 3254–3259 (2013). https://doi.org/10.1039/C3EE42216J
Ryu, J.; Lee, M.Y.; Song, M.G.; Baeck, S.-H.; Shim, S.E.; Qian, Y.: Highly selective removal of Hg(II) ions from aqueous solution using thiol-modified porous polyaminal-networked polymer. Sep. Purif. Technol. 250, 117120 (2020). https://doi.org/10.1016/j.seppur.2020.117120
Denizli, A.; Kesenci, K.; Arica, Y.; Pişkin, E.: Dithiocarbamate-incorporated monosize polystyrene microspheres for selective removal of mercury ions. React. Funct. Polym. 44(3), 235–243 (2000). https://doi.org/10.1016/S1381-5148(99)00099-1
Shen, J.; Ren, C.; Zeng, H.: Surprisingly high selectivity and high affinity in mercury recognition by H-bonded cavity-containing aromatic foldarands. J. Am. Chem. Soc. 139(15), 5387–5396 (2017). https://doi.org/10.1021/jacs.6b13342
Suriyakumar, S.; Bhardwaj, P.; Grace, A.N.; Stephan, A.M.: Role of polymers in enhancing the performance of electrochemical supercapacitors: a review. Batteries Supercaps 4(4), 571–584 (2021). https://doi.org/10.1002/batt.202000272
Meng, L.; Ren, S.; Ma, C.; Yu, Y.; Lou, Y.; Zhang, D.; Shi, Z.: Synthesis of a 2D nitrogen-rich π-conjugated microporous polymer for high performance lithium-ion batteries. Chem. Commun. 55(64), 9491–9494 (2019). https://doi.org/10.1039/C9CC04036F
Yang, L.; Cui, X.; Yang, Q.; Qian, S.; Wu, H.; Bao, Z.; Zhang, Z.; Ren, Q.; Zhou, W.; Chen, B.; Xing, H.: A single-molecule propyne trap: highly efficient removal of propyne from propylene with anion-pillared ultramicroporous materials. Adv. Mater. 30(10), 1705374 (2018). https://doi.org/10.1002/adma.201705374
Etaiw, S.E.-d.H., Marie, H., Shalaby, E.M., Farag, R.S., Elsharqawy, F.A.: Sensing and photocatalytic properties of nanosized Cu(I)CN organotin supramolecular coordination polymer based on pyrazine. Appl. Organomet. Chem. 33(9), e5114 (2019). https://doi.org/10.1002/aoc.5114
Skorjanc, T.; Shetty, D.; Valant, M.: Covalent organic polymers and frameworks for fluorescence-based sensors. ACS Sensors 6(4), 1461–1481 (2021). https://doi.org/10.1021/acssensors.1c00183
Palani, P.; Karpagam, S.: Conjugated polymers—a versatile platform for various photophysical, electrochemical and biomedical applications: a comprehensive review. New J. Chem. 45(41), 19182–19209 (2021). https://doi.org/10.1039/D1NJ04062F
Fayazi, M.; Taher, M.A.; Afzali, D.; Mostafavi, A.; Ghanei-Motlagh, M.: Synthesis and application of novel ion-imprinted polymer coated magnetic multi-walled carbon nanotubes for selective solid phase extraction of lead(II) ions. Mater. Sci. Eng. C 60, 365–373 (2016). https://doi.org/10.1016/j.msec.2015.11.060
Hou, Y.; Masoomi, M.Y.; Bagheri, M.; Morsali, A.; Joo, S.W.: Two reversible transformable mercury(II) coordination polymers as efficient adsorbents for removal of dibenzothiophene. RSC Adv. 5(99), 81356–81361 (2015). https://doi.org/10.1039/C5RA12686J
Lohse, M.S.; Bein, T.: Covalent organic frameworks: structures, synthesis, and applications. Adv. Funct. Mater. 28(33), 1705553 (2018). https://doi.org/10.1002/adfm.201705553
