Abstract
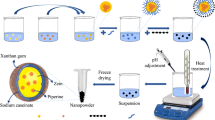
Ionotropic gelation followed by polyelectrolyte complexion was used for the synthesis of nanoparticles based on sodium alginate (NaAL) and chitosan (CS) for encapsulation of ibuprofen (IBP), Fourier transform infrared spectroscopy, differential scanning calorimetry, field-emission scanning electron microscopes, percentage of encapsulation efficiency and loading capacity have been used to confirm the synthesis and encapsulation of IBP. The results obtained show that they have a size of around 100 nm. In vitro drug release and evaluation of anti-inflammatory activity in different media indicate that the nanoparticles are pH-sensitive and permit the protection of the drug against total dissolution in the gastric medium, control its release, and it increases the solubility and biological activity of IBP. The analytical data of the in vitro drug release in the simulated gastric fluid (SGF), simulated interstitial fluid (SIF) and phosphate buffered saline (PBS) were fitted to the different kinetic models (zero-order, first-order, Higuchi, Korsmeyer–Pepass and Kopcha), and the results indicate that IBP is released by diffusion-controlled in SIF and PBS, and by erosion-controlled in the SGF. The toxicity of nanoparticles was tested against Artemia salina that displayed non-toxicity effects. In vitro biodegradation by lysozyme and all the results obtained indicated that a NaAL/CS nanoparticles are a promising system for IBP release that can achieve pseudo-zero-order kinetics.







Similar content being viewed by others
References
Arica, B.; Çaliş, S.; Atilla, P.; Durlu, N.T.; Çakar, N.; Kaş, H.S.; Hincal, A.A.: In vitro and in vivo studies of ibuprofen-loaded biodegradable alginate beads. J. Microencapsul. (2005). https://doi.org/10.1080/02652040400026319
Hattori, Y.; Haruna, Y.; Otsuka, M.: Dissolution process analysis using model-free Noyes–Whitney integral equation. Colloids. Surf. B. Biointerfaces (2013). https://doi.org/10.1016/j.colsurfb.2012.08.017
Liu, J.; Xiao, J.; Li, F.; Shi, Y.; Li, D.; Huang, Q.: Chitosan-sodium alginate Nanoparticle as a delivery system for ε-polylysine: preparation, characterization and antimicrobial activity. Food Control (2018). https://doi.org/10.1016/j.foodcont.2018.04.020
Kumari, A.; Yadav, K.S.; Yadav, S.C.: Biodegradable polymeric Nanoparticles based drug delivery systems. Colloids Surf. B. Biointerfaces (2010). https://doi.org/10.1016/j.colsurfb.2009.09.001
Fessi, H.; Puisieux, F.; Devissaguet, JPh; Ammoury, N.; Benita, S.: Nanocapsule formation by interfacial polymer deposition following solvent displacement. Int. J. Pharm. (1989). https://doi.org/10.1016/0378-5173(89)90281-0
Niwa, T.; Takeuchi, H.; Hino, T.; Kunou, N.; Kawashima, Y.: Preparations of biodegradable Nanospheres of water-soluble and insoluble drugs with D, L-lactide/glycolide copolymer by a novel spontaneous emulsification solvent diffusion method, and the drug release behavior. J. Control. Release (1993). https://doi.org/10.1016/0168-3659(93)90097-O
Allémann, E.; Leroux, J.; Gurny, R.: In vitro extended-release properties of drug-loaded poly(DL-lactic acid) nanoparticles produced by a salting-out procedure. Pharm. Res. (1993). https://doi.org/10.1023/A:1018970030327
Rajaonarivony, M.; Vauthier, C.; Couarraze, G.; Puisieux, F.; Couvreur, P.: Development of a new drug carrier made from alginate. J. Pharm. Sci. (1993). https://doi.org/10.1002/jps.2600820909
