Abstract
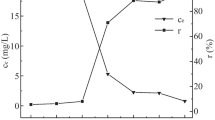
Rice husk-mediated magnetic biochar (RH-MBC) was manufactured in the present study by liquefaction method. It is applied on the anionic and cationic dyes to investigate their dye adsorption capacity from aqueous solutions. Characterization of the prepared materials has been conducted by the Fourier transform infrared spectroscopy (FTIR), X-ray diffraction (XRD), scanning electron microscope, energy-dispersive X-ray, transmission electron microscopy, Brunauer–Emmett–Teller, vibrating sample magnetometer and point of zero charge (PZC) analysis. From the analyses, it is revealed that the manufactured RH-MBC is super-magnetic and more porous with higher surface area compared with its parent materials. After investigating the adsorption data with different adsorption isotherm and kinetic models, the data were best-fitted with Freundlich model and pseudo-second-order model. The present material shows higher adsorption capacity for representative cationic dye CV (80.04 mg/g) rather than the anionic dye EBT (5.09 mg/g). From the FTIR, XRD, PZC and isotherm studies, it is revealed that the dyes adsorption onto the RH-MBC mainly happened due to the presence of the functional groups like C–O, Fe3O4, –OH, C=C and C–O. It is concluded that the novel functionalized RH-MBC is an approach to use the waste material (rice husk) to use for wastewater treatment and highly efficient for cationic dyes adsorption. Additionally, due to its super-magnetic properties, it is easily separable by using external magnetic field.



















Similar content being viewed by others
References
Guo, W.; Wang, S.; Wang, Y.; Lu, S.; Gao, Y.: Sorptive removal of phenanthrene from aqueous solutions using magnetic and non-magnetic rice husk-derived biochars. R. Soc. Open Sci. 5, 172382 (2018). https://doi.org/10.1098/rsos.172382
Chieng, H.I.; Lim, L.B.L.; Priyantha, N.: Sorption characteristics of peat from Brunei Darussalam for the removal of rhodamine B dye from aqueous solution: adsorption isotherms, thermodynamics, kinetics and regeneration studies. Desalin Water Treat 55, 664–677 (2015). https://doi.org/10.1080/19443994.2014.919609
Salleh, M.A.M.; Mahmoud, D.K.; Karim, W.A.W.A.; Idris, A.: Cationic and anionic dye adsorption by agricultural solid wastes: a comprehensive review. Desalination 280, 1–13 (2011). https://doi.org/10.1016/j.desal.2011.07.019
Zhao, Y.; Li, J.; Zhao, L.; Zhang, S.; Huang, Y.; Wu, X.; Wang, X.: Synthesis of amidoxime-functionalized Fe3O4@SiO2 core–shell magnetic microspheres for highly efficient sorption of U(VI). Chem. Eng. J. 235, 275–283 (2014). https://doi.org/10.1016/j.cej.2013.09.034
Nguyen, T.A.H.; Ngo, H.H.; Guo, W.S.; Zhang, J.; Liang, S.; Yue, Q.Y.; Li, Q.; Nguyen, T.V.: Applicability of agricultural waste and by-products for adsorptive removal of heavy metals from wastewater. Bioresour. Technol. 148, 574–585 (2013). https://doi.org/10.1016/j.biortech.2013.08.124
Chuah, T.G.; Jumasiah, A.; Azni, I.; Katayon, S.; Thomas Choong, S.Y.: Rice husk as a potentially low-cost biosorbent for heavy metal and dye removal: an overview. Desalination 175, 305–316 (2005). https://doi.org/10.1016/j.desal.2004.10.014
Chen, Y.; Zhu, Y.; Wang, Z.; Li, Y.; Wang, L.; Ding, L.; Gao, X.; Ma, Y.; Guo, Y.: Application studies of activated carbon derived from rice husks produced by chemical–thermal process—a review. Adv. Colloid Interface Sci. 163, 39–52 (2011). https://doi.org/10.1016/j.cis.2011.01.006
Guo, Y.; Zhang, H.; Tao, N.; Liu, Y.; Qi, J.; Wang, Z.; Xu, H.: Adsorption of malachite green and iodine on rice husk-based porous carbon. Mater. Chem. Phys. 82, 107–115 (2003). https://doi.org/10.1016/S0254-0584(03)00191-3
