Abstract
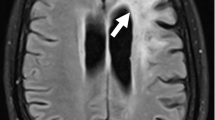
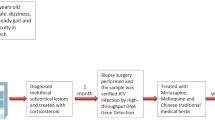
Progressive multifocal leukoencephalopathy (PML) is a demyelinating disease of the central nervous system with a poor prognosis and is primarily caused by JC virus (JCV) with a mutation called prototype. We encountered a case of PML with moderate progression and analyzed the mutational patterns of JCV in the cerebrospinal fluid (CSF). A 19-year-old Japanese woman with mild neurological symptoms was diagnosed with combined immunodeficiency following pneumocystis pneumonia. Brain magnetic resonance imaging scan showed multiple brain lesions, and real-time polymerase chain reaction testing detected JCV in the CSF, leading to the diagnosis of PML. The disease course of PML was stable after administration of mefloquine and mirtazapine with immunoglobulin replacement therapy. In the JCV genome cloned from the patient CSF, DNA sequences of the gene encoding the capsid protein (VP1) and the non-coding control region exhibited small mutations. However, they were quite similar to those of the archetype JCV, which persists asymptomatically in healthy individuals. These findings provide insight into the mutational characteristics of JCV in PML with mild symptoms and progression.


Similar content being viewed by others
References
Cortese I, Reich DS, Nath A (2021) Progressive multifocal leukoencephalopathy and the spectrum of JC virus-related disease. Nat Rev Neurol 17:37–51. https://doi.org/10.1038/s41582-020-00427-y
Ferrante P, Delbue S, Pagani E, Mancuso R, Marzocchetti A, Borghi E, Maserati R, Bestetti A, Cinque P (2003) Analysis of JC virus genotype distribution and transcriptional control region rearrangements in human immunodeficiency virus-positive progressive multifocal leukoencephalopathy patients with and without highly active antiretroviral treatment. J Neurovirol 9(Suppl 1):42–46. https://doi.org/10.1080/13550280390195405
Gorelik L, Reid C, Testa M, Brickelmaier M, Bossolasco S, Pazzi A, Bestetti A, Carmillo P, Wilson E, McAuliffe M, Tonkin C, Carulli JP, Lugovskoy A, Lazzarin A, Sunyaev S, Simon K, Cinque P (2011) Progressive multifocal leukoencephalopathy (PML) development is associated with mutations in JC virus capsid protein VP1 that change its receptor specificity. J Infect Dis 204:103–114. https://doi.org/10.1093/infdis/jir198
Gosert R, Kardas P, Major EO, Hirsch HH (2010) Rearranged JC virus noncoding control regions found in progressive multifocal leukoencephalopathy patient samples increase virus early gene expression and replication rate. J Virol 84:10448–10456. https://doi.org/10.1128/JVI.00614-10
Hadjadj J, Guffroy A, Delavaud C, Taieb G, Meyts I, Fresard A, Streichenberger N, L’Honneur AS, Rozenberg F, D’Aveni M, Aguilar C, Rosain J, Picard C, Mahlaoui N, Lecuit M, Hermine O, Lortholary O, Suarez F (2019) Progressive multifocal leukoencephalopathy in primary immunodeficiencies. J Clin Immunol 39:55–64. https://doi.org/10.1007/s10875-018-0578-8
Iannetta M, Bellizzi A, Lo Menzo S, Anzivino E, D’Abramo A, Oliva A, D’Agostino C, d’Ettorre G, Pietropaolo V, Vullo V, Ciardi MR (2013) HIV-associated progressive multifocal leukoencephalopathy: longitudinal study of JC virus non-coding control region rearrangements and host immunity. J Neurovirol 19:274–279. https://doi.org/10.1007/s13365-013-0167-9
McIlroy D, Halary F, Bressollette-Bodin C (2019) Intra-patient viral evolution in polyomavirus-related diseases. Philos Trans R Soc Lond B Biol Sci 374:20180301. https://doi.org/10.1098/rstb.2018.0301
Nakamichi K, Kishida S, Tanaka K, Suganuma A, Sano Y, Sano H, Kanda T, Maeda N, Kira J, Itoh A, Kato N, Tomimoto H, Kurane I, Lim CK, Mizusawa H, Saijo M (2013) Sequential changes in the non-coding control region sequences of JC polyomaviruses from the cerebrospinal fluid of patients with progressive multifocal leukoencephalopathy. Arch Virol 158:639–650. https://doi.org/10.1007/s00705-012-1532-3