Vauthier, C.; Bouchemal, K.: Methods for the preparation and manufacture of polymeric nanoparticles. Pharm. Res. (2009). https://doi.org/10.1007/s11095-008-9800-3
Thakur, S.; Sharma, B.; Verma, A.; Chaudhary, J.; Tamulevicius, S.; Thakur, V.K.: Recent progress in sodium alginate based sustainable hydrogels for environmental applications. J. Clean. Prod. (2018). https://doi.org/10.1016/j.jclepro.2018.06.259
Sarmento, B.; Ribeiro, A.J.; Veiga, F.; Ferreira, D.C.; Neufeld, R.J.: Insulin-loaded nanoparticles are prepared by alginate ionotropic pre-gelation followed by chitosan polyelectrolyte complexation. J. Nanosci. Nanotechnol. (2007). https://doi.org/10.1166/jnn.2007.609
Henao, E.; Delgado, E.; Contreras, H.; Quintana, G.: Polyelectrolyte complexation versus ionotropic gelation for chitosan-based hydrogels with carboxymethylcellulose, carboxymethyl starch, and alginic acid. Int. J. Chem. Eng (2018). https://doi.org/10.1155/2018/3137167
Boddohi, S.; Moore, N.; Johnson, P.A.; Kipper, M.J.: Polysaccharide-based polyelectrolyte complex nanoparticles from chitosan, heparin, and hyaluronan. Biomacromolecules (2009). https://doi.org/10.1021/bm801513e
Nalini, T.; Khaleel Basha, S.; Majeeth, A.M.S.; Sugantha Kumari, V.; Kaviyarasu, K.: Development and characterization of alginate/chitosan nanoparticulate system for hydrophobic drug encapsulation. J Drug Deliv Sci Technol (2019). https://doi.org/10.1016/j.jddst.2019.04.002
Naskar, S.; Sharma, S.; Kuotsu, K.: Chitosan-based nanoparticles: an overview of biomedical applications and its preparation. J Drug Deliv Sci Technol (2019). https://doi.org/10.1016/j.jddst.2018.10.022
Prabaharan, M.: Chitosan-based nanoparticles for tumor-targeted drug delivery. Int. J. Biol. Macromol. (2014). https://doi.org/10.1016/j.ijbiomac.2014.10.052
Yadav, G.; Bansal, M.; Thakur, N.; Sargam Khare, P.: Multilayer tablets and their drug release kinetic models for oral controlled drug delivery system. Middle-East J. Sci. Res. (2013). https://doi.org/10.5829/idosi.mejsr.2013.16.06.75176
Peppas, N.A.; Sahlin, J.J.: A simple equation for the description of solute release. II. Coupling of diffusion and relaxation. Int. J. Pharm., Int (1989). https://doi.org/10.1016/0378-5173(89)90306-2
Kopcha, M.; Lordi, N.G.; Tojo, K.J.: Evaluation of release from selected thermosoftening vehicles. J. Pharm. Pharmacol. (1991). https://doi.org/10.1111/j.2042-7158.1991.tb03
Karthik, K.; Ravi, M.; BharathRathnaKumar, P.: Evaluation of anti-inflammatory activity of canthium parviflorum by in vitro method. Biotech. Pharm. Res, Indian J (2013). https://doi.org/10.21276/ajptr
Gomathi, T.; Sudha, P.N.; Florence, J.A.K.; Venkatesan, J.; Sukumaran, A.: Fabrication of letrozole formulation using chitosan nanoparticles through ionic gelation method. Int. J. Biol. Macromol. (2017). https://doi.org/10.1016/j.ijbiomac.2017.01.147
Michael, A.S.; Thompson, C.G.; Abramovitz, M.: Artemia salina as a test organism for bioassay. Science (1956). https://doi.org/10.1126/science.123.3194.464
Solairaj, D.; Rameshthangam, P.; Muthukumaran, P.; Wilson, J.: Studies on electrochemical glucose sensing, antimicrobial activity and cytotoxicity of fabricated copper nanoparticle immobilized chitin nanostructure. Int. J. Biol. Macromol. (2017). https://doi.org/10.1016/j.ijbiomac.2017.03.147