Agrafioti, E.; Kalderis, D.; Diamadopoulos, E.: Arsenic and chromium removal from water using biochars derived from rice husk, organic solid wastes and sewage sludge. J. Environ. Manag. 133, 309–314 (2014). https://doi.org/10.1016/j.jenvman.2013.12.007
Denyes, M.J.; Langlois, V.S.; Rutter, A.; Zeeb, B.A.: The use of biochar to reduce soil PCB bioavailability to Cucurbita pepo and Eisenia fetida. Sci. Total Environ. 437, 76–82 (2012). https://doi.org/10.1016/j.scitotenv.2012.07.081
Lamichhane, S.; Bal Krishna, K.C.; Sarukkalige, R.: Polycyclic aromatic hydrocarbons (PAHs) removal by sorption: a review. Chemosphere 148, 336–353 (2016). https://doi.org/10.1016/j.chemosphere.2016.01.036
Li, H.; Qu, R.; Li, C.; Guo, W.; Han, X.; He, F.; Ma, Y.; Xing, B.: Selective removal of polycyclic aromatic hydrocarbons (PAHs) from soil washing effluents using biochars produced at different pyrolytic temperatures. Bioresour. Technol. 163, 193–198 (2014). https://doi.org/10.1016/j.biortech.2014.04.042
Chen, Y.; Yang, H.; Wang, X.; Zhang, S.; Chen, H.: Biomass-based pyrolytic polygeneration system on cotton stalk pyrolysis: influence of temperature. Bioresour. Technol. 107, 411–418 (2012). https://doi.org/10.1016/j.biortech.2011.10.074
Shang, J.; Pi, J.; Zong, M.; Wang, Y.; Li, W.; Liao, Q.: Chromium removal using magnetic biochar derived from herb-residue. J. Taiwan Inst. Chem. Eng. 68, 289–294 (2016). https://doi.org/10.1016/j.jtice.2016.09.012
Trakal, L.; Veselská, V.; Šafařík, I.; Vítková, M.; Číhalová, S.; Komárek, M.: Lead and cadmium sorption mechanisms on magnetically modified biochars. Bioresour. Technol. 203, 318–324 (2016). https://doi.org/10.1016/j.biortech.2015.12.056
Liu, S.; Huang, B.; Chai, L.; Liu, Y.; Zeng, G.; Wang, X.; Zeng, W.; Shang, M.; Deng, J.; Zhou, Z.: Enhancement of As(V) adsorption from aqueous solution by a magnetic chitosan/biochar composite. RSC Adv. 7, 10891–10900 (2017). https://doi.org/10.1039/C6RA27341F
Wang, P.; Wang, X.; Yu, S.; Zou, Y.; Wang, J.; Chen, Z.; Alharbi, N.S.; Alsaedi, A.; Hayat, T.; Chen, Y.; Wang, X.: Silica coated Fe3O4 magnetic nanospheres for high removal of organic pollutants from wastewater. Chem. Eng. J. 306, 280–288 (2016). https://doi.org/10.1016/j.cej.2016.07.068
Saucier, C.; Karthickeyan, P.; Ranjithkumar, V.; Lima, E.C.; Dos Reis, G.S.; De Brum, I.A.S.: Efficient removal of amoxicillin and paracetamol from aqueous solutions using magnetic activated carbon. Environ. Sci. Pollut. Res. 24, 5918–5932 (2017). https://doi.org/10.1007/s11356-016-8304-7
Liu, Y.; Yuan, X.; Huang, H.; Wang, X.; Wang, H.; Zeng, G.: Thermochemical liquefaction of rice husk for bio-oil production in mixed solvent (ethanol–water). Fuel Process. Technol. 112, 93–99 (2013). https://doi.org/10.1016/j.fuproc.2013.03.005
Park, J.; Hung, I.; Gan, Z.; Rojas, O.J.; Lim, K.H.; Park, S.: Activated carbon from biochar: influence of its physicochemical properties on the sorption characteristics of phenanthrene. Bioresour. Technol. 149, 383–389 (2013). https://doi.org/10.1016/j.biortech.2013.09.085
Gupta, P.L.; Jung, H.; Tiwari, D.; Kong, S.-H.; Lee, S.-M.: Insight into the mechanism of Cd(II) and Pb(II) removal by sustainable magnetic biosorbent precursor to Chlorella vulgaris. J. Taiwan Inst. Chem. Eng. 71, 206–213 (2017). https://doi.org/10.1016/j.jtice.2016.12.007
Ali, I.; Peng, C.; Lin, D.; Saroj, D.; Naz, I.; Khan, Z.M.; Sultan, M.; Ali, M.: Encapsulated green magnetic nanoparticles for the removal of toxic Pb2+ and Cd2+ from water: development, characterization and application. J. Environ. Manag. 234, 273–289 (2019). https://doi.org/10.1016/j.jenvman.2018.12.112
