Abstract
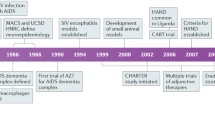
HIV-associated neurocognitive disorder (HAND) remains a common cause of cognitive impairment and persists in 15–55% of HIV+ individuals in the combination antiretroviral therapy (CART) era. CART is now the primary treatment for HAND, but it is effective in only a subset of patients. In the pre-CART era, HIV-associated dementia was the most common form of HAND. However, in CART-treated patients, the prevalence of HIV-associated dementia has declined substantially, and milder stages of HAND, i.e., ANI and MND predominate. HIV+ patients with mild neurocognitive disorder (MND) can still have significant functional impairment in some activities of daily living. There have been several other significant changes in the clinical features of HAND in the CART era. The mean survival for an individual diagnosed with HIV dementia has increased dramatically. In HIV+ individuals on CART with a suppressed systemic viral load, the majority of individuals with HAND remain stable, with a small proportion showing deterioration. Extrapyramidal signs are now less common in patients with HAND on CART. In the CART era, HAND may have a mixed pattern of both cortical and subcortical features with greater deficits in executive functioning and working memory. Despite the milder clinical phenotype, in the CART era, patients with HAND still have persistent laboratory and neuroimaging abnormalities in the central nervous system even with systemic viral suppression. As the HIV+ patient population ages, cerebrovascular disease risk factors such as hypertension, diabetes, and hypercholesterolemia are increasingly recognized as risk factors for cognitive impairment in HIV+ patients on CART. HAND remains a common neurological condition globally in the CART era, necessitating the need for new animal models to examine pathogenesis and potential treatments for HAND.
Similar content being viewed by others
References
Akolo C, Royal W 3rd, Cherner M, Okwuasaba K, Eyzaguirre L, Adebiyi R et al (2014) Neurocognitive impairment associated with predominantly early stage HIV infection in Abuja, Nigeria. J Neurovirol 20(4):380–387
Antinori A, Arendt G, Becker JT, Brew BJ, Byrd DA, Cherner M et al (2007) Updated research nosology for HIV-associated neurocognitive disorders. Neurology 69(18):1789–1799
Bandaru VV, McArthur JC, Sacktor N, Cutler RG, Knapp EL, Mattson MP et al (2007) Associative and predictive biomarkers of dementia in HIV-1-infected patients. Neurology 68(18):1481–1487
Becker JT, Kingsley L, Mullen J, Cohen B, Martin E, Miller EN et al (2009) Vascular risk factors, HIV serostatus, and cognitive dysfunction in gay and bisexual men. Neurology 73(16):1292–1299
Becker JT, Sanders J, Madsen SK, Ragin A, Kingsley L, Maruca V et al (2011) Subcortical brain atrophy persists even in HAART-regulated HIV disease. Brain Imaging Behav 5(2):77–85
Becker J, Maruca V, Kingsley L, Sanders J, Alger JR, Barker PB et al (2012) Factors affecting brain structure in men with disease in the post-HAART era. Neuroradiology 2:113–121
Carey CL, Woods SP, Gonzalez R, Conover E, Marcotte TD, Grant I et al (2004) Predictive validity of global deficit scores in detecting neuropsychological impairment in HIV infection. J Clin Exp Neuropsychol 26(3):307–319
Cassol E, Misra V, Dutta A, Morgello S, Gabuzda D (2014) Cerebrospinal fluid metabolomics reveals altered waste clearance and accelerated aging in HIV patients with neurocognitive impairment. AIDS (London, England) 28(11):1579–1591
Childs EA, Lyles RH, Selnes OA, Chen B, Miller EN, Cohen BA et al (1999) Plasma viral load and CD4 lymphocytes predict HIV-associated dementia and sensory neuropathy. Neurology 52(3):607–613
Dickens AM, Anthony DC, Deutsch R, Mielke MM, Claridge TD, Grant I et al (2015) Cerebrospinal fluid metabolomics implicate bioenergetic adaptation as a neural mechanism regulating shifts in cognitive states of HIV-infected patients. AIDS (London, England) 29(5):559–569
Dore GJ, McDonald A, Li Y, Kaldor JM, Brew BJ (2003) Marked improvement in survival following AIDS dementia complex in the era of highly active antiretroviral therapy. AIDS (London, England) 17(10):1539–1545
Grant I, Franklin DR Jr, Deutsch R, Woods SP, Vaida F, Ellis RJ et al (2014) Asymptomatic HIV-associated neurocognitive impairment increases risk for symptomatic decline. Neurology 82(23):2055–2062
Hagberg L, Cinque P, Gisslen M, Brew BJ, Spudich S, Bestetti A et al (2010) Cerebrospinal fluid neopterin: an informative biomarker of central nervous system immune activation in HIV-1 infection. AIDS Res Ther 7:15
Heaton RK, Franklin DR, Ellis RJ, McCutchan JA, Letendre SL, Leblanc S et al (2011) HIV-associated neurocognitive disorders before and during the era of combination antiretroviral therapy: differences in rates, nature, and predictors. J Neurovirol 17(1):3–16
Jessen Krut J, Mellberg T, Price RW, Hagberg L, Fuchs D, Rosengren L et al (2014) Biomarker evidence of axonal injury in neuroasymptomatic HIV-1 patients. PLoS One 9(2):e88591