Meyer, B.N.; Ferrigni, N.R.; Putnam, J.E.; Jacobsen, L.B.; Nichols, D.E.; McLaughlin, J.L.: Brine shrimp: a convenient general bioassay for active plant constituents. Planta Med. (1982). https://doi.org/10.1055/s-2007-971236
Hirai, A.; Odani, H.; Nakajima, A.: Determination of degree of deacetylation of chitosan by 1H NMR spectroscopy. Polym. Bull. (1991). https://doi.org/10.1007/BF00299352
Smitha, B.; Sridhar, S.; Khan, A.A.: Chitosan–sodium alginate polyion complexes as fuel cell membranes. Eur. Polym J. (2005). https://doi.org/10.1016/j.eurpolymj.2005.02.018
Rathore, H.S.; Senthilvelan, T.; Vasantharaja, R.; Abraham, L.S.; Prakash, D.; Sivagnanam, U.T.; Gupta, S.: Fabrication and characterization of chitosan film impregnated ciprofloxacin drug: a comparative study. ISBAB (2019). https://doi.org/10.1016/j.bcab.2019.101078
Pereira, F.S.; da Silva Agostini, D.L.; Job, A.E.: Thermal studies of chitin–chitosan derivatives. J. Therm. Anal. Calorim. (2013). https://doi.org/10.1007/s10973-012-2835-z
Wang, Y.; Pitto-barry, A.; Habtemariam, A.; Romero-canelon, I.; Sadler, P.J.; Barry, N.P.E.: Nanoparticles of chitosan conjugated to organo-ruthenium complexes. Inorg. Chem. Front. (2016). https://doi.org/10.1039/c6qi00115g
Ramukutty, S.; Ramachandran, E.: Growth, spectral and thermal studies of ibuprofen crystals. Cryst. Res. Technol. (2012). https://doi.org/10.1002/crat.201100394
Sarmento, B.; Ferreira, D.; Veiga, F.; Ribeiro, A.: Characterization of insulin-loaded alginate nanoparticles produced by ionotropic pre-gelation through DSC and FTIR studies. Carbohydr. Polym. (2006). https://doi.org/10.1016/j.carbpol.2006.02.008
Voo, W.P.; Lee, B.B.; Idris, A.; Islam, A.; Tey, B.; Chan, E.S.: Production of ultra-high concentration calcium alginate beads with prolonged dissolution profile. RSC Adv. (2015). https://doi.org/10.1039/C5RA03862F
Saravanan, M.; Bhaskar, K.; Srinivasa, R.G.; Dhanaraju, M.D.: Ibuprofen-loaded ethylcellulose/polystyrene microspheres: an approach to get prolonged drug release with reduced burst effect and low ethylcellulose content. J. Microencapsul. (2003). https://doi.org/10.1080/0265204031000093087
Singh, R.; Lillard, J.W.: Nanoparticle-based targeted drug delivery. Exp. Mol. Pathol. (2012). https://doi.org/10.1016/j.yexmp.2008.12.004
Longmire, M.; Choyke, P.L.; Kobayashi, H.: Clearance properties of nano-sized particles and molecules as imaging agents: considerations and caveats. Nanomedicine (London) (2012). https://doi.org/10.2217/17435889.3.5.703
Li, P.; Dai, Y.; Zhang, J.; Wang, A.; Wei, Q.: Chitosan-alginate nanoparticles as a novel drug delivery system for nifedipine. Int. J. Biomed. Sci. 4, 221–228 (2008)
Bajpai, S.K.; Sharma, S.: Investigation of swelling/degradation behaviour of alginate beads crosslinked with Ca2+ and Ba2+ ions. React. Funct. Polym. (2004). https://doi.org/10.1016/j.reactfunctpolym.2004.01.002
Acknowledgements
The authors would like to thank the biotechnology research centre (CRBt) for their supporting.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bensouiki, S., Belaib, F., Sindt, M. et al. Evaluation of Anti-inflammatory Activity and In Vitro Drug Release of Ibuprofen-Loaded Nanoparticles Based on Sodium Alginate and Chitosan. Arab J Sci Eng 45, 7599–7609 (2020). https://doi.org/10.1007/s13369-020-04720-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-020-04720-2