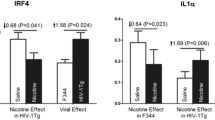
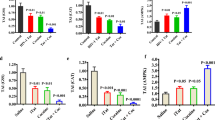
Abstract
Oxidative stress plays an important role in the progression of HIV-1 infection. Nicotine can either protect neurons from neurodegeneration or induce oxidative stress, depending on its dose and degree of oxidative stress impairment. However, the relationship between nicotine and oxidative stress in the HIV-1-infected individuals remains largely unknown. The purpose of this study was to determine the effect of nicotine on expression of genes related to the glutathione (GSH)-centered antioxidant system and oxidative stress in the nucleus accumbens (NAc) and ventral tegmental area (VTA) of HIV-1 transgenic (HIV-1Tg) and F344 control rats. Adult HIV-1Tg and F344 rats received nicotine (0.4 mg/kg, base, s.c.) or saline injections once per day for 27 days. At the end of treatment, various brain regions including the NAc and VTA were collected from each rat. Following total RNA extraction and complementary DNA (cDNA) synthesis of each sample, quantitative reverse transcription PCR (RT-PCR) analysis was performed for 43 oxidative-stress-related genes. Compared with F344 control rats, HIV-1Tg rats showed a significant downregulation of genes involved in ATPase and cyctochrome oxidase at the messenger RNA (mRNA) level in both regions. Further, we found a significant downregulation of Gstm5 in the NAc and upregulation of Cox1, Cox3, and Gsta6 in the VTA of HIV-1Tg rats. HIV-1Tg rats showed brain-region-specific responses to chronic nicotine treatment. This response resulted in a change in the expression of genes involved in antioxidant mechanisms including the downregulation of genes such as Atp5h, Calml1, Gpx7, Gstm5, Gsr, and Gsta6 and upregulation of Sod1 in the NAc, as well as downregulation of genes like Cox5a, Gpx4, Gpx6, Gpx7, Gstm5, and Sod1 in the VTA of HIV-1Tg rats. Together, we conclude that chronic nicotine treatment has a dual effect on the antioxidant defense system and oxidative-stress-induced apoptosis signaling in HIV-1Tg rats. These findings suggest that nicotine has a negative effect on response to oxidative stress and antioxidant processes in HIV-1 Tg rat brain, especially in the VTA.

Similar content being viewed by others
References
Aksenov MY, Hasselrot U, Bansal AK, Wu G, Nath A, Anderson C, Mactutus CF, Booze RM (2001) Oxidative damage induced by the injection of HIV-1 tat protein in the rat striatum. Neurosci Lett 305:5–8
Boven LA, Gomes L, Hery C, Gray F, Verhoef J, Portegies P, Tardieu M, Nottet HS (1999) Increased peroxynitrite activity in AIDS dementia complex: implications for the neuropathogenesis of HIV-1 infection. J Immunol 162:4319–4327
Chaban Y, Boekema EJ, Dudkina NV (2014) Structures of mitochondrial oxidative phosphorylation supercomplexes and mechanisms for their stabilisation. Biochim Biophys Acta Bioenerg 1837:418–426
Churchill MJ, Cowley DJ, Wesselingh SL, Gorry PR, Gray LR (2014) HIV-1 transcriptional regulation in the central nervous system and implications for HIV cure research. J Neurovirol
Cui WY, Wang J, Wei J, Cao J, Chang SL, Gu J, Li MD (2012) Modulation of innate immune-related pathways in nicotine-treated SH-SY5Y cells. Amino Acids 43:1157–1169
Fratiglioni L, Wang HX (2000) Smoking and Parkinson’s and Alzheimer’s disease: review of the epidemiological studies. Behav Brain Res 113:117–120
Gallo C, Renzi P, Loizzo S, Loizzo A, Piacente S, Festa M, Caputo M, Tecce MF, Capasso A (2010) Potential therapeutic effects of vitamin e and C on placental oxidative stress induced by nicotine: an in vitro evidence. Open Biochem J 4:77–82
Guan ZZ, Nordberg A, Mousavi M, Rinne JO, Hellstrom-Lindahl E (2002) Selective changes in the levels of nicotinic acetylcholine receptor protein and of corresponding mRNA species in the brains of patients with Parkinson’s disease. Brain Res 956:358–366
Kaul M, Zheng J, Okamoto S, Gendelman HE, Lipton SA (2005) HIV-1 infection and AIDS: consequences for the central nervous system. Cell Death Differ 12(Suppl 1):878–892
Kihara T, Shimohama S, Sawada H, Kimura J, Kume T, Kochiyama H, Maeda T, Akaike A (1997) Nicotinic receptor stimulation protects neurons against beta-amyloid toxicity. Ann Neurol 42:159–163
Kimura T, Kameoka M, Ikuta K (1993) Amplification of superoxide anion generation in phagocytic cells by HIV-1 infection. FEBS Lett 326:232–236