Nakamichi K, Kawamoto M, Ishii J, Saijo M (2019) Improving detection of JC virus by ultrafiltration of cerebrospinal fluid before polymerase chain reaction for the diagnosis of progressive multifocal leukoencephalopathy. BMC Neurol 19:252. https://doi.org/10.1186/s12883-019-1476-2
Pfister LA, Letvin NL, Koralnik IJ (2001) JC virus regulatory region tandem repeats in plasma and central nervous system isolates correlate with poor clinical outcome in patients with progressive multifocal leukoencephalopathy. J Virol 75:5672–5676. https://doi.org/10.1128/JVI.75.12.5672-5676.2001
Reid CE, Li H, Sur G, Carmillo P, Bushnell S, Tizard R, McAuliffe M, Tonkin C, Simon K, Goelz S, Cinque P, Gorelik L, Carulli JP (2011) Sequencing and analysis of JC virus DNA from natalizumab-treated PML patients. J Infect Dis 204:237–244. https://doi.org/10.1093/infdis/jir256
Seppälä H, Virtanen E, Saarela M, Laine P, Paulín L, Mannonen L, Auvinen P, Auvinen E (2017) Single-molecule sequencing revealing the presence of distinct JC polyomavirus populations in patients with progressive multifocal leukoencephalopathy. J Infect Dis 215:889–895. https://doi.org/10.1093/infdis/jiw399
Sugimoto C, Ito D, Tanaka K, Matsuda H, Saito H, Sakai H, Fujihara K, Itoyama Y, Yamada T, Kira J, Matsumoto R, Mori M, Nagashima K, Yogo Y (1998) Amplification of JC virus regulatory DNA sequences from cerebrospinal fluid: diagnostic value for progressive multifocal leukoencephalopathy. Arch Virol 143:249–262. https://doi.org/10.1007/s007050050284
Sunyaev SR, Lugovskoy A, Simon K, Gorelik L (2009) Adaptive mutations in the JC virus protein capsid are associated with progressive multifocal leukoencephalopathy (PML). PLoS Genet 5:e1000368. https://doi.org/10.1371/journal.pgen.1000368
Thompson AJ, Banwell BL, Barkhof F, Carroll WM, Coetzee T, Comi G, Correale J, Fazekas F, Filippi M, Freedman MS, Fujihara K, Galetta SL, Hartung HP, Kappos L, Lublin FD, Marrie RA, Miller AE, Miller DH, Montalban X, Mowry EM, Sorensen PS, Tintoré M, Traboulsee AL, Trojano M, Uitdehaag BMJ, Vukusic S, Waubant E, Weinshenker BG, Reingold SC, Cohen JA (2018) Diagnosis of multiple sclerosis: 2017 revisions of the McDonald criteria. Lancet Neurol 17:162–173. https://doi.org/10.1016/S1474-4422(17)30470-2
Yogo Y, Kitamura T, Sugimoto C, Ueki T, Aso Y, Hara K, Taguchi F (1990) Isolation of a possible archetypal JC virus DNA sequence from nonimmunocompromised individuals. J Virol 64:3139–3143. https://doi.org/10.1128/JVI.64.6.3139-3143.1990
Acknowledgements
The authors would like to thank the patient in this study.
Funding
This work was supported by the Research Committee of Prion Disease and Slow Virus Infection, Research on Policy Planning and Evaluation for Rare and Intractable Diseases, Health and Labour Sciences Research Grants, The Ministry of Health, Labour and Welfare, Japan, and by JSPS KAKENHI (Grant Number 21K07450). We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Contributions
Kosuke Iwami, Masaaki Matsushima, Azusa Nagai, Shinichi Shirai, Sho Nakakubo, Ikuko Takahashi-Iwata, Masafumi Yamada, and Ichiro Yabe collected and interpreted the clinical data. Kazuo Nakamichi completed virological analyses. Kosuke Iwami and Kazuo Nakamichi wrote the manuscript and prepared the figures. Kosuke Iwami and Kazuo Nakamichi contributed equally to this work. All authors have critically revised and approved the manuscript.
Corresponding authors
Ethics declarations
Informed consent
The CSF was collected for clinical care, and written informed consent was obtained from the patient and her family for the use of the specimen for research purposes. The study was performed in accordance with the ethical standards of the Declaration of Helsinki after approval from the research institution.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Iwami, K., Nakamichi, K., Matsushima, M. et al. Progressive multifocal leukoencephalopathy with mild clinical conditions and detection of archetype-like JC virus in cerebrospinal fluid. J. Neurovirol. 27, 917–922 (2021). https://doi.org/10.1007/s13365-021-01017-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-021-01017-4