Islam, T.; Peng, C.: Synthesis of carbon embedded silica and zeolite from rice husk to remove trace element from aqueous solutions: characterization, optimization and equilibrium studies. Sep. Sci. Technol. (2019). https://doi.org/10.1080/01496395.2019.1658781
Rovani, S.; Censi, M.T.; Pedrotti, S.L.; Lima, É.C.; Cataluña, R.; Fernandes, A.N.: Development of a new adsorbent from agro-industrial waste and its potential use in endocrine disruptor compound removal. J. Hazard. Mater. 271, 311–320 (2014). https://doi.org/10.1016/j.jhazmat.2014.02.004
Shi, Q.; Li, A.; Zhu, Z.; Liu, B.: Adsorption of naphthalene onto a high-surface-area carbon from waste ion exchange resin. J. Environ. Sci. 25, 188–194 (2013). https://doi.org/10.1016/S1001-0742(12)60017-5
Saha, P.; Chowdhury, S.; Gupta, S.; Kumar, I.: Insight into adsorption equilibrium, kinetics and thermodynamics of Malachite Green onto clayey soil of Indian origin. Chem. Eng. J. 165, 874–882 (2010). https://doi.org/10.1016/j.cej.2010.10.048
Wang, S.; Zhu, Z.H.: Effects of acidic treatment of activated carbons on dye adsorption. Dyes Pigments 75, 306–314 (2007). https://doi.org/10.1016/j.dyepig.2006.06.005
de Luna, M.D.G.; Flores, E.D.; Genuino, D.A.D.; Futalan, C.M.; Wan, M.-W.: Adsorption of Eriochrome Black T (EBT) dye using activated carbon prepared from waste rice hulls—optimization, isotherm and kinetic studies. J. Taiwan Inst. Chem. Eng. 44, 646–653 (2013). https://doi.org/10.1016/j.jtice.2013.01.010
Mane, V.S.; Mall, I.D.; Srivastava, V.C.: Kinetic and equilibrium isotherm studies for the adsorptive removal of brilliant green dye from aqueous solution by rice husk ash. J. Environ. Manag. 84, 390–400 (2007). https://doi.org/10.1016/j.jenvman.2006.06.024
Filho, N.C.; Venancio, E.C.; Barriquello, M.F.; Hechenleitner, A.A.; Pineda, E.A.G.: Methylene blue adsorption onto modified lignin from sugarcane baggasse. Eclét. Quím. 32, 63–70 (2007). https://doi.org/10.1590/S0100-46702007000400009
Khattri, S.; Singh, M.K.: Colour removal from dye wastewater using sugarcane dust as an adsorbent. Adsorpt. Sci. Technol. 17, 269–282 (1999). https://doi.org/10.1177/026361749901700404
Nigam, K.; Mccallum, A.K.; Thrun, S.; Mitchell, T.: Text classification from labeled and unlabeled documents using EM. Mach. Learn. 39, 103–134 (2000). https://doi.org/10.1023/A:1007692713085
Kavitha, D.; Namasivayam, C.: Experimental and kinetic studies on methylene blue adsorption by coir pith carbon. Bioresour. Tech. 98, 14–21 (2007). https://doi.org/10.1016/j.biortech.2005.12.008
Liversidge, R.M.; Lloyd, G.J.; Wase, D.A.J.; Forster, C.F.: Removal of Basic Blue 41 dye from aqueous solution by linseed cake. Process Biochem. 32, 473–477 (1997). https://doi.org/10.1016/S0032-9592(96)00107-0
Tan, I.A.W.; Ahmad, A.L.; Hameed, B.H.: Adsorption of basic dye using activated carbon prepared from oil palm shell: batch and fixed bed studies. Desalination 225, 13–28 (2008). https://doi.org/10.1016/j.desal.2007.07.005
Huang, H.; Yuan, X.; Zeng, G.; Liu, Y.; Li, H.; Yin, J.; Wang, X.: Thermochemical liquefaction of rice husk for bio-oil production with sub- and supercritical ethanol as solvent. J. Anal. Appl. Pyrolysis 102, 60–67 (2013). https://doi.org/10.1016/j.jaap.2013.04.002
Senthilkumaar, S.; Kalaamani, P.; Porkodi, K.; Varadarajan, P.R.; Subburaam, C.V.: Adsorption of dissolved reactive red dye from aqueous phase onto activated carbon prepared from agricultural waste. Bioresour. Technol. 97, 1618–1625 (2006). https://doi.org/10.1016/j.biortech.2005.08.001
Thue, P.S.; Sophia, A.C.; Lima, E.C.; Wamba, A.G.N.; de Alencar, W.S.; dos Reis, G.S.; Rodembusch, F.S.; Dias, S.L.P.: Synthesis and characterization of a novel organic–inorganic hybrid clay adsorbent for the removal of Acid Red 1 and Acid Green 25 from aqueous solutions. J. Clean. Prod. 171, 30–44 (2018). https://doi.org/10.1016/j.jclepro.2017.09.278