Manzar, M.S., Haladu, S.A., Zubair, M., Mu'azu, N.D., Qureshi, A., Blaisi, N.I., Garrison, T.F., Al Hamouz, O.C.S.: Synthesis and characterization of a series of cross-linked polyamines for removal of Erichrome Black T from aqueous solution. Chin. J. Chem. Eng. 32, 341–352 (2021). https://doi.org/10.1016/j.cjche.2020.09.052
Zeng, H.; Wang, L.; Zhang, D.; Yan, P.; Nie, J.; Sharma, V.K.; Wang, C.: Highly efficient and selective removal of mercury ions using hyperbranched polyethylenimine functionalized carboxymethyl chitosan composite adsorbent. Chem. Eng. J. 358, 253–263 (2019). https://doi.org/10.1016/j.cej.2018.10.001
Abdelnaby, M.M., Cordova, K.E., Abdulazeez, I., Alloush, A.M., Al-Maythalony, B.A., Mankour, Y., Alhooshani, K., Saleh, T.A., Al Hamouz, O.C.S.: Novel porous organic polymer for the concurrent and selective removal of hydrogen sulfide and carbon dioxide from natural gas streams. ACS Appl. Mater. Interfaces 12(42), 47984–47992 (2020). https://doi.org/10.1021/acsami.0c14259
Zhong, J.; Wang, B.; Sun, K.; Duan, J.: Hyperbranched polyethylenimine–based polymeric nanoparticles: synthesis, properties, and an application in selective response to copper ion. Colloid Polym. Sci. 299(10), 1577–1586 (2021). https://doi.org/10.1007/s00396-021-04885-8
Englert, C.; Tauhardt, L.; Hartlieb, M.; Kempe, K.; Gottschaldt, M.; Schubert, U.S.: Linear poly(ethylene imine)-based hydrogels for effective binding and release of DNA. Biomacromol 15(4), 1124–1131 (2014). https://doi.org/10.1021/bm4017572
Chaudhuri, H.; Gupta, R.; Dash, S.: Efficient synthesis of branched polyamine based thermally stable heterogeneous catalyst for knoevenagel condensation at room temperature. Catal. Lett. 148(6), 1703–1713 (2018). https://doi.org/10.1007/s10562-018-2368-6
Tarase, M.V.; Zade, A.B.; Gurnule, W.B.: Kinetics of thermal degradation studies of some new terpolymers derived from 2,4-dihydroxypropiophenone, oxamide, and formaldehyde. J. Appl. Polym. Sci. 116(2), 619–627 (2010). https://doi.org/10.1002/app.30844
Alloush, A.M., Abdelnaby, M.M., Cordova, K.E., Qasem, N.A.A., Al-Maythalony, B.A., Jalilov, A., Mankour, Y., Al Hamouz, O.C.S.: Selectively capturing carbon dioxide from mixed gas streams using a new microporous organic copolymer. Microp. Mesop. Mater. 305, 110391 (2020). https://doi.org/10.1016/j.micromeso.2020.110391
Wang, J.; Deng, B.; Chen, H.; Wang, X.; Zheng, J.: Removal of aqueous Hg(II) by polyaniline: sorption characteristics and mechanisms. Environ. Sci. Technol. 43(14), 5223–5228 (2009). https://doi.org/10.1021/es803710k
Foti, C.; Giuffrè, O.; Lando, G.; Sammartano, S.: Interaction of inorganic Mercury(II) with polyamines, polycarboxylates, and amino acids. J. Chem. Eng. Data 54(3), 893–903 (2009). https://doi.org/10.1021/je800685c
Liu, J., Ma, Y., Zhang, Y., Shao, G.: Novel negatively charged hybrids. 3. Removal of Pb2+ from aqueous solution using zwitterionic hybrid polymers as adsorbent. J. Hazard. Mater. 173, 438–444 (2010). https://doi.org/10.1016/j.jhazmat.2009.08.097
Minceva, M., Markovska, L., Meshko, V.: Removal of Zn2+, Cd2+, and Pb2+ from binary aqueous solution by natural zeolite and granulated activated carbon. Maced. J. Chem. Chem. Eng. 26, 125–134 (2007).