Knott V, Shah D, Millar A, McIntosh J, Fisher D, Blais C, Ilivitsky V (2012) Nicotine, auditory sensory memory, and sustained attention in a human ketamine model of schizophrenia: moderating influence of a hallucinatory trait. Front Pharmacol 3:172
Ladha JS, Tripathy MK, Mitra D (2005) Mitochondrial complex I activity is impaired during HIV-1-induced T-cell apoptosis. Cell Death Differ 12:1417–1428
Lee HM, Hallberg LM, Greeley GH Jr, Englander EW (2010) Differential inhibition of mitochondrial respiratory complexes by inhalation of combustion smoke and carbon monoxide, in vivo, in the rat brain. Inhal Toxicol 22:770–777
Levin ED, Hao I, Burke DA, Cauley M, Hall BJ, Rezvani AH (2014) Effects of tobacco smoke constituents, anabasine and anatabine, on memory and attention in female rats. J Psychopharmacol 28:915–922
Linert W, Bridge MH, Huber M, Bjugstad KB, Grossman S, Arendash GW (1999) In vitro and in vivo studies investigating possible antioxidant actions of nicotine: relevance to Parkinson’s and Alzheimer’s diseases. Biochim Biophys Acta 1454:143–152
Louboutin JP, Agrawal L, Reyes BA, van Bockstaele EJ, Strayer DS (2012) Gene delivery of antioxidant enzymes inhibits human immunodeficiency virus type 1 gp120-induced expression of caspases. Neuroscience 214:68–77
Marshall MM, Kirk GD, Caporaso NE, McCormack MC, Merlo CA, Hague JC, Mehta SH, Engels EA (2011) Tobacco use and nicotine dependence among HIV-infected and uninfected injection drug users. Addict Behav 36:61–67
Matta SG, Balfour DJ, Benowitz NL, Boyd RT, Buccafusco JJ, Caggiula AR, Craig CR, Collins AC, Damaj MI, Donny EC, Gardiner PS, Grady SR, Heberlein U, Leonard SS, Levin ED, Lukas RJ, Markou A, Marks MJ, McCallum SE, Parameswaran N, Perkins KA, Picciotto MR, Quik M, Rose JE, Rothenfluh A, Schafer WR, Stolerman IP, Tyndale RF, Wehner JM, Zirger JM (2007) Guidelines on nicotine dose selection for in vivo research. Psychopharmacology (Berlin) 190:269–319
Menon NK (2001a) Neuropathy associated with norplant. J Fam Plann Reprod Health Care 27:241
Menon NK (2001b) Single-handed practice—the reality. Br J Gen Pract 51:1014–1015
Miro O, Lopez S, Martinez E, Pedrol E, Milinkovic A, Deig E, Garrabou G, Casademont J, Gatell JM, Cardellach F (2004) Mitochondrial effects of HIV infection on the peripheral blood mononuclear cells of HIV-infected patients who were never treated with antiretrovirals. Clin Infect Dis 39:710–716
Mollace V, Nottet HS, Clayette P, Turco MC, Muscoli C, Salvemini D, Perno CF (2001) Oxidative stress and neuroAIDS: triggers, modulators and novel antioxidants. Trends Neurosci 24:411–416
Nath A, Anderson C, Jones M, Maragos W, Booze R, Mactutus C, Bell J, Hauser KF, Mattson M (2000) Neurotoxicity and dysfunction of dopaminergic systems associated with AIDS dementia. J Psychopharmacol 14:222–227
Navarro A, Boveris A (2010) Brain mitochondrial dysfunction in aging, neurodegeneration, and Parkinson’s disease. Front Aging Neurosci 2
Nesil T, Cao J, Yang Z, Chang SL, Li MD (2015) Nicotine attenuates the effect of HIV-1 proteins on the neural circuits of working and contextual memories. Mol Brain 8:43
Nie H, Wang Z, Zhao W, Lu J, Zhang C, Lok K, Wang Y, Shen H, Xu Z, Yin M (2013) New nicotinic analogue ZY-1 enhances cognitive functions in a transgenic mice model of Alzheimer’s disease. Neurosci Lett 537:29–34
Nordberg A, Hellstrom-Lindahl E, Lee M, Johnson M, Mousavi M, Hall R, Perry E, Bednar I, Court J (2002) Chronic nicotine treatment reduces beta-amyloidosis in the brain of a mouse model of Alzheimer’s disease (APPsw). J Neurochem 81:655–658
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates, 4th edn. New York, Academic Press
Perez-Matute P, Perez-Martinez L, Blanco JR, Oteo JA (2013) Role of mitochondria in HIV infection and associated metabolic disorders: focus on nonalcoholic fatty liver disease and lipodystrophy syndrome. Oxidative Med Cell Longev 2013:493413
Picciotto MR, Zoli M (2008) Neuroprotection via nAChRs: the role of nAChRs in neurodegenerative disorders such as Alzheimer’s and Parkinson’s disease. Front Biosci 13:492–504
Price TO, Ercal N, Nakaoke R, Banks WA (2005) HIV-1 viral proteins gp120 and Tat induce oxidative stress in brain endothelial cells. Brain Res 1045:57–63