Leite, A.J.B.; Lima, E.C.; dos Reis, G.S.; Thue, P.S.; Saucier, C.; Rodembusch, F.S.; Dias, S.L.P.; Umpierres, C.S.; Dotto, G.L.: Hybrid adsorbents of tannin and APTES (3-aminopropyltriethoxysilane) and their application for the highly efficient removal of acid red 1 dye from aqueous solutions. J. Environ. Chem. Eng. 5, 4307–4318 (2017). https://doi.org/10.1016/j.jece.2017.08.022
Kamaraj, R.; Vasudevan, S.: Evaluation of electrocoagulation process for theremoval of strontium and cesium from aqueoussolution. Chem. Eng. Res. Des. 93, 522–530 (2015). https://doi.org/10.1016/j.cherd.2014.03.021
Kamaraj, R.; Ganesan, P.; Vasudevan, S.: Removal of lead from aqueous solutions by electrocoagulation: isotherm, kinetics and thermodynamic studies. Int. J. Environ. Sci. Technol. 12, 683–692 (2015). https://doi.org/10.1007/s13762-013-0457-z
Vasudevan, S.; Lakshmi, J.; Sozhan, G.: Optimization of electrocoagulation process for the simultaneous removal of mercury, lead, and nickel from contaminated water. Environ. Sci. Pollut. Res. 19, 2734–2744 (2012). https://doi.org/10.1007/s11356-012-0773-8
Vasudevan, S.; Lakshmi, J.; Sozhan, G.: Simultaneous removal of Co, Cu, and Cr from water by electrocoagulation. Toxicol. Environ. Chem. 94(10), 1930–1940 (2012). https://doi.org/10.1080/02772248.2012.742898
Igwe, J.C.; Abia, A.A.: A bioseparation process for removing heavy metals from waste water using biosorbents. Afr. J. Biotechnol. 5, 1167–1179 (2006)
Langmuir, I.: the adsorption of gases on plane surfaces of glass, mica and platinum. J. Am. Chem. Soc. 40, 1361–1403 (1918). https://doi.org/10.1021/ja02242a004
Vasudevan, S.; Lakshmi, J.; Packiyam, M.: Electrocoagulation studies on removal of cadmium using magnesium electrode. J. Appl. Electrochem. 40, 2023–2032 (2010). https://doi.org/10.1007/s10800-010-0182-y
Vasudevan, S.; Sozhan, G.; Ravichandran, S.; Jayaraj, J.; Lakshmi, J.; Sheela, S.M.: Studies on the removal of phosphate from drinking water by electrocoagulation process. Ind. Eng. Chem. Res. 47, 2018–2023 (2018). https://doi.org/10.1021/ie0714652
Bedoui, A.; Ahmadi, M.F.; Bensalah, N.; Gadri, A.: Comparative study of Eriochrome black T treatment by BDD-anodic oxidation and Fenton process. Chem. Eng. J. 146, 98–104 (2009). https://doi.org/10.1016/j.cej.2008.05.029
Naushad, M.; Vasudevan, S.; Sharma, G.; Kumar, A.; ALOthman, Z.A.: Adsorption kinetics, isotherms, and thermodynamic studies for Hg2+ adsorption from aqueous medium using alizarin red-S-loaded amberlite IRA-400 resin. Des. Water Treat. (2015). https://doi.org/10.1080/19443994.2015.1090914
Vasudevan, S.; Lakshmi, J.; Sozhan, G.: Effects of alternating and direct current in electrocoagulation process on the removal of cadmium from water. J. Hazard. Mater. 192, 26–34 (2011). https://doi.org/10.1016/j.jhazmat.2011.04.081
Kasperiski, F.M.; Lima, E.C.; dos Reis, G.S.; da Costa, J.B.; Dotto, G.L.; Dias, S.L.P.; Cunha, M.R.; Pavan, F.A.; Correa, C.S.: Preparation of CTAB-functionalized aqai stalk and its efficient application as adsorbent for the removal of Direct Blue 15 and Direct Red 23 dyes from aqueous media. Chem. Eng. Commun. 205(10), 1520–1536 (2018). https://doi.org/10.1080/00986445.2018.1458028
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Islam, T., Peng, C., Ali, I. et al. Synthesis of Rice Husk-Derived Magnetic Biochar Through Liquefaction to Adsorb Anionic and Cationic Dyes from Aqueous Solutions. Arab J Sci Eng 46, 233–246 (2021). https://doi.org/10.1007/s13369-020-04537-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13369-020-04537-z