Wang, X.S., Miao, H.H., He, W., Shen, H.L.: Competitive adsorption of Pb(II), Cu(II), and Cd(II) ions on wheat-residue derived black carbon. J. Chem. Eng. Data 56, 444–449 (2011). https://doi.org/10.1021/je101079w
Boparai, H.K., Joseph, M., O'Carroll, D.M.: Kinetics and thermodynamics of cadmium ion removal by adsorption onto nano zerovalent iron particles. J. Hazard. Mater. 186, 458–465 (2011). https://doi.org/10.1016/j.jhazmat.2010.11.029
Mohapatra, M., Rout, K., Mohapatra, B.K., Anand, S.: Sorption behavior of Pb(II) and Cd(II) on iron ore slime and characterization of metal ion loaded sorbent. J. Hazard. Mater. 166, 1506–1513 (2009).
Ozcan, A., Ozcan, A.S., Gok, O.: Adsorption kinetics and isotherms of anionic dye of Reactive Blue 19 from aqueous solutions onto DTMA-sepiolite. In: 2007, pp. 225–249. Nova Science Publishers, Inc.
Wu, F.C.; Tseng, R.L.; Juang, R.S.: Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. Chem. Eng. J. 153(1–3), 1–8 (2009)
Al-Yaari, M.; Saleh, T.A.; Saber, O.: Removal of mercury from polluted water by a novel composite of polymer carbon nanofiber: kinetic, isotherm, and thermodynamic studies. RSC Adv. 11(1), 380–389 (2021). https://doi.org/10.1039/D0RA08882J
Al-Ghouti, M.A.; Da’ana, D.; Abu-Dieyeh, M.; Khraisheh, M.: Adsorptive removal of mercury from water by adsorbents derived from date pits. Sci. Rep. 9(1), 15327 (2019). https://doi.org/10.1038/s41598-019-51594-y
Cao, R.; Fan, M.; Hu, J.; Ruan, W.; Xiong, K.; Wei, X.: Optimizing low-concentration mercury removal from aqueous solutions by reduced graphene oxide-supported Fe3O4 composites with the aid of an artificial neural network and genetic algorithm. Materials (Basel) 10(11), 1279 (2017). https://doi.org/10.3390/ma10111279
Luo, F.; Chen, J.L.; Dang, L.L.; Zhou, W.N.; Lin, H.L.; Li, J.Q.; Liu, S.J.; Luo, M.B.: High-performance Hg2+ removal from ultra-low-concentration aqueous solution using both acylamide- and hydroxyl-functionalized metal–organic framework. J. Mater. Chem. A 3(18), 9616–9620 (2015). https://doi.org/10.1039/C5TA01669J
Girginova, P.I.; Daniel-da-Silva, A.L.; Lopes, C.B.; Figueira, P.; Otero, M.; Amaral, V.S.; Pereira, E.; Trindade, T.: Silica coated magnetite particles for magnetic removal of Hg2+ from water. J. Colloid Interface Sci. 345(2), 234–240 (2010). https://doi.org/10.1016/j.jcis.2010.01.087
Looney, B.B.; Denham, M.E.; Vangelas, K.M.; Bloom, N.S.: Removal of mercury from low-concentration aqueous streams using chemical reduction and air stripping. J. Environ. Eng. 129(9), 819–825 (2003). https://doi.org/10.1061/(ASCE)0733-9372(2003)129:9(819)
Xiong, Y.Y.; Li, J.Q.; Gong, L.L.; Feng, X.F.; Meng, L.N.; Zhang, L.; Meng, P.P.; Luo, M.B.; Luo, F.: Using MOF-74 for Hg2+ removal from ultra-low concentration aqueous solution. J. Solid State Chem. 246, 16–22 (2017). https://doi.org/10.1016/j.jssc.2016.10.018
Acknowledgements
The authors would like to thank the support provided by Chemistry Department, King Fahd University of Petroleum and Minerals.
Author information
Authors and Affiliations
Contributions
Rakan AlRashidi was involved methodology of the work, investigation, formal analysis, resources, and software. Othman Charles S. Al Hamouz helped in visualization, supervision, investigation, and writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
AlRashidi, R.B., Al Hamouz, O.C.S. Synthesis of Cross-Linked Pyrazine-Based Polymers for Selective Removal of Mercury(II) Ions from Wastewater Solutions. Arab J Sci Eng 47, 7207–7218 (2022). https://doi.org/10.1007/s13369-022-06833-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-022-06833-2