Joska JA, Westgarth-Taylor J, Myer L, Hoare J, Thomas KG, Combrinck M et al (2011) Characterization of HIV-associated neurocognitive disorders among individuals starting antiretroviral therapy in South Africa. AIDS Behav 15(6):1197–1203
Kamat A, Lyons JL, Misra V, Uno H, Morgello S, Singer EJ et al (1999) Monocyte activation markers in cerebrospinal fluid associated with impaired neurocognitive testing in advanced HIV infection. J Acquir Immune Defic Syndr 60(3):234–243
Kanmogne GD, Kuate CT, Cysique LA, Fonsah JY, Eta S, Doh R et al (2010) HIV-associated neurocognitive disorders in sub-Saharan Africa: a pilot study in Cameroon. BMC Neurol 10:60
Kelly CM, van Oosterhout JJ, Ngwalo C, Stewart RC, Benjamin L, Robertson KR et al (2014) HIV associated neurocognitive disorders (HAND) in Malawian adults and effect on adherence to combination anti-retroviral therapy: a cross sectional study. PLoS One 9(6):e98962
Kwasa J, Cettomai D, Lwanya E, Osiemo D, Oyaro P, Birbeck GL et al (2012) Lessons learned developing a diagnostic tool for HIV-associated dementia feasible to implement in resource-limited settings: pilot testing in Kenya. PLoS One 7(3):e32898
Lawler K, Mosepele M, Ratcliffe S, Seloilwe E, Steele K, Nthobatsang R et al (2010) Neurocognitive impairment among HIV-positive individuals in Botswana: a pilot study. J Int AIDS Soc 13:15
Lundgren JD, Babiker AG, Gordin F, Emery S, Grund B, Sharma S et al (2015) Initiation of antiretroviral therapy in early asymptomatic HIV infection. N Engl J Med 373(9):795–807
Luther VP, Wilkin AM (2007) HIV infection in older adults. Clin Geriatr Med 23(3):567–583 vii
Marder SR (2013) First and second generation antipsychotics: translating the results from pragmatic trials into clinical practice. Epidemiol Psychiat Sci 22(3):239–240
McArthur JC, Steiner J, Sacktor N, Nath A (2010) Human immunodeficiency virus-associated neurocognitive disorders: mind the gap. Ann Neurol 67(6):699–714
Mielke MM, Bandaru VV, McArthur JC, Chu M, Haughey NJ (2010) Disturbance in cerebral spinal fluid sphingolipid content is associated with memory impairment in subjects infected with the human immunodeficiency virus. J Neurovirol 16(6):445–456
Robertson KR (2017) Can we afford to wait? ART and the CNS Conference on Retroviruses and Opportunistic Infections (CROI). Conference on Retroviruses and Opportunistic Infections (CROI); Feb. 13, 2017; Seattle, WA
Royal W 3rd, Cherner M, Carr J, Habib AG, Akomolafe A, Abimiku A et al (2012) Clinical features and preliminary studies of virological correlates of neurocognitive impairment among HIV-infected individuals in Nigeria. J Neurovirol 18(3):191–199
Sacktor N (2017) Antiretrovirals Improve HAND Stage in HIV+ patients with subtype D and A in Uganda. Conference on Retroviruses and Opportunistic Infections (CROI); Seattle, WA
Sacktor N, Tarwater PM, Skolasky RL, McArthur JC, Selnes OA, Becker J et al (2001) CSF antiretroviral drug penetrance and the treatment of HIV-associated psychomotor slowing. Neurology 57(3):542–544
Sacktor N, Weiguo Y, Zhou Y, Roosa H, Skolasky R, Wong D, and Mohamed M (2014) Brain Amyloid Deposition is increased in a subset of older HIV+ individuals Conference on Retroviruses and Opportunistic Infections (CROI); Boston MA
Sacktor N, Skolasky RL, Seaberg E, Munro C, Becker JT, Martin E et al (2016) Prevalence of HIV-associated neurocognitive disorders in the Multicenter AIDS Cohort Study. Neurology 86(4):334–340
Saylor D, Sacktor N (2016) Cognitive impairment among older individuals with HIV infection. Curr Geriatr Rep 5(2):63–70
Saylor D, Dickens AM, Sacktor N, Haughey N, Slusher B, Pletnikov M et al (2016) HIV-associated neurocognitive disorder—pathogenesis and prospects for treatment. Nat Rev Neurol 12(5):309
Vago L, Bonetto S, Nebuloni M, Duca P, Carsana L, Zerbi P et al (2002) Pathological findings in the central nervous system of AIDS patients on assumed antiretroviral therapeutic regimens: retrospective study of 1597 autopsies. AIDS (London, England) 16(14):1925–1928
Valcour V, Shikuma C, Shiramizu B (2004) Higher frequency of dementia in older HIV-1 individuals: the Hawaii aging with HIV-1 cohort. Neurology 63:822–827
Vivithanaporn P, Heo G, Gamble J, Krentz HB, Hoke A, Gill MJ et al (2010) Neurologic disease burden in treated HIV/AIDS predicts survival: a population-based study. Neurology 75(13):1150–1158
Wright EJ, Grund B, Robertson K, Brew BJ, Roediger M, Bain MP et al (2010) Cardiovascular risk factors associated with lower baseline cognitive performance in HIV-positive persons. Neurology 75(10):864–873
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that he has no conflict of interest.
Rights and permissions
About this article
Cite this article
Sacktor, N. Changing clinical phenotypes of HIV-associated neurocognitive disorders. J. Neurovirol. 24, 141–145 (2018). https://doi.org/10.1007/s13365-017-0556-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-017-0556-6