Quik M, Bordia T, O’Leary K (2007) Nicotinic receptors as CNS targets for Parkinson’s disease. Biochem Pharmacol 74:1224–1234
Ravikumar R, Flora G, Geddes JW, Hennig B, Toborek M (2004) Nicotine attenuates oxidative stress, activation of redox-regulated transcription factors and induction of proinflammatory genes in compressive spinal cord trauma. Brain Res Mol Brain Res 124:188–198
Sener G, Kapucu C, Paskaloglu K, Ayanoglu-Dulger G, Arbak S, Ersoy Y, Alican I (2004) Melatonin reverses urinary system and aorta damage in the rat due to chronic nicotine administration. J Pharm Pharmacol 56:359–366
Sener G, Ozer Sehirli A, Ipci Y, Cetinel S, Cikler E, Gedik N, Alican I (2005) Taurine treatment protects against chronic nicotine-induced oxidative changes. Fundam Clin Pharmacol 19:155–164
Soto-Otero R, Mendez-Alvarez E, Hermida-Ameijeiras A, Lopez-Real AM, Labandeira-Garcia JL (2002) Effects of (−)-nicotine and (−)-cotinine on 6-hydroxydopamine-induced oxidative stress and neurotoxicity: relevance for Parkinson’s disease. Biochem Pharmacol 64:125–135
Stack EC, Ferro JL, Kim J, Del Signore SJ, Goodrich S, Matson S, Hunt BB, Cormier K, Smith K, Matson WR, Ryu H, Ferrante RJ (2008) Therapeutic attenuation of mitochondrial dysfunction and oxidative stress in neurotoxin models of Parkinson’s disease. Biochim Biophys Acta 1782:151–162
Subramaniam SR, Chesselet MF (2013) Mitochondrial dysfunction and oxidative stress in Parkinson’s disease. Prog Neurobiol 106–107:17–32
Tolnay M, Spillantini MG, Goedert M, Ulrich J, Langui D, Probst A (1997) Argyrophilic grain disease: widespread hyperphosphorylation of tau protein in limbic neurons. Acta Neuropathol 93:477–484
Tripathy MK, Mitra D (2010) Differential modulation of mitochondrial OXPHOS system during HIV-1 induced T-cell apoptosis: up regulation of complex-IV subunit COX-II and its possible implications. Apoptosis 15:28–40
Uttara B, Singh AV, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7:65–74
VanCott TC, Mascola JR, Kaminski RW, Kalyanaraman V, Hallberg PL, Burnett PR, Ulrich JT, Rechtman DJ, Birx DL (1997) Antibodies with specificity to native gp120 and neutralization activity against primary human immunodeficiency virus type 1 isolates elicited by immunization with oligomeric gp160. J Virol 71:4319–4330
Wang J, Kim JM, Donovan DM, Becker KG, Li MD (2009) Significant modulation of mitochondrial electron transport system by nicotine in various rat brain regions. Mitochondrion 9:186–195
Wang J, Cui W, Wei J, Sun D, Gutala R, Gu J, Li MD (2011) Genome-wide expression analysis reveals diverse effects of acute nicotine exposure on neuronal function-related genes and pathways. Front Psychiatry 2:5
Wei J, Wang J, Dwyer JB, Mangold J, Cao J, Leslie FM, Li MD (2011) Gestational nicotine treatment modulates cell death/survival-related pathways in the brains of adolescent female rats. Int J Neuropsychopharmacol 14:91–106
Winer J, Jung CK, Shackel I, Williams PM (1999) Development and validation of real-time quantitative reverse transcriptase-polymerase chain reaction for monitoring gene expression in cardiac myocytes in vitro. Anal Biochem 270:41–49
Yildiz D, Ercal N, Armstrong DW (1998) Nicotine enantiomers and oxidative stress. Toxicology 130:155–165
Zeng H, Zhang Y, Peng L, Shao H, Menon NK, Yang J, Salomon AR, Freidland RP, Zagorski MG (2001) Nicotine and amyloid formation. Biol Psychiatry 49:248–257
Zhang J, Kamdar O, Le W, Rosen GD, Upadhyay D (2009) Nicotine induces resistance to chemotherapy by modulating mitochondrial signaling in lung cancer. Am J Respir Cell Mol Biol 40:135–146
Zhao SF, Cui WY, Cao JR, Luo C, Fan LJ, Li MD (2014) Impact of maternal nicotine exposure on expression of myelin-related genes in zebrafish larvae. Zebrafish 11:10–16
Acknowledgments
The project was supported, in part, by US National Institutes of Health grants DA-012844 to MDL, DA-016149 to SLC, and DA-026356 to SLC and MDL.
Conflict of interest
The authors declare no conflict of interest with the study or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, G., Nesil, T., Cao, J. et al. Nicotine mediates expression of genes related to antioxidant capacity and oxidative stress response in HIV-1 transgenic rat brain. J. Neurovirol. 22, 114–124 (2016). https://doi.org/10.1007/s13365-015-0375-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13365-015-0375-6